J Periodontal Implant Sci.
2016 Oct;46(5):303-319. 10.5051/jpis.2016.46.5.303.
The influence of root surface distance to alveolar bone and periodontal ligament on periodontal wound healing
- Affiliations
-
- 1Division of Periodontology and Implantology, Department of Biomedical and Neuromotor Sciences, Alma Mater Studiorum, University of Bologna School of Dentistry, Bologna, Italy. m.montevecchi@unibo.it
- 2BITTA Laboratory, Rizzoli Orthopaedic Institute, Bologna, Italy.
- 3Preclinical and Surgical Studies Laboratory, Rizzoli Orthopaedic Institute, Bologna, Italy.
- 4Faculty of Veterinary Medicine, Teramo University, Teramo, Italy.
- KMID: 2354929
- DOI: http://doi.org/10.5051/jpis.2016.46.5.303
Abstract
- PURPOSE
The purpose of this animal study was to perform a 3-dimensional micro-computed tomography (micro-CT) analysis in order to investigate the influence of root surface distance to the alveolar bone and the periodontal ligament on periodontal wound healing after a guided tissue regeneration (GTR) procedure.
METHODS
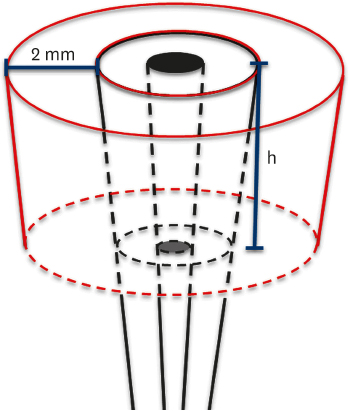
Three adult Sus scrofa domesticus specimens were used. The study sample included 6 teeth, corresponding to 2 third mandibular incisors from each animal. After coronectomy, a circumferential bone defect was created in each tooth by means of calibrated piezoelectric inserts. The experimental defects had depths of 3 mm, 5 mm, 7 mm, 9 mm, and 11 mm, with a constant width of 2 mm. One tooth with no defect was used as a control. The defects were covered with a bioresorbable membrane and protected with a flap. After 6 months, the animals were euthanised and tissue blocks were harvested and preserved for micro-CT analysis.
RESULTS
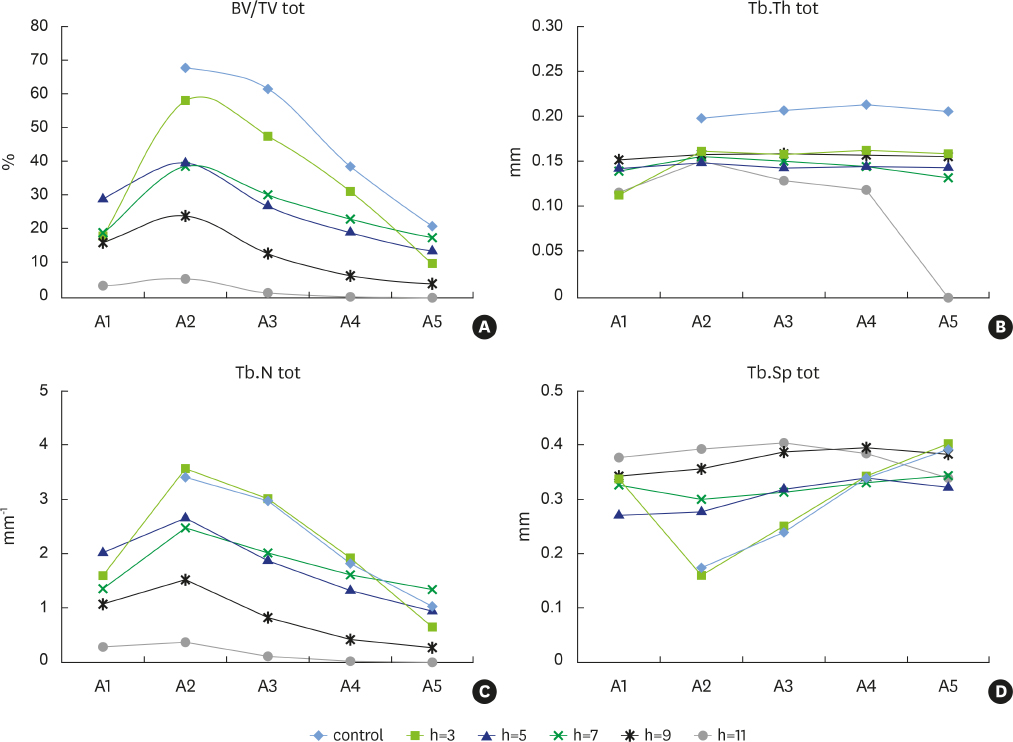
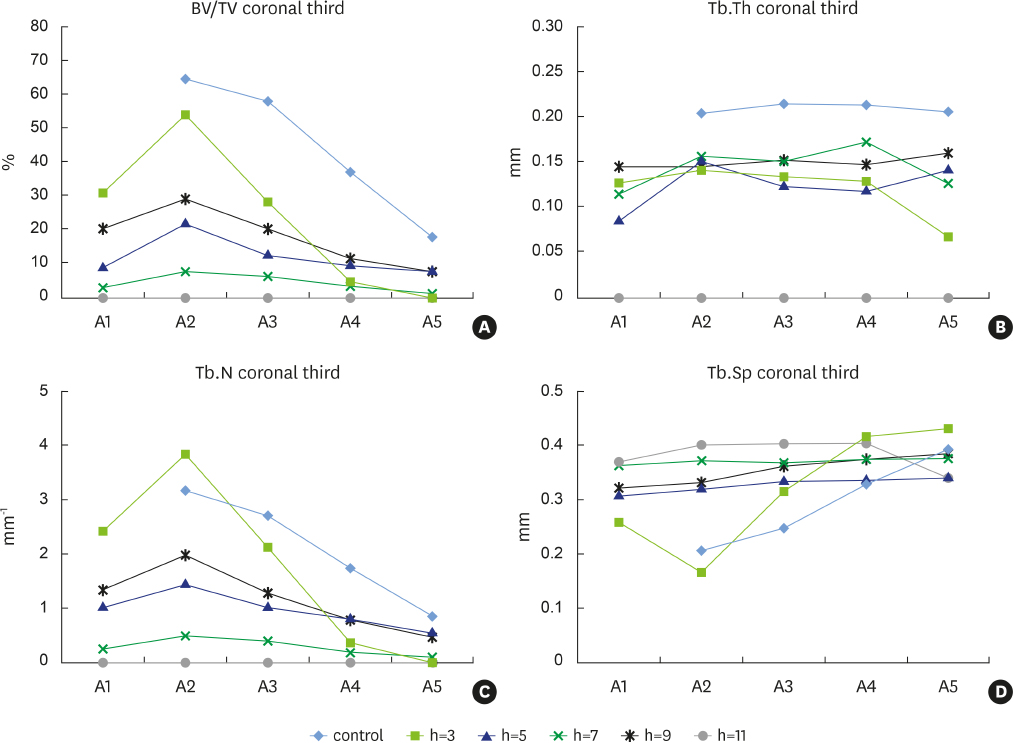
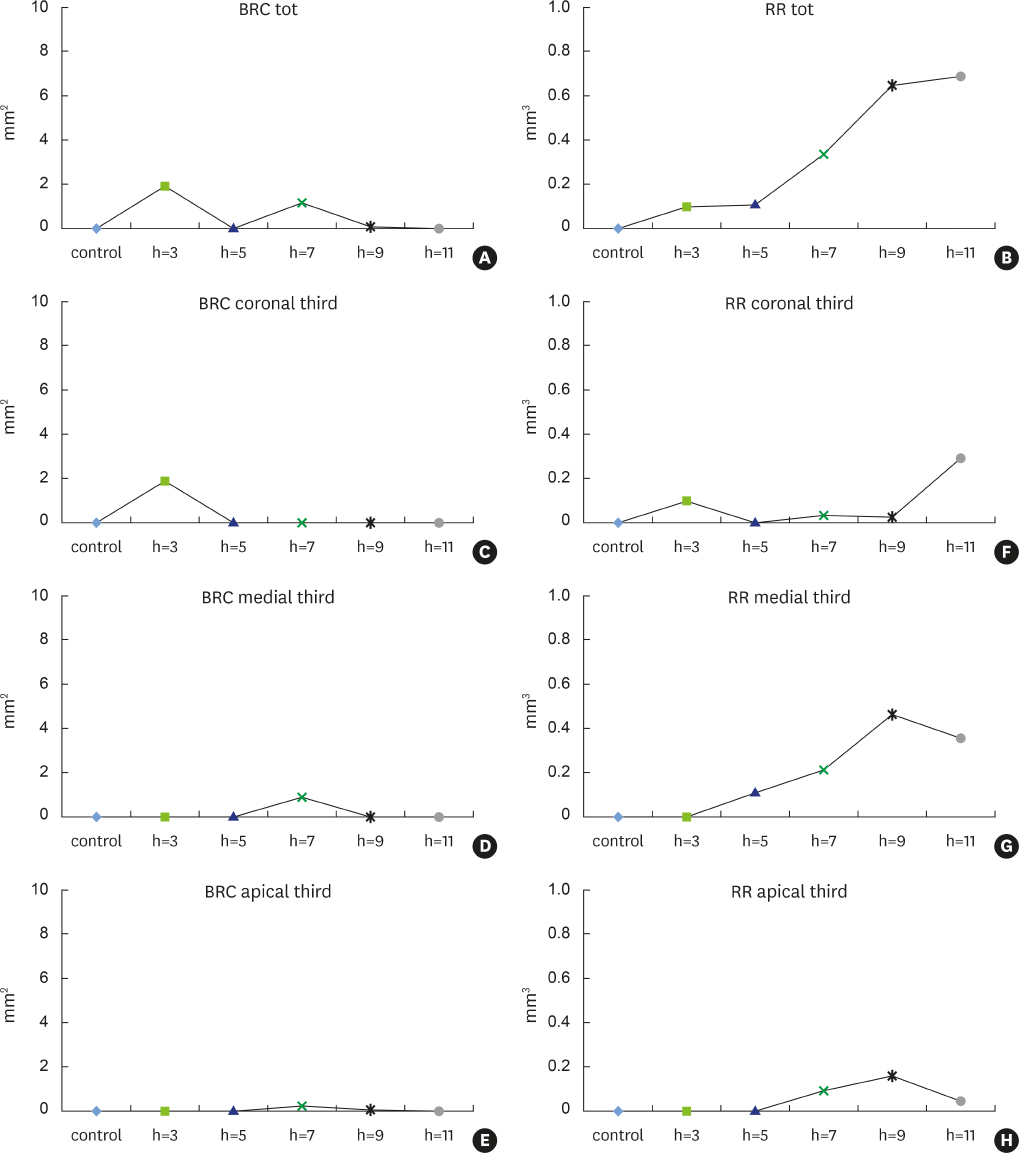
New alveolar bone was consistently present in all experimental defects. Signs of root resorption were observed in all samples, with the extent of resorption directly correlated to the vertical extent of the defect; the medial third of the root was the most commonly affected area. Signs of ankylosis were recorded in the defects that were 3 mm and 7 mm in depth. Density and other indicators of bone quality decreased with increasing defect depth.
CONCLUSIONS
After a GTR procedure, the periodontal ligament and the alveolar bone appeared to compete in periodontal wound healing. Moreover, the observed decrease in bone quality indicators suggests that intrabony defects beyond a critical size cannot be regenerated. This finding may be relevant for the clinical application of periodontal regeneration, since it implies that GTR has a dimensional limit.
Keyword
MeSH Terms
Figure
Reference
-
1. Cortellini P, Bowers GM. Periodontal regeneration of intrabony defects: an evidence-based treatment approach. Int J Periodontics Restorative Dent. 1995; 15:128–145.2. American Academy of Periodontology. Glossary of periodontal terms. 4th ed. Chicago (IL): American Academy of Periodontology;2001.3. Wikesjö UM, Selvig KA. Periodontal wound healing and regeneration. Periodontol 2000. 1999; 19:21–39.
