Korean J Ophthalmol.
2015 Dec;29(6):382-388. 10.3341/kjo.2015.29.6.382.
Comparison of Serum Cortisol and Testosterone Levels in Acute and Chronic Central Serous Chorioretinopathy
- Affiliations
-
- 1Department of Ophthalmology, North Eastern Indira Gandhi Regional Institute of Health and Medical Sciences, Shillong, India. natungtanie@gmail.com
- KMID: 2363845
- DOI: http://doi.org/10.3341/kjo.2015.29.6.382
Abstract
- PURPOSE
To compare the levels of serum cortisol and testosterone in acute and chronic central serous chorio-retinopathy (CSC).
METHODS
Serum cortisol and testosterone levels in 30 patients with either acute or chronic CSC were evaluated using chemiluminescent immunoassay.
RESULTS
The mean age was 42.43 +/- 6.37 years (range, 32 to 56 years). The mean 8:00 to 9.00 a.m. serum cortisol level was 12.61 +/- 4.74 microg/dL (range, 6.58 to 27.42 microg/dL). The mean serum testosterone level was 5.88 +/- 1.57 ng/dL (range, 2.81 to 9.94 ng/dL). The mean visual acuity was 20 / 65.07 +/- 40.56 (range, 20 / 25 to 20 / 200). There was no statistically significant difference in the mean levels of serum cortisol and testosterone between the acute and chronic cases (p > 0.05), but there was a statistically significant difference in the mean presenting visual acuity in the two groups (p < 0.05).
CONCLUSIONS
All except one patient in the acute group had normal levels of serum cortisol. Testosterone levels were within the normal range in both the acute and chronic cases of CSC. There is unlikely to be any statistically significant difference in the mean levels of serum cortisol and testosterone between the acute and chronic cases, but there may be a statistically significant difference in the mean presenting visual acuity in these groups.
MeSH Terms
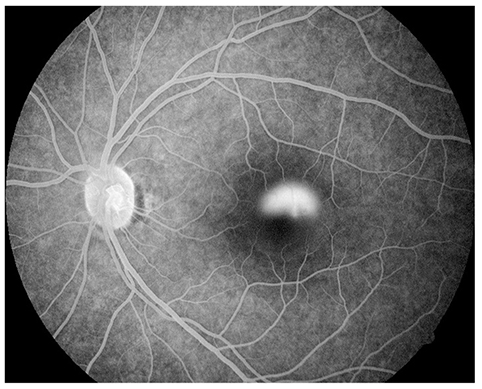
Figure
Reference
-
1. Meyerle CB, Spaide RF. Central serous chorioretinopathy. In : Albert DM, Miller JW, Azar DT, Blodi BA, editors. Albert & Jakobiec's principles and practice of ophthalmology. 3rd ed. Philadelphia: Saunders Elsevier;2008. p. 1871–1880.2. Klais CM, Ober MD, Ciardella AP. . Central serous chorioretinopathy. In : Klais CM, Ober MD, Ciardella AP, editors. Retina. 4th ed. Philadalphia: Elsevier Mosby;2006. p. 1135–1161.3. Garg SP, Dada T, Talwar D, Biswas NR. Endogenous cortisol profile in patients with central serous chorioretinopathy. Br J Ophthalmol. 1997; 81:962–964.4. Pastor-Idoate S, Pena D, Herreras JM. Adrenocortical adenoma and central serous chorioretinopathy: a rare association? Case Rep Ophthalmol. 2011; 2:327–332.5. Zakir SM, Shukla M, Simi ZU, et al. Serum cortisol and testosterone levels in idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 2009; 57:419–422.6. Thoelen AM, Bernasconi PP, Schmid C, Messmer EP. Central serous chorioretinopathy associated with a carcinoma of the adrenal cortex. Retina. 2000; 20:98–99.7. Chalisgaonkar C, Chouhan S, Lakhtakia S, et al. Central serous chorioretinopathy and endogenous cortisol: is there an association? Indian J Ophthalmol. 2010; 58:449–450.8. Tufan HA, Gencer B, Comez AT. Serum cortisol and testosterone levels in chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2013; 251:677–680.9. Rocha EM, Wickham LA, da Silveira LA, et al. Identification of androgen receptor protein and 5alpha-reductase mRNA in human ocular tissues. Br J Ophthalmol. 