J Korean Ophthalmol Soc.
2011 Sep;52(9):1089-1093.
Effects of Pupil Dilation and Constriction Agents on Trabecular Meshwork Cells
- Affiliations
-
- 1Department of Ophthalmology, Catholic University of Daegu College of Medicine, Daegu, Korea. jwkim@cu.ac.kr
Abstract
- PURPOSE
To investigate the effects of pupil dilation and constriction agents on the survival and production of nitric oxide (NO) in cultured human trabecular meshwork cells (HTMC).
METHODS
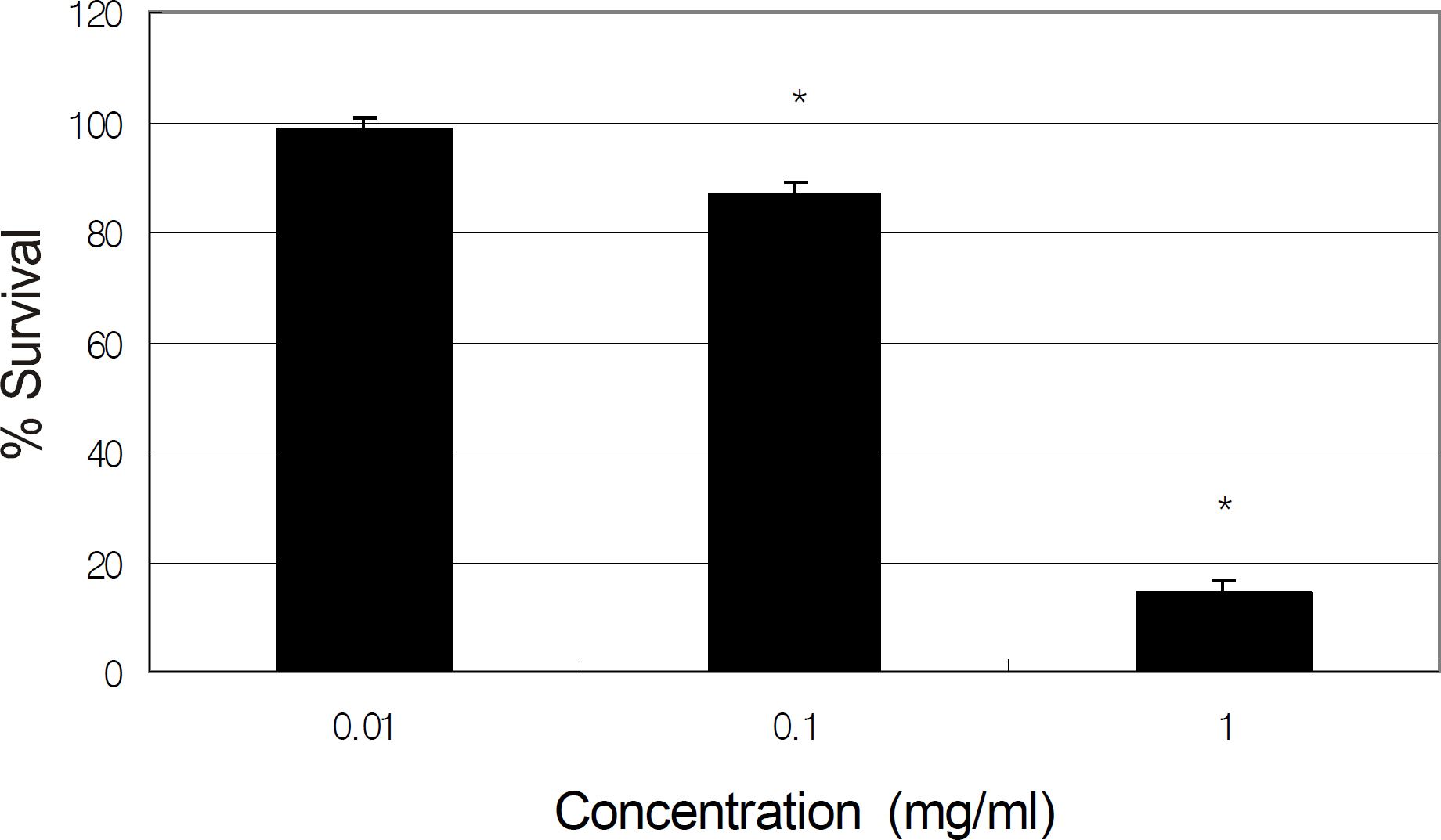
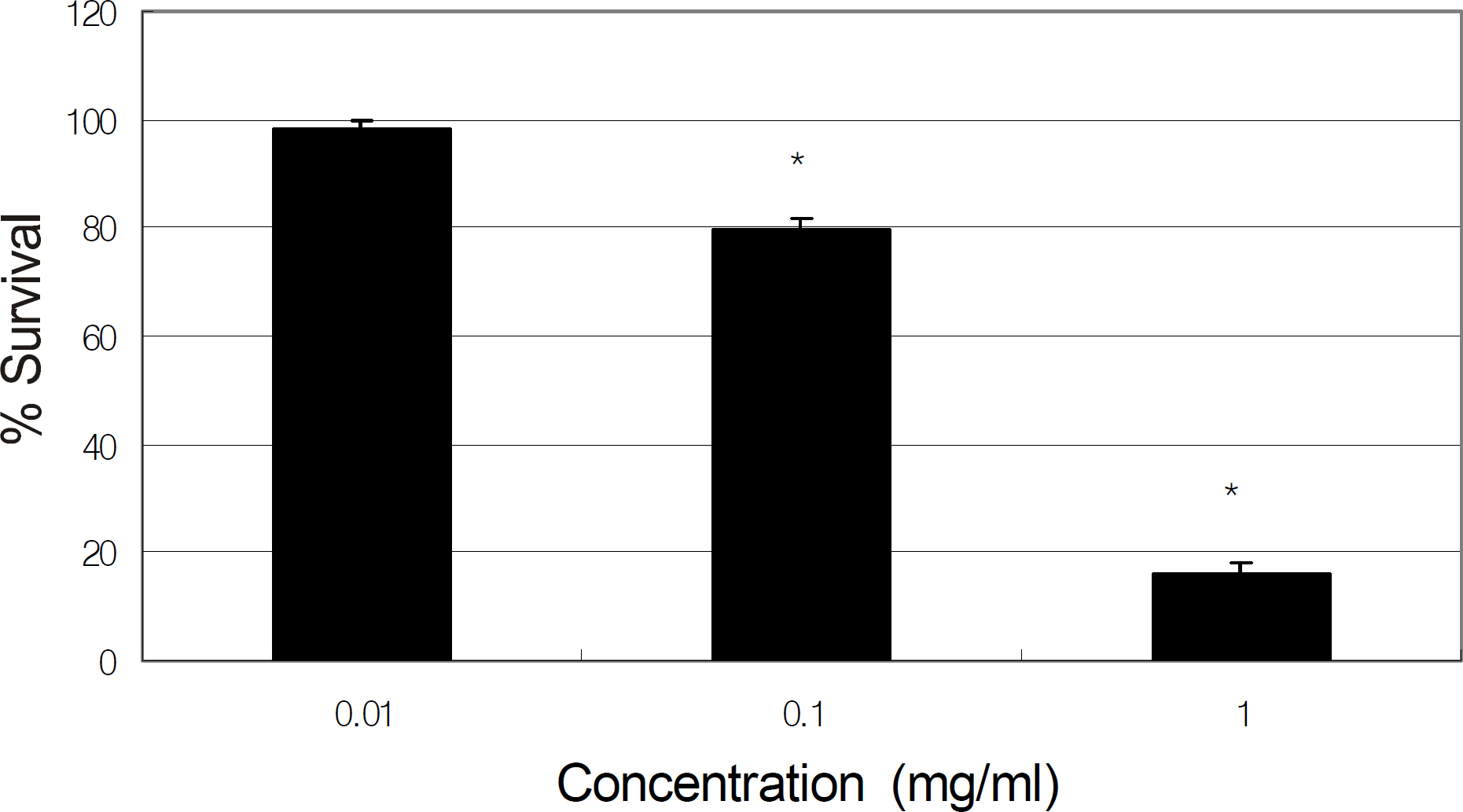
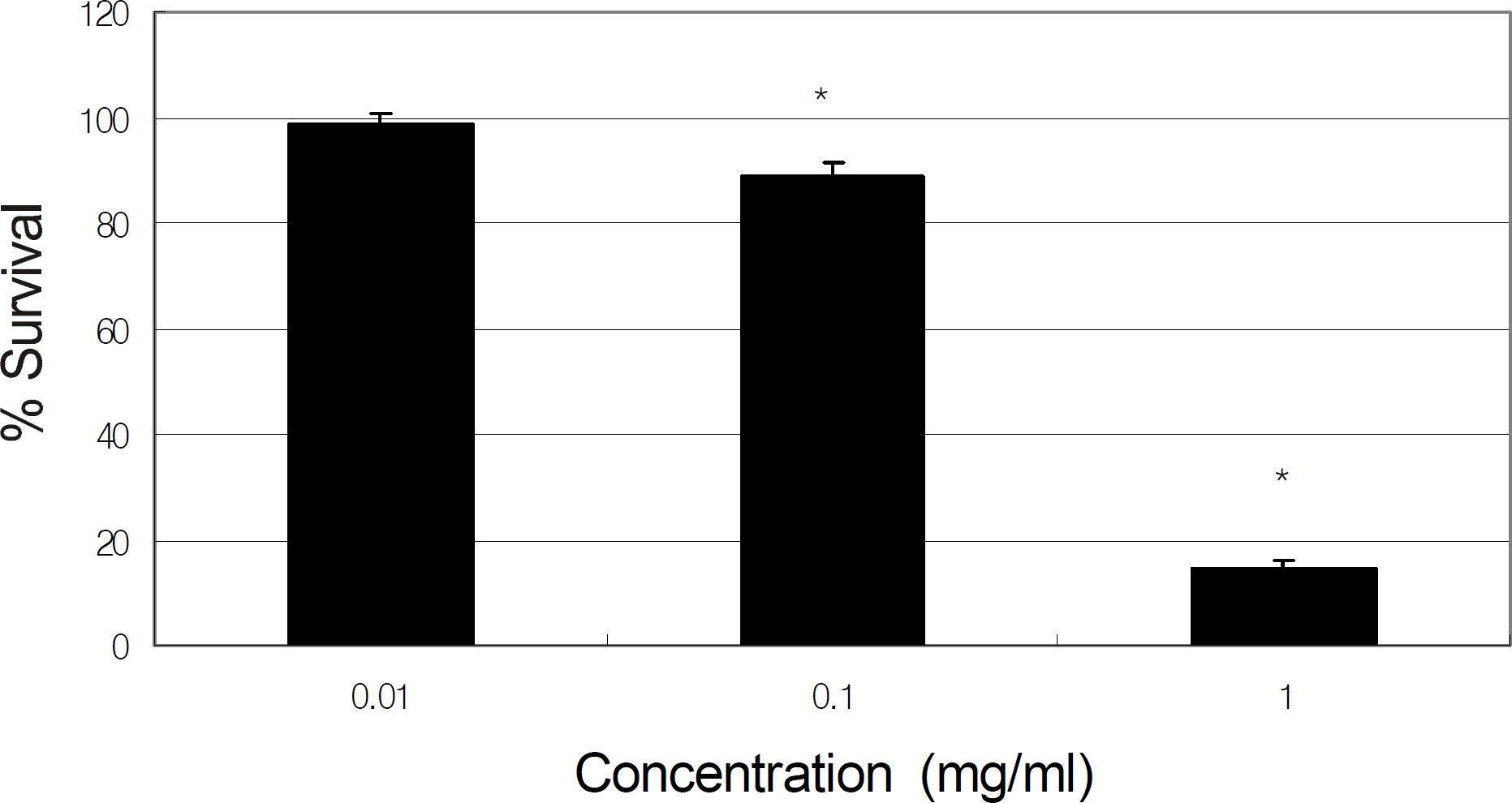