Article4. Sculean A, Nikolidakis D, Schwarz F. Regeneration of periodontal tissues: combinations of barrier membranes and grafting materials-biological foundation and preclinical evidence: a systematic review. J Clin Periodontol. 2008; 35:106–116.
Article5. Trombelli L. Which reconstructive procedures are effective for treating the periodontal intraosseous defect? Periodontol 2000. 2005; 37:88–105.
Article6. Karring T, Nyman S, Lindhe J, Sirirat M. Potentials for root resorption during periodontal wound healing. J Clin Periodontol. 1984; 11:41–52.
Article7. Melcher AH. On the repair potential of periodontal tissues. J Periodontol. 1976; 47:256–260.
Article8. Magnusson I, Claffey N, Bogle G, Garrett S, Egelberg J. Root resorption following periodontal flap procedures in monkeys. J Periodontal Res. 1985; 20:79–85.
Article9. Klinge B, Nilvéus R, Egelberg J. Bone regeneration pattern and ankylosis in experimental furcation defects in dogs. J Clin Periodontol. 1985; 12:456–464.
Article10. Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004; 364:149–155.
Article11. Nagatomo K, Komaki M, Sekiya I, Sakaguchi Y, Noguchi K, Oda S, et al. Stem cell properties of human periodontal ligament cells. J Periodontal Res. 2006; 41:303–310.
Article12. Listgarten MA, Rosenberg MM. Histological study of repair following new attachment procedures in human periodontal lesions. J Periodontol. 1979; 50:333–344.
Article13. Linghorne WJ, O’Connell DC. Studies in the regeneration and reattachment of supporting structures of the teeth; soft tissue reattachment. J Dent Res. 1950; 29:419–428.
Article14. Karring T, Nyman S, Gottlow J, Laurell L. Development of the biological concept of guided tissue regeneration-animal and human studies. Periodontol 2000. 1993; 1:26–35.
Article15. Karring T, Cortellini P. Regenerative therapy: furcation defects. Periodontol 2000. 1999; 19:115–137.
Article16. Polimeni G, Susin C, Wikesjö UM. Regenerative potential and healing dynamics of the periodontium: a critical-size supra-alveolar periodontal defect study. J Clin Periodontol. 2009; 36:258–264.
Article17. Graziani F, Laurell L, Tonetti M, Gottlow J, Berglundh T. Periodontal wound healing following GTR therapy of dehiscence-type defects in the monkey: short-, medium- and long-term healing. J Clin Periodontol. 2005; 32:905–914.
Article18. Laurell L, Bose M, Graziani F, Tonetti M, Berglundh T. The structure of periodontal tissues formed following guided tissue regeneration therapy of intra-bony defects in the monkey. J Clin Periodontol. 2006; 33:596–603.
Article19. Samuel JL, Woodall PF. Periodontal disease in feral pigs (Sus scrofa) from Queensland, Australia. J Wildl Dis. 1988; 24:201–206.20. Štembírek J, Kyllar M, Putnová I, Stehlík L, Buchtová M. The pig as an experimental model for clinical craniofacial research. Lab Anim. 2012; 46:269–279.
Article21. Hildebrand T, Rüegsegger P. Quantification of bone microarchitecture with the structure model index. Comput Methods Biomech Biomed Engin. 1997; 1:15–23.
Article22. Russell WM, Burch RL. The principles of humane experimental technique [Internet]. Baltimore (MD): Johns Hopkins University;cited 2015 Jun 24. Available from: http://altweb.jhsph.edu/pubs/books/humane_exp/het-toc.23. Pandis N. Sample calculation for split-mouth designs. Am J Orthod Dentofacial Orthop. 2012; 141:818–819.
Article24. Becker W, Becker BE. Periodontal regeneration: a contemporary re-evaluation. Periodontol 2000. 1999; 19:104–114.
