Cancer Res Treat.
2017 Jan;49(1):263-273. 10.4143/crt.2016.054.
Adjuvant Chemotherapy for Advanced Gastric Cancer in Elderly and Non-elderly Patients: Meta-Analysis of Randomized Controlled Trials
- Affiliations
-
- 1Department of Surgery, Konkuk University School of Medicine, Seoul, Korea.
- 2Department of Obstetrics and Gynecology, Konkuk University School of Medicine, Seoul, Korea.
- 3Department of Internal Medicine, Institute of Health Sciences, Gyeongsang National University School of Medicine, Jinju, Korea. lwshmo@hanmail.net
- 4Medical Library, Institute of Health Sciences, Gyeongsang National University School of Medicine, Jinju, Korea.
- 5Department of Preventive Medicine and Environmental Health, Institute of Health Sciences, Gyeongsang National University School of Medicine, Jinju, Korea.
- KMID: 2367524
- DOI: http://doi.org/10.4143/crt.2016.054
Abstract
- PURPOSE
This study evaluated the benefits of adjuvant chemotherapy on elderly patients with advanced gastric cancer (AGC) using meta-analysis of well-designed randomized controlled clinical studies.
MATERIALS AND METHODS
PubMed, Embase, and Cochrane were searched to retrieve clinical studies evaluating the benefits of adjuvant chemotherapy in the elderly with AGC. Hazards ratios (HRs) with 95% confidence intervals (CIs) were pooled across studies using a fixed-effects model.
RESULTS
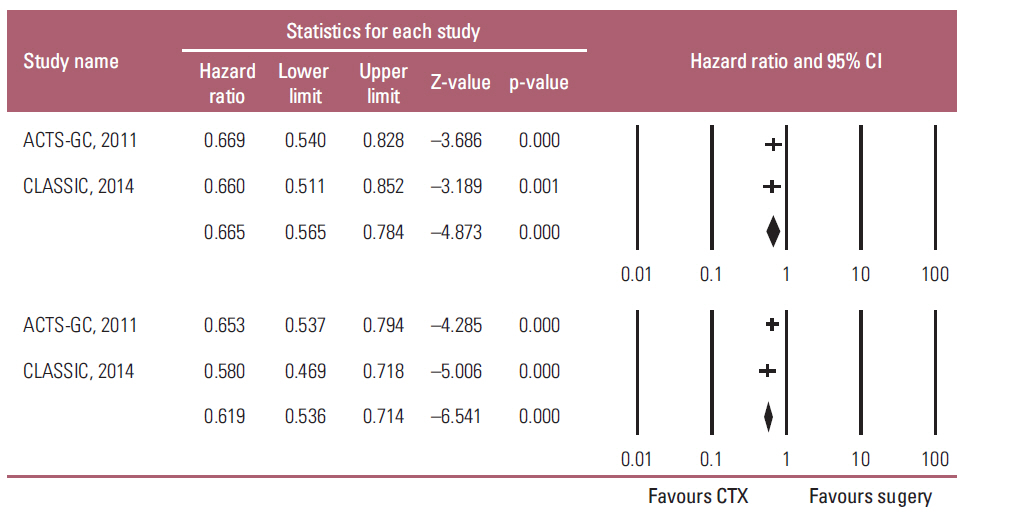
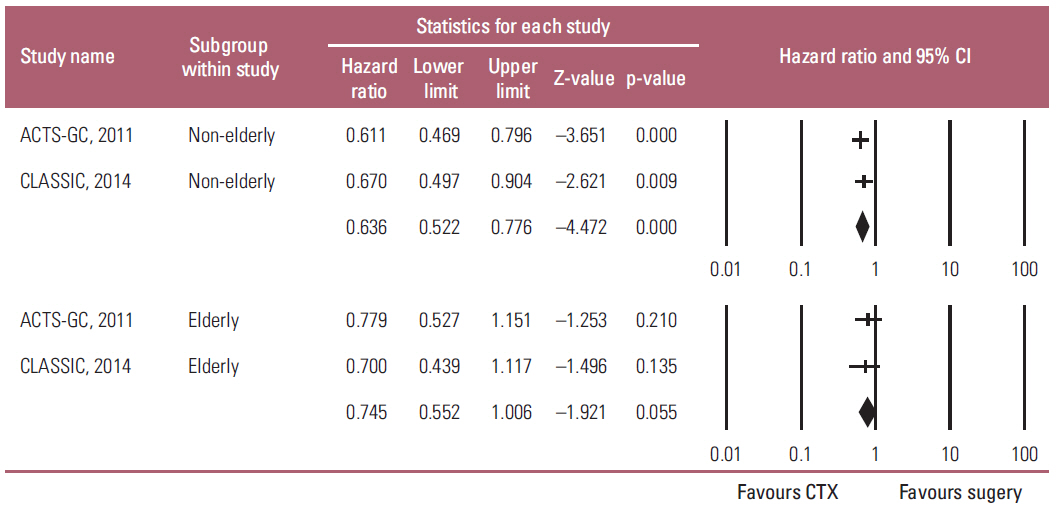
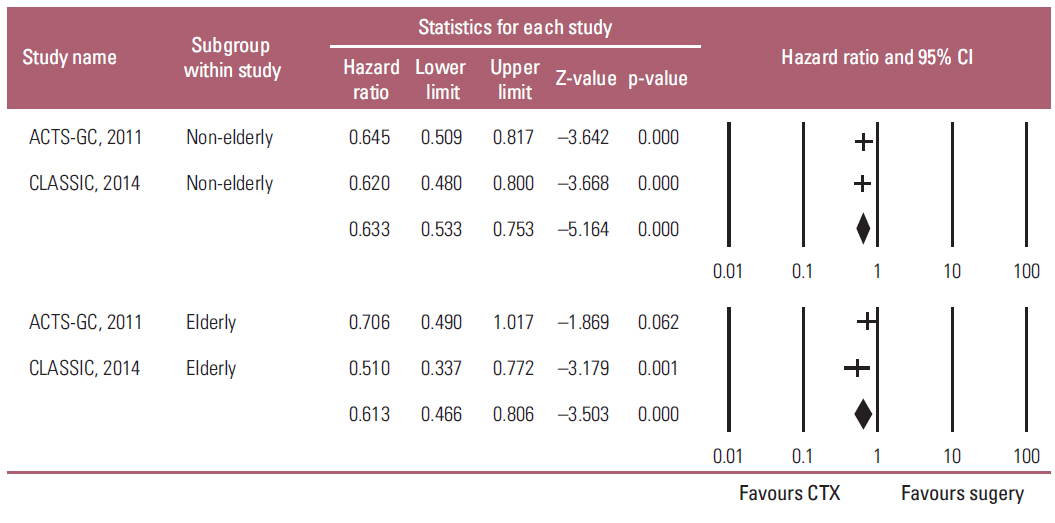
Two studies were included in this meta-analysis to estimate HR for the overall survival (OS), and relapse-free survival (RFS) between adjuvant chemotherapy and surgery in elderly and non-elderly patients. HR for OS in the elderly and non-elderly was 0.745 (95% CI, 0.552 to 1.006, p=0.055) and 0.636 (95% CI, 0.522 to 0.776; p < 0.001), respectively, which showed no heterogeneity regarding HR between the two groups (p(interaction)=0.389). HR for RFS in the elderly and non-elderly was 0.613 (95% CI, 0.466 to 0.806; p < 0.001) and 0.633 (95% CI, 0.533 to 0.753; p < 0.001), respectively (p(interaction)=0.846).