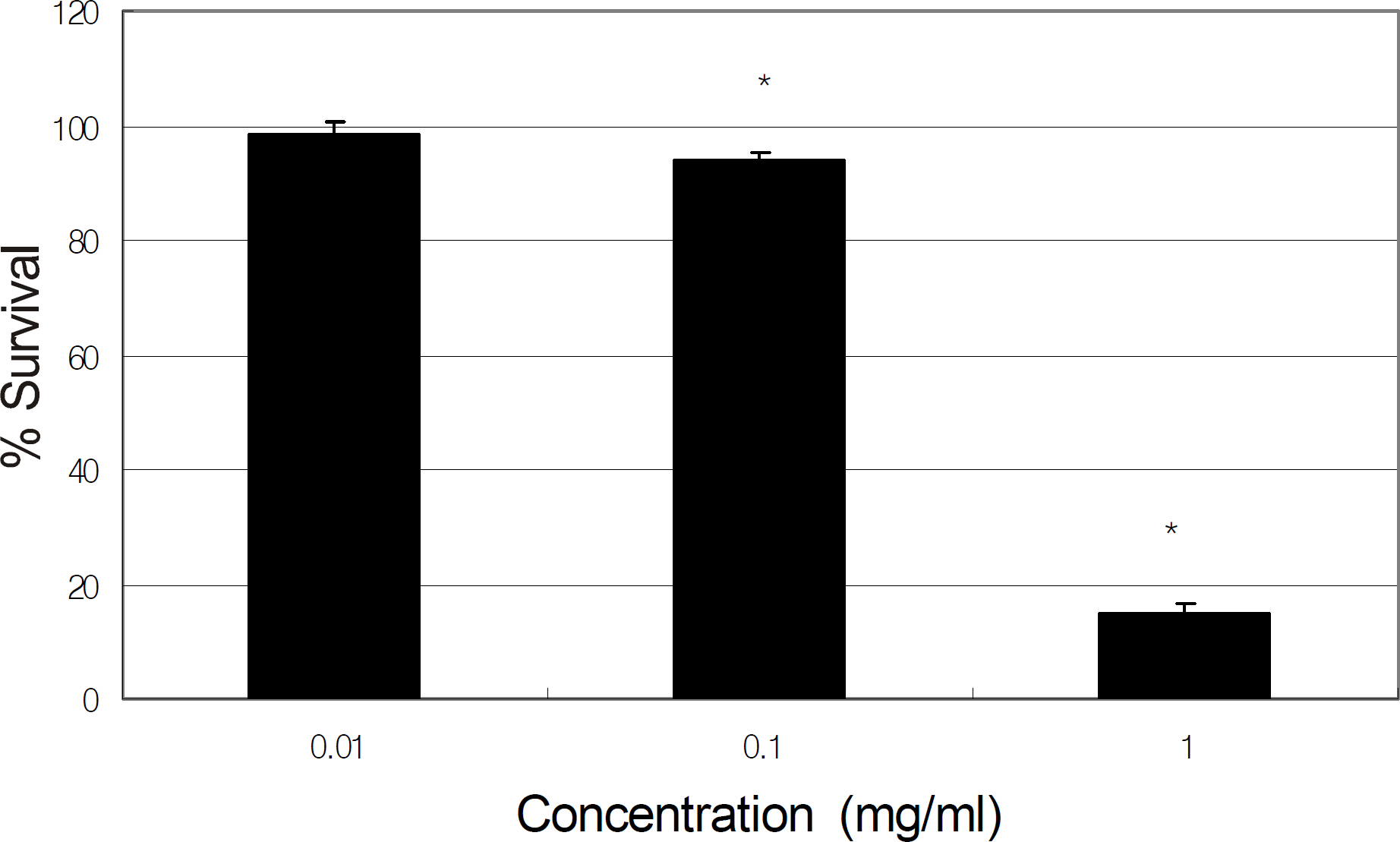
Primarily cultured HTMC were exposed to 0, 0.01, and 0.1 mg/ml of tropicamide, cyclopentolate, atropine, or pilocarpine for 2 hours. Cellular survival and production of NO were assessed using the MTT assay and Griess assay, respectively.
RESULTS
Tropicamide, cyclopentolate, atropine, and pilocarpine decreased cellular survival at the concentration of 0.1 mg. At the concentration of 0.01 mg/ml, all agents decreased production of NO to some extent, although the reduction was not statistically significant.
