Ann Lab Med.
2017 May;37(3):248-253. 10.3343/alm.2017.37.3.248.
Comparison of the QIAGEN artus HBV QS-RGQ Assay With the Roche COBAS AmpliPrep/COBAS TaqMan HBV Assay for Quantifying Viral DNA in Sera of Chronic Hepatitis B Patients
- Affiliations
-
- 1Medical Clinic Laboratory Department of U2Bio Co. Ltd., Seoul, Korea.
- 2Department of Laboratory Medicine, National Health Insurance Service Ilsan Hospital, Goyang, Korea.
- 3Department of Laboratory Medicine, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea. kimhs54@yuhs.ac
- KMID: 2369750
- DOI: http://doi.org/10.3343/alm.2017.37.3.248
Abstract
- BACKGROUND
Hepatitis B virus DNA quantification is essential for managing chronic hepatitis B (CHB). We compared the performance of artus HBV QS-RGQ (QIAGEN GmbH, Germany) and CAP/CTM v2.0 HBV assays (Roche Molecular Diagnostics, USA) in CHB patients.
METHODS
A comparative evaluation between two assays was performed with 508 clinical serum samples. Precision, linearity, and the limit of detection (LOD) of QS-RGQ assay was evaluated by using the WHO standard 97/750 and clinical samples.
RESULTS
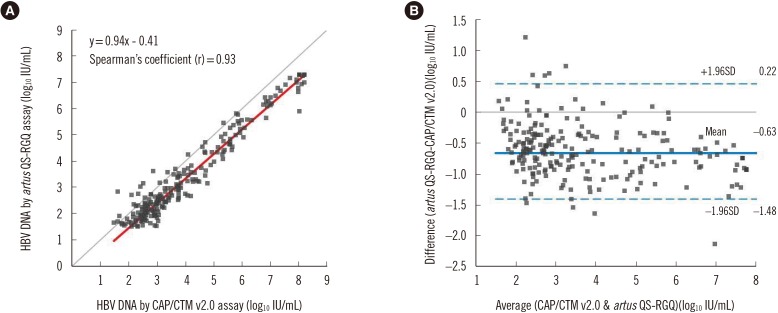
Detection rates and viral loads as determined QS-RGQ assay were significantly lower than those from the CAP/CTM v2.0 assay (52.8% vs 60.6%; 3.55±1.77 IU/mL vs 4.18±1.89 IU/mL, P<0.0001). The kappa coefficient between qualitative results was 0.79 (95% confidence interval, 0.74 to 0.85). Bland-Altman plot found a mean difference of (QS-RGQ − CAP/CTM v2.0)=−0.63 logâ‚â‚€ IU/mL (95% limit of agreement, −1.48 to 0.22). Repeatability and total imprecision (% CV) of the QS-RGQ assay were 1.0% and 1.1% at 2,000 IU/mL, and 0.7% and 1.4% at 20,000 IU/mL, respectively. Linearity of this assay ranged from 31.6 to 1.0±10â· IU/mL, and the LOD was 2.95 IU/mL.
CONCLUSIONS
The artus HBV QS-RGQ assay showed good performance but significantly decreased detection rate and viral load compared with CAP/CTM v2.0 assays. This assay recommends using plasma; however, we used stored serum because of the retrospective study design. Usually HBV DNA quantification is performed in plasma or serum, but sample type and clinical relevance of quantitative values should be considered when determining the clinical application of this reagent.
