Allergy Asthma Immunol Res.
2015 Mar;7(2):158-166. 10.4168/aair.2015.7.2.158.
Comparison of Intranasal Ciclesonide, Oral Levocetirizine, and Combination Treatment for Allergic Rhinitis
- Affiliations
-
- 1Department of Otorhinolaryngology, Yonsei University College of Medicine, Seoul, Korea. jhyoon@yuhs.ac
- 2Department of Otorhinolaryngology-Head and Neck Surgery, College of Medicine, Konkuk University, Seoul, Korea.
- 3Department of Otolaryngology, Ajou University School of Medicine, Suwon, Korea.
- 4Department of Otorhinolaryngology-Head and Neck Surgery, The Catholic University of Korea, Seoul, Korea.
- 5Department of Otorhinolaryngology-Head and Neck Surgery, College of Medicine, Kyungpook National University, Daegu, Korea.
- 6Department of Otorhinolaryngology-Head and Neck Surgery, College of Medicine, Yeungnam University, Daegu, Korea.
- 7Department of Otorhinolaryngology-Head and Neck Surgery, Korea University College of Medicine, Seoul, Korea.
- 8Department of Otorhinolaryngology-Head and Neck Surgery, School of Medicine, Kyunghee University, Seoul, Korea.
- 9Department of Otorhinolaryngology, Pusan National University School of Medicine, Busan, Korea.
- 10Department of Otorhinolaryngology, Seoul National University College of Medicine, Seoul, Korea.
- 11Department of Otorhinolaryngology-Head and Neck Surgery, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 12Department of Otolaryngology, Chungnam National University School of Medicine, Daejeon, Korea.
- KMID: 2365544
- DOI: http://doi.org/10.4168/aair.2015.7.2.158
Abstract
- PURPOSE
To evaluate the efficacy and safety of once-daily ciclesonide in comparison to both levocetirizine alone, and a ciclesonide/levocetirizine combination in patients with seasonal allergic rhinitis (SAR) and perennial allergic rhinitis (PAR).
METHODS
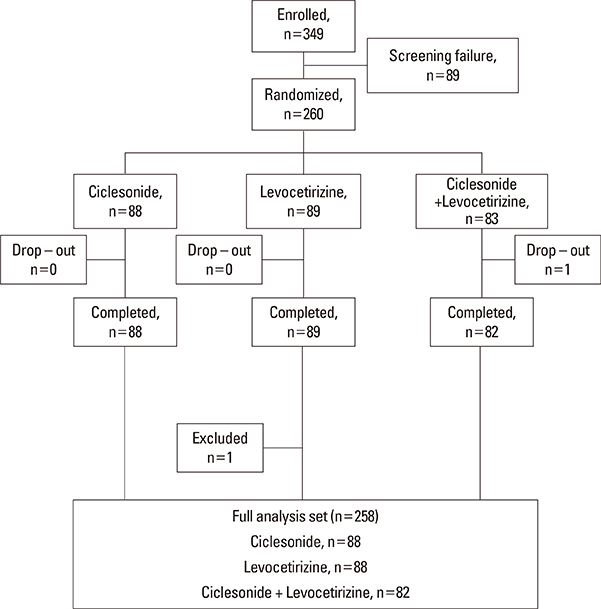
Subjects exhibiting moderate to severe allergic rhinitis for longer than 1 year were randomized in an open-label, 3-arm, parallel group, multicenter study. Subjects received 200 microg ciclesonide, 5 mg levocetirizine, or a combination of both. Changes from baseline until the end-of-study visit (2 weeks following) were evaluated by reflective total nasal symptom scores (rTNSSs), reflective total ocular symptom scores (rTOSSs), physician-assessed overall nasal signs and symptoms severity (PANS), and rhinoconjunctivitis quality-of-life questionnaires (RQLQ).