2000; 84:76–84.10. Ahad MA, Chua CN, Evans NM. Central serous chorioretinopathy associated with testosterone therapy. Eye (Lond). 2006; 20:503–505.11. Grieshaber MC, Staub JJ, Flammer J. The potential role of testosterone in central serous chorioretinopathy. Br J Ophthalmol. 2007; 91:118–119.12. Yannuzzi LA. Type A behavior and central serous chorioretinopathy. Trans Am Ophthalmol Soc. 1986; 84:799–845.13. Bouzas EA, Karadimas P, Pournaras CJ. Central serous chorioretinopathy and glucocorticoids. Surv Ophthalmol. 2002; 47:431–448.14. Carvalho-Recchia CA, Yannuzzi LA, Negrao S, et al. Corticosteroids and central serous chorioretinopathy. Ophthalmology. 2002; 109:1834–1837.15. Iida T, Spaide RF, Negrao SG, et al. Central serous chorioretinopathy after epidural corticosteroid injection. Am J Ophthalmol. 2001; 132:423–425.16. Spaide RF, Campeas L, Haas A, et al. Central serous chorioretinopathy in younger and older adults. Ophthalmology. 1996; 103:2070–2079.17. Spahn C, Wiek J, Burger T, Hansen L. Psychosomatic aspects in patients with central serous chorioretinopathy. Br J Ophthalmol. 2003; 87:704–708.18. Cunningham ET Jr, Alfred PR, Irvine AR. Central serous chorioretinopathy in patients with systemic lupus erythematosus. Ophthalmology. 1996; 103:2081–2090.19. Carpenter MT, O'Boyle JE, Enzenauer RW, et al. Choroiditis in systemic lupus erythematosus. Am J Ophthalmol. 1994; 117:535–536.20. Chumbley LC, Frank RN. Central serous retinopathy and pregnancy. Am J Ophthalmol. 1974; 77:158–160.21. Said-Ahmed K, Moustafa G, Fawzy M. Incidence and natural course of symptomatic central serous chorioretinopathy in pregnant women in a maternity hospital in Kuwait. Middle East Afr J Ophthalmol. 2012; 19:273–276.22. Haimovici R, Koh S, Gagnon DR, et al. Risk factors for central serous chorioretinopathy: a case-control study. Ophthalmology. 2004; 111:244–249.23. Tittl MK, Spaide RF, Wong D, et al. Systemic findings associated with central serous chorioretinopathy. Am J Ophthalmol. 1999; 128:63–68.24. Lee CS, Kang EC, Lee KS, et al. Central serous chorioretinopathy after renal transplantation. Retina. 2011; 31:1896–1903.25. Basu S, Das T, Padhi TR. Serous retinal detachment and multiple retinal pigment epithelial detachments, following hemodialysis for multi-organ failure. Indian J Ophthalmol. 2010; 58:261–262.26. Hussain D, Gass JD. Idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 1998; 46:131–137.27. Liu DT, Fok AT, Lam DS. An update on the diagnosis and management of central serous chorioretinopathy. Asia Pac J Ophthalmol (Phila). 2012; 1:296–302.28. Gilbert CM, Owens SL, Smith PD, Fine SL. Long-term follow-up of central serous chorioretinopathy. Br J Ophthalmol. 1984; 68:815–820.29. Kapetanios AD, Donati G, Bouzas E, et al. Serous central chorioretinopathy and endogenous hypercortisolemia. Klin Monbl Augenheilkd. 1998; 212:343–344.30. Haimovici R, Rumelt S, Melby J. Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology. 2003; 110:698–703.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Chronic Central Serous Chorioretinopathy Associated with Pituitary Adenoma
- A case of Atypical Central Serous Chorioretinopathy with Bullous Retinal Detachment
- Stellate Ganglion Block for Treatment of Central Serous Chorioretinopathy
- A Case of Central Serous Chorioretinopathy Following Systemic Corticosteroid Therapy
- Electronmicroscopic Study of the Effect of Hexamethonium on Serous Choriretinopathy in Rabbits