Korean Circ J.
2016 May;46(3):402-407. 10.4070/kcj.2016.46.3.402.
Levosimendan Reduces Lung Injury in a Canine Model of Cardiopulmonary Bypass
- Affiliations
-
- 1The First Ward of Cardiovascular Surgery Department of the First Affiliated Hospital of Anhui Medical University, Hefei, China. aydgsl@sina.com
- 2The Oncology Department of the Hefei Second People's Hospital, Hefei, China.
- KMID: 2344453
- DOI: http://doi.org/10.4070/kcj.2016.46.3.402
Abstract
- BACKGROUND AND OBJECTIVES
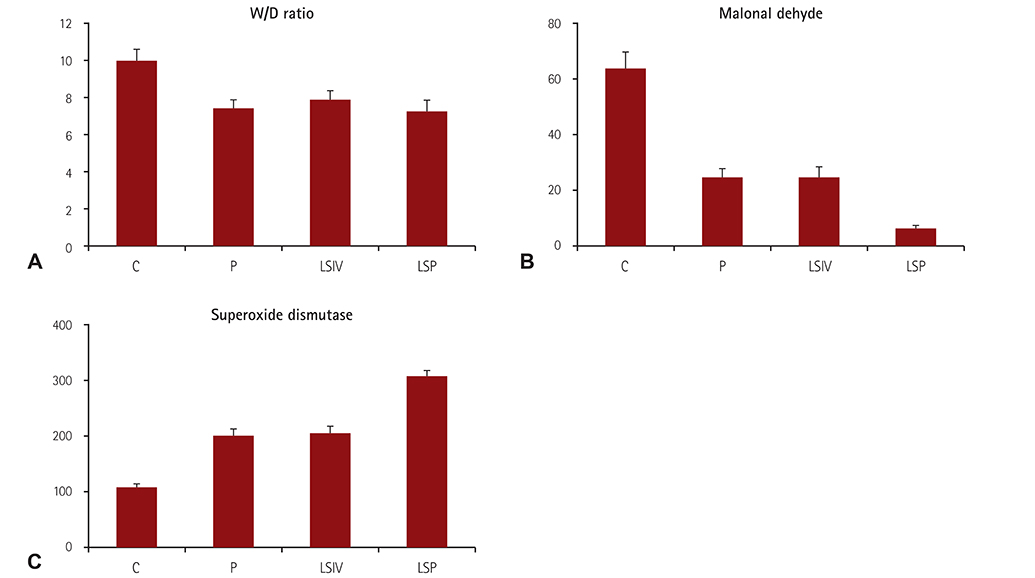
To explore the lung-protective effect of levosimendan (LS) during cardiopulmonary bypass in a canine model by determining the wet/dry weight (W/D) ratio of lung tissue, malonaldehyde (MDA) and superoxide dismutase (SOD) concentrations, and performing a histological evaluation.
MATERIALS AND METHODS
Thirty-two canines were divided randomly into four groups and underwent a routine aortic cross-clamping cardiopulmonary bypass procedure for 1 h, followed by recovery for 2 h. Animals were handled as follows: group C (means control group), no special treatment after aortic cross clamping; group P (means pulmonary artery perfusion group), pulmonary artery perfusion with cold oxygenated blood after aortic cross clamping; group LSIV (means intravenous injection of LS group), intravenous injection of LS (65 µg/kg) before thoracotomy, and the rest of the procedure was identical to the control group; group LPS (means pulmonary perfusion with LS group), pulmonary perfusion with cold oxygenated blood combined with LS (65 µg/kg) after aortic cross clamping. Lung tissues were removed and subjected to evaluation of pathological alterations, W/D ratio and MDA and SOD concentrations.
RESULTS
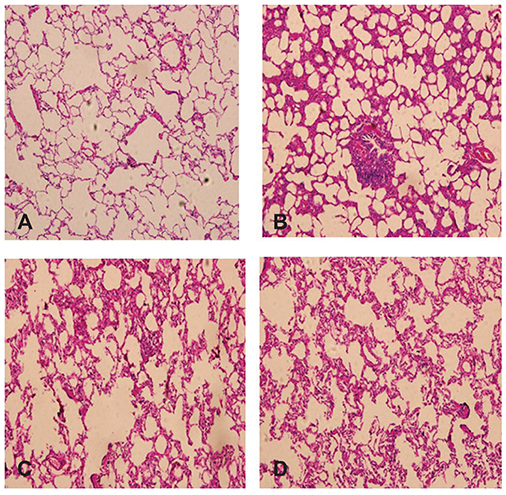
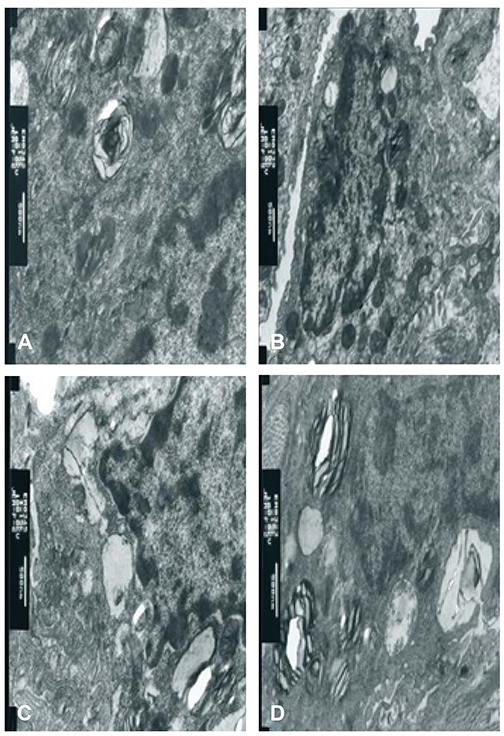
In group C, the W/D ratio and MDA concentration were higher, while the SOD concentrations were lower (p<0.05). Compared with groups P and LSIV, the MDA concentration was lower in group LPS, while that of SOD was higher (p<0.05); Light and electron microscopy indicated that LS intervention reduced impairment of lung tissues.