RESULTS
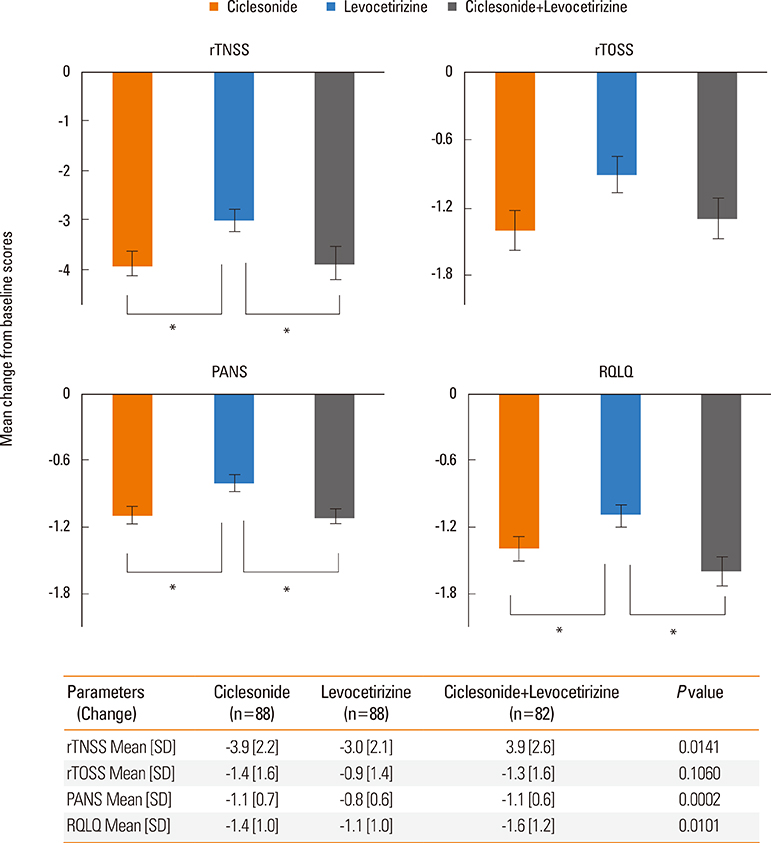
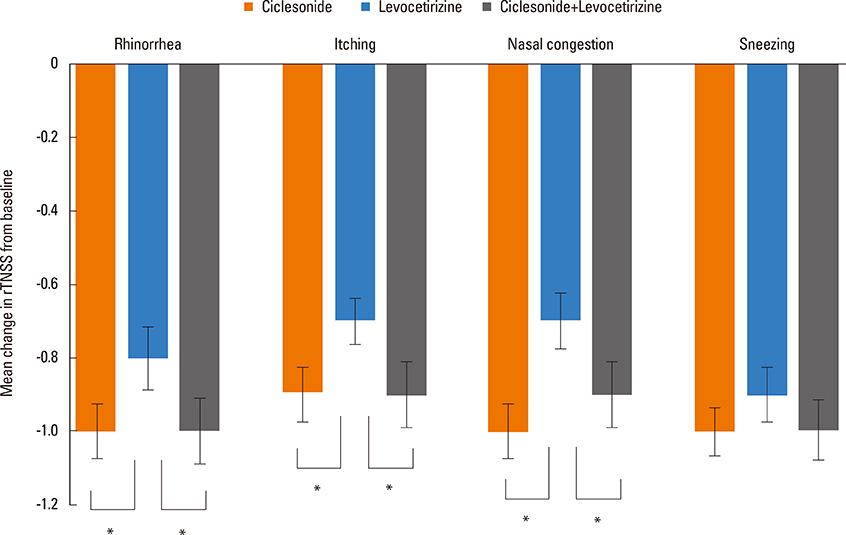
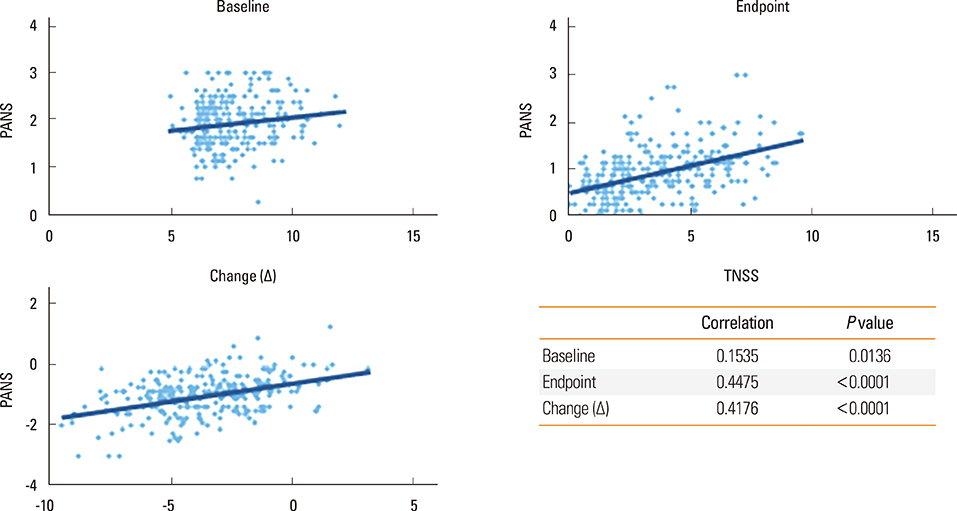
Significant improvements in rTNSS, PANS, and RQLQ in the ciclesonide monotherapy group were observed in comparison to the levocetirizine alone group. Three individual symptoms of rTNSS, including runny nose, nasal itching, and congestion, were improved in the ciclesonide-treated group. rTOSS scores for ciclesonide monotherapy improved from baseline, but no superiority over levocetirizine was shown. The absolute score and changes in rTNSS and PANS were positively correlated. Ciclesonide spray was more effective than levocetirizine in reducing nasal symptoms in both SAR and PAR patients. Ciclesonide and levocetrizine were well tolerated alone and in combination.
