Ann Lab Med.
2015 Nov;35(6):639-642. 10.3343/alm.2015.35.6.639.
Novel Pathogenic Variant (c.3178G>A) in the SMC1A Gene in a Family With Cornelia de Lange Syndrome Identified by Exome Sequencing
- Affiliations
-
- 1Department of Laboratory Medicine, Korea University College of Medicine, Seoul, Korea.
- 2Department of Molecular Cell Biology, Samsung Biomedical Research Institute, Sungkyunkwan University School of Medicine, Suwon, Korea.
- 3Department of Pediatrics, Catholic University of Daegu School of Medicine, Daegu, Korea. kimjk@cu.ac.kr
- 4Department of Laboratory Medicine and Genetics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. changski@skku.edu
- KMID: 2363269
- DOI: http://doi.org/10.3343/alm.2015.35.6.639
Abstract
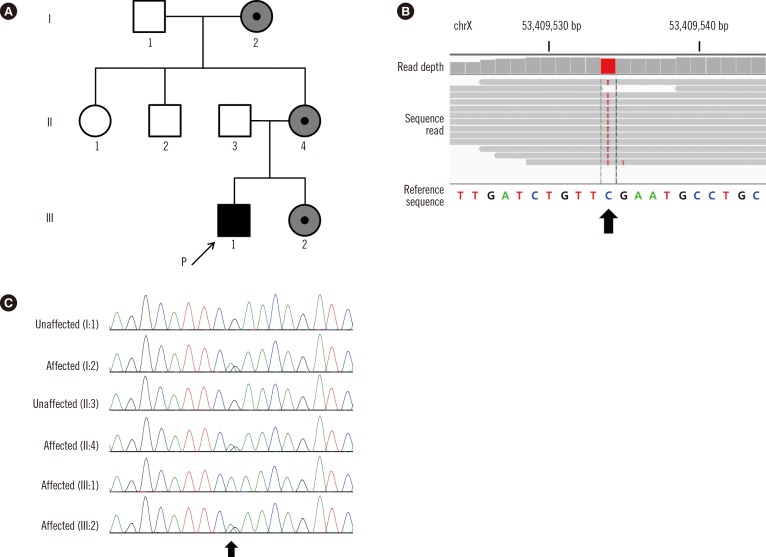
- Cornelia de Lange syndrome (CdLS) is a clinically and genetically heterogeneous congenital anomaly. Mutations in the NIPBL gene account for a half of the affected individuals. We describe a family with CdLS carrying a novel pathogenic variant of the SMC1A gene identified by exome sequencing. The proband was a 3-yr-old boy presenting with a developmental delay. He had distinctive facial features without major structural anomalies and tested negative for the NIPBL gene. His younger sister, mother, and maternal grandmother presented with mild mental retardation. By exome sequencing of the proband, a novel SMC1A variant, c.3178G>A, was identified, which was expected to cause an amino acid substitution (p.Glu1060Lys) in the highly conserved coiled-coil domain of the SMC1A protein. Sanger sequencing confirmed that the three female relatives with mental retardation also carry this variant. Our results reveal that SMC1A gene defects are associated with milder phenotypes of CdLS. Furthermore, we showed that exome sequencing could be a useful tool to identify pathogenic variants in patients with CdLS.
Keyword
MeSH Terms
-
Asian Continental Ancestry Group/genetics
Base Sequence
Cell Cycle Proteins/*genetics
Child, Preschool
Chromosomal Proteins, Non-Histone/*genetics
DNA
DNA Mutational Analysis
De Lange Syndrome/diagnosis/*genetics
Female
Heterozygote
Humans
Male
Pedigree
Phenotype
Polymorphism, Single Nucleotide
Proteins/genetics
Republic of Korea
Cell Cycle Proteins
Chromosomal Proteins, Non-Histone
DNA
Proteins
Figure
Reference
-
1. Liu J, Krantz ID. Cornelia de Lange syndrome, cohesin, and beyond. Clin Genet. 2009; 76:303–314. PMID: 19793304.
Article2. Kline AD, Grados M, Sponseller P, Levy HP, Blagowidow N, Schoedel C, et al. Natural history of aging in Cornelia de Lange syndrome. Am J Med Genet C Semin Med Genet. 2007; 145C:248–260. PMID: 17640042.
Article3. Deardorff MA, Kaur M, Yaeger D, Rampuria A, Korolev S, Pie J, et al. Mutations in cohesin complex members SMC3 and SMC1A cause a mild variant of cornelia de Lange syndrome with predominant mental retardation. Am J Hum Genet. 2007; 80:485–494. PMID: 17273969.
Article4. Kline AD, Krantz ID, Sommer A, Kliewer M, Jackson LG, FitzPatrick DR, et al. Cornelia de Lange syndrome: clinical review, diagnostic and scoring systems, and anticipatory guidance. Am J Med Genet A. 2007; 143A:1287–1296. PMID: 17508425.
Article5. Ansari M, Poke G, Ferry Q, Williamson K, Aldridge R, Meynert AM, et al. Genetic heterogeneity in Cornelia de Lange syndrome (CdLS) and CdLS-like phenotypes with observed and predicted levels of mosaicism. J Med Genet. 2014; 51:659–668. PMID: 25125236.6. Mannini L, Cucco F, Quarantotti V, Krantz ID, Musio A. Mutation spectrum and genotype-phenotype correlation in Cornelia de Lange syndrome. Hum Mutat. 2013; 34:1589–1596. PMID: 24038889.
Article7. Gillis LA, McCallum J, Kaur M, DeScipio C, Yaeger D, Mariani A, et al. NIPBL mutational analysis in 120 individuals with Cornelia de Lange syndrome and evaluation of genotype-phenotype correlations. Am J Hum Genet. 2004; 75:610–623. PMID: 15318302.
Article8. Park KH, Lee ST, Ki CS, Byun SY. Cornelia de Lange Syndrome with NIPBL gene mutation: a case report. J Korean Med Sci. 2010; 25:1821–1823. PMID: 21165303.
Article9. Park HD, Ki CS, Kim JW, Kim WT, Kim JK. Clinical and genetic analysis of Korean patients with Cornelia de Lange syndrome: two novel NIPBL mutations. Ann Clin Lab Sci. 2010; 40:20–25. PMID: 20124326.10. Musio A, Selicorni A, Focarelli ML, Gervasini C, Milani D, Russo S, et al. X-linked Cornelia de Lange syndrome owing to SMC1L1 mutations. Nat Genet. 2006; 38:528–530. PMID: 16604071.
Article11. Hirano T. At the heart of the chromosome: SMC proteins in action. Nat Rev Mol Cell Biol. 2006; 7:311–322. PMID: 16633335.
Article12. Liu J, Feldman R, Zhang Z, Deardorff MA, Haverfield EV, Kaur M, et al. SMC1A expression and mechanism of pathogenicity in probands with X-Linked Cornelia de Lange syndrome. Hum Mutat. 2009; 30:1535–1542. PMID: 19701948.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A family with X-linked Cornelia de Lange syndrome due to a novel SMC1A missense mutation identified by multi-gene panel sequencing
- Legg-Perthes Disease Associated with Cornelia de Lange Syndrome: A Case Report
- Two cases of Cornelia de Lange syndrome
- Dental Findings in Cornelia De Lange Syndrome
- Clinical and molecular characteristics of Korean children with Cornelia de Lange syndrome