CONCLUSION
Meta-analysis suggests that the benefit of adjuvant chemotherapy to the elderly is not big enough to reach statistical significance while the HR for OS is less than 1 (0.745) and no heterogeneity are observed regarding the HR between the elderly and non-elderly patients.
Keyword
Figure
Reference
-
References
1. Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG, Lee JS. Cancer statistics in Korea: incidence, mortality, survival and prevalence in 2010. Cancer Res Treat. 2013; 45:1–14.
Article2. Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG, Lee JS. Prediction of cancer incidence and mortality in Korea, 2013. Cancer Res Treat. 2013; 45:15–21.
Article3. Neugut AI, Matasar M, Wang X, McBride R, Jacobson JS, Tsai WY, et al. Duration of adjuvant chemotherapy for colon cancer and survival among the elderly. J Clin Oncol. 2006; 24:2368–75.
Article4. Sakuramoto S, Sasako M, Yamaguchi T, Kinoshita T, Fujii M, Nashimoto A, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med. 2007; 357:1810–20.
Article5. Sasako M, Sakuramoto S, Katai H, Kinoshita T, Furukawa H, Yamaguchi T, et al. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol. 2011; 29:4387–93.
Article6. Noh SH, Park SR, Yang HK, Chung HC, Chung IJ, Kim SW, et al. Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014; 15:1389–96.
Article7. Bang YJ, Kim YW, Yang HK, Chung HC, Park YK, Lee KH, et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a phase 3 open-label, randomised controlled trial. Lancet. 2012; 379:315–21.
Article8. Bouche O, Ychou M, Burtin P, Bedenne L, Ducreux M, Lebreton G, et al. Adjuvant chemotherapy with 5-fluorouracil and cisplatin compared with surgery alone for gastric cancer: 7-year results of the FFCD randomized phase III trial (8801). Ann Oncol. 2005; 16:1488–97.9. Nakajima T, Kinoshita T, Nashimoto A, Sairenji M, Yamaguchi T, Sakamoto J, et al. Randomized controlled trial of adjuvant uracil-tegafur versus surgery alone for serosa-negative, locally advanced gastric cancer. Br J Surg. 2007; 94:1468–76.