Keyword
MeSH Terms
Figure
Reference
-
1. World Health Organization. Hepatitis B. Updated on Jul 2016. http://www.who.int/mediacentre/factsheets/fs204/en/.2. Oakes K. Hepatitis B: prevalence and pathophysiology. Nurs Times. 2014; 110:12–16.3. Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009; 50:661–662. PMID: 19714720.4. Martin P, Lau DT, Nguyen MH, Janssen HL, Dieterich DT, Peters MG, et al. A treatment algorithm for the management of chronic hepatitis B virus infection in the United States: 2015 Update. Clin Gastroenterol Hepatol. 2015; 13:2071–2087. PMID: 26188135.5. González R, Torres P, Castro E, Barbolla L, Candotti D, Koppelman M, et al. Efficacy of hepatitis B virus (HBV) DNA screening and characterization of acute and occult HBV infections among blood donors from Madrid, Spain. Transfusion. 2010; 50:221–230. PMID: 19682332.6. Jiang JN, Huang ZL, He LX, Huang YH, Su MH, Xie R, et al. Residual amount of HBV DNA in serum is related to relapse in chronic hepatitis B patients after cessation of nucleos(t)ide analogs. J Clin Gastroenterol. 2015; 49:323–328. PMID: 25014234.7. Stelzl E, Muller Z, Marth E, Kessler HH. Rapid quantification of hepatitis B virus DNA by automated sample preparation and real-time PCR. J Clin Microbiol. 2004; 42:2445–2449. PMID: 15184417.8. Poljak M, Lepej SZ, Rode OD. Recent developments in serologic and molecular diagnosis of hepatitis B and C. Acta medica Croatica. 2013; 67:281–290. PMID: 24984327.9. Ismail AM, Sivakumar J, Anantharam R, Dayalan S, Samuel P, Fletcher GJ, et al. Performance characteristics and comparison of Abbott and artus real-time systems for hepatitis B virus DNA quantification. J Clin Microbiol. 2011; 49:3215–3221. PMID: 21795507.10. Yeh ML, Huang CF, Hsieh MY, Huang JF, Dai CY, Yu ML, et al. Comparison of the Abbott RealTime HBV assay with the Roche Cobas AmpliPrep/Cobas TaqMan HBV assay for HBV DNA detection and quantification. J Clin Virol. 2014; 60:206–214. PMID: 24809730.11. Brichler S, Dziri S, Duhant A, Faleu G, Gordien E. P0543: Comparative evaluation of the artus HBV QS-RGQ assay and the roche cobas mpliprep/cobas taqman V.2 HBV test for the quantification of HBV DNA in plasma. J Hepatol. 2015; 62:S518–S519.12. Fielder H, Javed S, Marques C, Wall G, Lange C, Tomlinson D. Quantitative detection of hepatitis B virus (HBV) DNA with the novel artus® HBV QS-RGQ Kit version 2. J Clin Virol. 2015; 70:S85.13. Allice T, Cerutti F, Pittaluga F, Varetto S, Gabella S, Marzano A, et al. COBAS AmpliPrep-COBAS TaqMan hepatitis B virus (HBV) test: a novel automated real-time PCR assay for quantification of HBV DNA in plasma. J Clin Microbiol. 2007; 45:828–834. PMID: 17229858.14. Chevaliez S, Bouvier-Alias M, Laperche S, Hézode C, Pawlotsky JM. Performance of version 2.0 of the Cobas AmpliPrep/Cobas TaqMan real-time PCR assay for hepatitis B virus DNA quantification. J Clin Microbiol. 2010; 48:3641–3647. PMID: 20720031.15. Caliendo AM, Valsamakis A, Bremer JW, Ferreira-Gonzalez A, Granger S, Sabatini L, et al. Multilaboratory evaluation of real-time PCR tests for hepatitis B virus DNA quantification. J Clin Microbiol. 2011; 49:2854–2858. PMID: 21697326.16. Lok AS. Natural history and control of perinatally acquired hepatitis B virus infection. Dig Dis. 1992; 10:46–52.17. Funk ML, Rosenberg DM, Lok AS. World-wide epidemiology of HBeAg-negative chronic hepatitis B and associated precore and core promoter variants. J Viral Hepat. 2002; 9:52–61. PMID: 11851903.18. Lindh M, Hannoun C, Malmström S, Lindberg J, Norkrans G. Lamivudine resistance of hepatitis B virus masked by coemergence of mutations in probe region of the COBAS AMPLICOR assay. J Clin Microbiol. 2006; 44:2587–2589. PMID: 16825388.19. Yeh ML, Huang CF, Huang CI, Liu SF, Yang HL, Hsieh MY, et al. Abbott RealTime HBV assay is more sensitive in detection of low viral load and little impacted by drug resistant mutation in chronic hepatitis B patients under nucleot(s)ide analogues therapy. PLoS One. 2014; 9:e101790. PMID: 25000502.20. Alestig E, Hannoun C, Horal P, Lindh M. Phylogenetic origin of hepatitis B virus strains with precore C-1858 variant. J Clin Microbiol. 2001; 39:3200–3203. PMID: 11526151.21. Alexopoulou A, Karayiannis P. HBeAg negative variants and their role in the natural history of chronic hepatitis B virus infection. World J Gastroenterol. 2014; 20:7644–7652. PMID: 24976702.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Performance of Cobas TaqMan MTB for Detection of Mycobacterium tuberculosis Complex in Respiratory Specimens
- Performance Evaluation of Aptima HBV and HCV Quant Assays in the Panther System
- Long-Term Stability of HBV, HCV, and HIV-1 National Reference Standards for in vitro Diagnostic Medical Devices Intended to Be Used for the Nucleic Acid Amplification Test
- HBV DNA Quantitation Using Real-time PCR
- Comparison of COBAS AmpliPrep/COBAS TaqMan HCV Qualitative Test v2.0 with COBAS AMPLICOR Hepatitis C Virus Test v2.0 for the Qualitative Detection of Hepatitis C Virus RNA in Korean Clinical Samples