CONCLUSIONS
Our results provide support for an AR and its Impact on Asthma (ARIA) recommendation stipulating that ciclesonide is superior to levocetirizine for the treatment of AR, with tolerable safety. Addition of levocetirizine to ciclesonide did not give further clinical benefit over monotherapy.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Anti-Interleukin-9 Antibody Increases the Effect of Allergen-Specific Immunotherapy in Murine Allergic Rhinitis
Ji-Hyeon Shin, Do Hyun Kim, Boo-Young Kim, Sung Won Kim, Se Hwan Hwang, Joohyung Lee, Soo Whan Kim
Allergy Asthma Immunol Res. 2017;9(3):237-246. doi: 10.4168/aair.2017.9.3.237.
Reference
-
1. Nathan RA. The burden of allergic rhinitis. Allergy Asthma Proc. 2007; 28:3–9.2. Brozek JL, Bousquet J, Baena-Cagnani CE, Bonini S, Canonica GW, Casale TB, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines: 2010 revision. J Allergy Clin Immunol. 2010; 126:466–476.3. Postma DS, Sevette C, Martinat Y, Schlösser N, Aumann J, Kafé H. Treatment of asthma by the inhaled corticosteroid ciclesonide given either in the morning or evening. Eur Respir J. 2001; 17:1083–1088.4. Chapman KR, Patel P, D'Urzo AD, Alexander M, Mehra S, Oedekoven C, et al. Maintenance of asthma control by once-daily inhaled ciclesonide in adults with persistent asthma. Allergy. 2005; 60:330–337.5. Mutch E, Nave R, McCracken N, Zech K, Williams FM. The role of esterases in the metabolism of ciclesonide to desisobutyryl-ciclesonide in human tissue. Biochem Pharmacol. 2007; 73:1657–1664.6. Stoeck M, Riedel R, Hochhaus G, Häfner D, Masso JM, Schmidt B, et al. In vitro and in vivo anti-inflammatory activity of the new glucocorticoid ciclesonide. J Pharmacol Exp Ther. 2004; 309:249–258.7. Sunovion Pharmaceuticals Inc (US). Highlights of prescribing information. OMNARIS® (ciclesonide) nasal spray. Initial U.S. approval: 2006. Marlborough (MA): Sunovion Pharmaceuticals Inc.;2006.8. Niphadkar P, Jagannath K, Joshi JM, Awad N, Boss H, Hellbardt S, et al. Comparison of the efficacy of ciclesonide 160 microg QD and budesonide 200 microg BID in adults with persistent asthma: a phase III, randomized, double-dummy, open-label study. Clin Ther. 2005; 27:1752–1763.9. Ratner P, Jacobs R, Mohar D, Huang H, Desai SY, Hinkle J. Evaluation of the efficacy and safety of ciclesonide hydrofluoroalkane nasal aerosol, 80 or 160 μg once daily, for the treatment of seasonal allergic rhinitis. Ann Allergy Asthma Immunol. 2010; 105:471–479.10. Berger WE, Mohar DE, LaForce C, Raphael G, Desai SY, Huang H, et al. A 26-week tolerability study of ciclesonide nasal aerosol in patients with perennial allergic rhinitis. Am J Rhinol Allergy. 2012; 26:302–307.11. Benninger M, Farrar JR, Blaiss M, Chipps B, Ferguson B, Krouse J, et al. Evaluating approved medications to treat allergic rhinitis in the United States: an evidence-based review of efficacy for nasal symptoms by class. Ann Allergy Asthma Immunol. 2010; 104:13–29.12. Barnes ML, Ward JH, Fardon TC, Lipworth BJ. Effects of levocetirizine as add-on therapy to fluticasone in seasonal allergic rhinitis. Clin Exp Allergy. 2006; 36:676–684.13. Chung YJ, Cho IK, Lee KI, Bae SH, Lee JW, Chung PS, et al. Seasonal specificity of seasonal allergens and validation of the ARIA classification in Korea. Allergy Asthma Immunol Res. 2013; 5:75–80.14. Bousquet J, Annesi-Maesano I, Carat F, Léger D, Rugina M, Pribil C, et al. Characteristics of intermittent and persistent allergic rhinitis: DREAMS study group. Clin Exp Allergy. 2005; 35:728–732.15. Fujieda S, Kurono Y, Okubo K, Ichimura K, Enomoto T, Kawauchi H, et al. Examination, diagnosis and classification for Japanese allergic rhinitis: Japanese guideline. Auris Nasus Larynx. 2012; 39:553–556.16. Demoly P, Calderon MA, Casale T, Scadding G, Annesi-Maesano I, Braun JJ, et al. Assessment of disease control in allergic rhinitis. Clin Transl Allergy. 2013; 3:7.17. Hojo M, Ohta K, Iikura M, Mizutani T, Hirashima J, Sugiyama H. Clinical usefulness of a guideline-based screening tool for the diagnosis of allergic rhinitis in asthmatics: the Self Assessment of Allergic Rhinitis and Asthma questionnaire. Respirology. 2013; 18:1016–1021.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Proparacaine in a Mouse Model of Allergic Rhinitis
- Effects of Levocetirizine on Quality of Life in the Patients with Allergic Rhinitis
- Prevalence, comorbidities, diagnosis, and treatment of nonallergic rhinitis: real-world comparison with allergic rhinitis
- Evidences for Local Allergic Rhinitis
- Current trends in treatment of allergic rhinitis