Article10. Cascinu S, Labianca R, Barone C, Santoro A, Carnaghi C, Cassano A, et al. Adjuvant treatment of high-risk, radically resected gastric cancer patients with 5-fluorouracil, leucovorin, cisplatin, and epidoxorubicin in a randomized controlled trial. J Natl Cancer Inst. 2007; 99:601–7.
Article11. Fazio N, Biffi R, Curigliano G, Lorizzo K, Zampino MG, de Braud F, et al. Re: Adjuvant treatment of high-risk, radically resected gastric cancer patients with 5-fluorouracil, leucovorin, cisplatin, and epidoxorubicin in a randomized controlled trial. J Natl Cancer Inst. 2007; 99:1345–6.
Article12. De Vita F, Giuliani F, Orditura M, Maiello E, Galizia G, Di Martino N, et al. Adjuvant chemotherapy with epirubicin, leucovorin, 5-fluorouracil and etoposide regimen in resected gastric cancer patients: a randomized phase III trial by the Gruppo Oncologico Italia Meridionale (GOIM 9602 Study). Ann Oncol. 2007; 18:1354–8.
Article13. Paganini-Hill A, Kawas CH, Corrada MM. Activities and mortality in the elderly: the Leisure World cohort study. J Gerontol A Biol Sci Med Sci. 2011; 66:559–67.
Article14. Molineux M. Social and productive activities in elderly people. Activity (occupation) is important for survival. BMJ. 2000; 320:184–5.15. Tournigand C, Andre T, Bonnetain F, Chibaudel B, Lledo G, Hickish T, et al. Adjuvant therapy with fluorouracil and oxaliplatin in stage II and elderly patients (between ages 70 and 75 years) with colon cancer: subgroup analyses of the Multicenter International Study of Oxaliplatin, Fluorouracil, and Leucovorin in the Adjuvant Treatment of Colon Cancer trial. J Clin Oncol. 2012; 30:3353–60.
Article16. Huguenin P, Beer KT, Allal A, Rufibach K, Friedli C, Davis JB, et al. Concomitant cisplatin significantly improves locoregional control in advanced head and neck cancers treated with hyperfractionated radiotherapy. J Clin Oncol. 2004; 22:4665–73.
Article17. Hogberg T, Signorelli M, de Oliveira CF, Fossati R, Lissoni AA, Sorbe B, et al. Sequential adjuvant chemotherapy and radiotherapy in endometrial cancer: results from two randomised studies. Eur J Cancer. 2010; 46:2422–31.18. Inoshita N, Yanagisawa A, Arai T, Kitagawa T, Hirokawa K, Kato Y. Pathological characteristics of gastric carcinomas in the very old. Jpn J Cancer Res. 1998; 89:1087–92.
Article19. Arai T, Esaki Y, Inoshita N, Sawabe M, Kasahara I, Kuroiwa K, et al. Pathologic characteristics of gastric cancer in the elderly: a retrospective study of 994 surgical patients. Gastric Cancer. 2004; 7:154–9.
Article20. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–9.21. Habu H, Endo M. Gastric cancer in elderly patients: results of surgical treatment. Hepatogastroenterology. 1989; 36:71–4.22. Choi JY, Shim KN, Roh SH, Tae CH, Kim SE, Jung HK, et al. Clinicopathological characteristics of gastric cancer and survival improvement by surgical treatment in the elderly. Korean J Gastroenterol. 2011; 58:9–19.
Article23. Kitamura K, Yamaguchi T, Taniguchi H, Hagiwara A, Yamane T, Sawai K, et al. Clinicopathological characteristics of gastric cancer in the elderly. Br J Cancer. 1996; 73:798–802.
Article24. Edelman DS, Russin DJ, Wallack MK. Gastric cancer in the elderly. Am Surg. 1987; 53:170–3.25. Ishigami S, Natsugoe S, Saihara T, Hokita S, Tokushige M, Watanabe T, et al. Clinical and pathologic features of early gastric cancer in elderly patients. Hepatogastroenterology. 1997; 44:1164–8.26. Li LS, Morales JC, Veigl M, Sedwick D, Greer S, Meyers M, et al. DNA mismatch repair (MMR)-dependent 5-fluorouracil cytotoxicity and the potential for new therapeutic targets. Br J Pharmacol. 2009; 158:679–92.
Article27. Carethers JM, Smith EJ, Behling CA, Nguyen L, Tajima A, Doctolero RT, et al. Use of 5-fluorouracil and survival in patients with microsatellite-unstable colorectal cancer. Gastroenterology. 2004; 126:394–401.
Article28. An JY, Kim H, Cheong JH, Hyung WJ, Kim H, Noh SH. Microsatellite instability in sporadic gastric cancer: its prognostic role and guidance for 5-FU based chemotherapy after R0 resection. Int J Cancer. 2012; 131:505–11.
Article29. Zuckerman IH, Rapp T, Onukwugha E, Davidoff A, Choti MA, Gardner J, et al. Effect of age on survival benefit of adjuvant chemotherapy in elderly patients with Stage III colon cancer. J Am Geriatr Soc. 2009; 57:1403–10.
Article