Article25. Karring T, Nyman S, Lindhe J. Healing following implantation of periodontitis affected roots into bone tissue. J Clin Periodontol. 1980; 7:96–105.
Article26. Nyman S, Karring T, Lindhe J, Plantén S. Healing following implantation of periodontitis-affected roots into gingival connective tissue. J Clin Periodontol. 1980; 7:394–401.
Article27. Gottlow J, Nyman S, Karring T. Healing following citric acid conditioning of roots implanted into bone and gingival connective tissue. J Periodontal Res. 1984; 19:214–220.
Article28. Crigger M, Bogle GC, Garrett S, Gantes BG. Repair following treatment of circumferential periodontal defects in dogs with collagen and expanded polytetrafluoroethylene barrier membranes. J Periodontol. 1996; 67:403–413.
Article29. Klinge B, Nilvéus R, Kiger RD, Egelberg J. Effect of flap placement and defect size on healing of experimental furcation defects. J Periodontal Res. 1981; 16:236–248.
Article30. Karring T, Isidor F, Nyman S, Lindhe J. New attachment formation on teeth with a reduced but healthy periodontal ligament. J Clin Periodontol. 1985; 12:51–60.
Article31. Andreasen JO. The effect of splinting upon periodontal healing after replantation of permanent incisors in monkeys. Acta Odontol Scand. 1975; 33:313–323.
Article32. Nasjleti CE, Castelli WA, Caffesse RG. The effects of different splinting times on replantation of teeth in monkeys. Oral Surg Oral Med Oral Pathol. 1982; 53:557–566.
Article33. Kristerson L, Andreasen JO. The effect of splinting upon periodontal and pulpal healing after autotransplantation of mature and immature permanent incisors in monkeys. Int J Oral Surg. 1983; 12:239–249.
Article34. Bauss O, Schilke R, Fenske C, Engelke W, Kiliaridis S. Autotransplantation of immature third molars: influence of different splinting methods and fixation periods. Dent Traumatol. 2002; 18:322–328.
Article35. Gandolfi MG, Parrilli AP, Fini M, Prati C, Dummer PM. 3D micro-CT analysis of the interface voids associated with Thermafil root fillings used with AH Plus or a flowable MTA sealer. Int Endod J. 2013; 46:253–263.
Article36. Park CH, Abramson ZR, Taba M Jr, Jin Q, Chang J, Kreider JM, et al. Three-dimensional micro-computed tomographic imaging of alveolar bone in experimental bone loss or repair. J Periodontol. 2007; 78:273–281.
Article37. de Paula Reis MV, Moura CC, Soares PB, Leoni GB, Souza-Neto MD, Barbosa DZ, et al. Histologic and micro-computed tomographic analyses of replanted teeth stored in different kind of media. J Endod. 2014; 40:665–669.
Article38. Martini L, Staffa G, Giavaresi G, Salamanna F, Parrilli A, Serchi E, et al. Long-term results following cranial hydroxyapatite prosthesis implantation in a large skull defect model. Plast Reconstr Surg. 2012; 129:625e–635e.
Article39. Vercellotti T, Nevins ML, Kim DM, Nevins M, Wada K, Schenk RK, et al. Osseous response following resective therapy with piezosurgery. Int J Periodontics Restorative Dent. 2005; 25:543–549.40. Do MJ, Kim K, Lee H, Cha S, Seo T, Park HJ, et al. Development of animal experimental periodontitis models. J Periodontal Implant Sci. 2013; 43:147–152.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The effects of Acellular dermal matrix on the healing of 1 wall intrabony defects in dogs
- EFFECTS OF BONE MORPHOGENETIC PROTEIN ON THE HEALING OF PERIODONTIUM AFTER TOOTH REPLANTATION OF THE RAT
- ULTRASTRUCTURAL INVESTIGATIONS OF THE INTERFACE BETWEEN CULTURED PERIODONTAL LIGAMENT CELLS AND TITANIUM
- An experimental study on the periodontal tissue reaction to tooth movement in the rat
- Micro-computed tomography analysis of changes in the periodontal ligament and alveolar bone proper induced by occlusal hypofunction of rat molars