CONCLUSION
Our findings suggest that LS plays an important role in protecting lung tissues.
MeSH Terms
Figure
Reference
-
1. Qiu W, Zheng L, Gu H, Chen D, Chen Y. Comparison between adult and infant lung injury in a rabbit ischemia-reperfusion mode. J Thorac Cardiovasc Surg. 2008; 136:352–359.2. Kim JC, Shim JK, Lee S, Yoo YC, Yang SY, Kwak YL. Effect of combined remote ischemic preconditioning and postconditioning on pulmonary function in valvular heart surgery. Chest. 2012; 142:467–475.3. Rong J, Ye S, Liang MY, et al. Receptor for advanced glycation end products involved in lung ischemia reperfusion injury in cardiopulmonary bypass attenuated by controlled oxygen reperfusion in a canine model. ASAIO J. 2013; 59:302–308.4. Rong J, Ye S, Wu ZK, et al. Controlled oxygen reperfusion protects the lung against early ischemia-reperfusion injury in cardiopulmonary bypasses by downregulating high mobility group box 1. Exp Lung Res. 2012; 38:183–191.5. Wang C, Li D, Qian Y, Wang J, Jing H. Increased matrix metalloproteinase-9 activity and mRNA expression in lung injury following cardiopulmonary bypass. Lab Invest. 2012; 92:910–916.6. Qi D, Gao MX, Yu Y. Intratracheal antitumor necrosis factor-α antibody attenuates lung tissue damage following cardiopulmonary bypass. Artif Organs. 2013; 37:142–149.7. Maniatis NA, Kardara M, Hecimovich D, et al. Role of caveolin-1 expression in the pathogenesis of pulmonary edema in ventilator-induced lung injury. Pulm Circ. 2012; 2:452–460.8. Radovits T, Beller CJ, Groves JT, et al. Effects of FP15, a peroxynitrite decomposition catalyst on cardiac and pulmonary function after cardiopulmonary bypass. Eur J Cardiothorac Surg. 2012; 41:391–396.9. Gecit I, Kavak S, Yüksel MB, et al. Effect of short-term treatment with levosimendan on oxidative stress in renal tissues of rats. Toxicol Ind Health. 2014; 30:47–51.10. Paraskevaidis IA, Parissis JT, Kremastinos D. Anti-inflammatory and anti-apoptotic effects of levosimendan in decompensated heart failure: a novel mechanism of drug-induced improvement in contractile performance of the failing heart. Curr Med Chem Cardiovasc Hematol Agents. 2005; 3:243–247.11. Markou T, Makridou Z, Galatou E, Lazou A. Multiple signalling pathways underlie the protective effect of levosimendan in cardiac myocytes. Eur J Pharmacol. 2011; 667:298–305.12. Caimmi PP, Molinari C, Uberti F, et al. Intracoronary levosimendan prevents myocardial ischemic damages and activates survival signaling through ATP-sensitive potassium channel and nitric oxide. Eur J Cardiothorac Surg. 2011; 39:e59–e67.13. Hönisch A, Theuring N, Ebner B, Waqner C, Strasser RH, Weinbrenner C. Postconditioning with levosimendan reduces the infarct size involving the PI3K pathway and KATP-channel activation but is independent of PDE-III inhibition. Basic Res Cardiol. 2010; 105:155–167.14. Ozturk T, Gok S, Nese N. Levosimendan attenuates reperfusion injury in an isolated perfused rat heart model. J Cardiothorac Vasc Anesth. 2010; 24:624–628.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- New Inotropic Agent-Levosimendan
- A scientific analysis of changes in hemostatic parameters following cardiopulmonary bypass procedure
- Delayed Ascending Aorta Replacement in Blunt Chest Trauma with Aortic Injury
- Anesthetic experience in patient for single lung transplantation with previous contralateral pneumonectomy: A case report
- Unilateral Bronchospasm during Cardiopulmonary Bypass: A case report