CONCLUSIONS
Pupil dilation and constriction agents may be toxic to HTMC if used at high concentrations or if used frequently in the short-term but may not affect trabecular outflow.
MeSH Terms
Figure
Reference
-
References
1. Abraham SV. Mydriatic glaucoma: a statistical study. Arch Ophthalmol. 1933; 10:757–62.2. Nelson ME, Orton HP. Counteracting the effects of mydriatics: Does it benefit the patient? Arch Ophthalmol. 1987; 105:486–9.3. Siam GA, de Barros DS, Gheith ME, et al. The amount of intraocular pressure rise during pharmacological pupillary dilatation is an indicator of the likelihood of future progression of glaucoma. Br J Ophthalmol. 2007; 91:1170–2.
Article4. Wiederholt M. Direct involvement of trabecular meshwork in the regulation of aqueous humor outflow. Curr Opin Ophthalmol. 1998; 9:46–9.
Article5. Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991; 43:109–42.6. Bredt DS, Snyder SH. Nitric oxide: a physiologic messenger molecule. Annu Rev Biochem. 1994; 63:175–95.
Article7. Brüne B, Knethen A, Sandau KB. Nitric oxide and its role in apoptosis. Eur J Pharmacol. 1998; 351:261–72.
Article8. Nathanson JA, McKee M. Identification of an extensive system of nitric oxide-producing cells in the ciliary muscle and outflow pathway of the human eye. Invest Ophthalmol Vis Sci. 1995; 36:1765–73.9. Geyer O, Podos SM, Mittag T. Nitric oxide synthase activity in tissues of the bovine eye. Graefes Arch Clin Exp Ophthalmol. 1997; 235:786–93.
Article10. Meyer P, Champion C, Schlotzer-Schrehardt U, et al. Localization of nitric oxide synthase isoforms in porcine ocular tissues. Curr Eye Res. 1999; 18:375–80.
Article11. Schuman JS, Erickson K, Nathanson JA. Nitrovasodilator effects on intraocular pressure and outflow facility in monkeys. Exp Eye Res. 1994; 58:99–105.
Article12. Wang RF, Podos SM. Effect of the topical application of nitro-glycerin on intraocular pressure in normal and glaucomatous monkeys. Exp Eye Res. 1995; 60:337–9.
Article13. Nathanson JA, McKee M. Alterations of ocular nitric oxide synthase in human glaucoma. Invest Ophthalmol Vis Sci. 1995; 36:1774–84.14. Matsuo T. Basic nitric oxide production is enhanced by hydraulic pressure in cultured human trabecular cells. Br J Ophthalmol. 2000; 84:631–5.15. Schneemann A, Dijkstra BG, van den Berg TJ, et al. Nitric ox-ide/guanylate cyclase pathways and flow in anterior segment perfusion. Graefes Arch Clin Exp Ophthalmol. 2002; 240:936–41.
Article16. Galassi F, Renieri G, Sodi A, et al. Nitric oxide proxies and ocular perfusion pressure in primary open angle glaucoma. Br J Ophthalmol. 2004; 88:757–60.
Article17. Ellis DZ, Dismuke WM, Chokshi BM. Characterization of soluble guanylate cyclase in NO-induced increases in aqueous humor outflow facility and in the trabecular meshwork. Invest Ophthalmol Vis Sci. 2009; 50:1808–13.
Article18. Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.
Article19. Green LC, Wagner DA, Glogowski J, et al. Analysis of nitrate, nitrite and [15 N]nitrate in biological fluids. Anal Biochem. 1982; 126:131–8.20. Polansky JR, Weinreb RN, Baxter JD, Alvarado J. Human trabecular cells. I. Establishment in tissue culture and growth characteristics. Invest Ophthalmol Vis Sci. 1979; 18:1043–9.21. Alvarado JA, Wood I, Polansky JR. Human trabecular cells. II. Growth pattern and ultrastructural characteristics. Invest Ophthalmol Vis Sci. 1982; 23:464–78.22. Kawa JE, Higginbotham EJ, Chang IL, Yue BY. Effects of anti-glaucoma medications on bovine trabecular meshwork cells in vitro. Exp Eye Res. 1993; 57:557–65.
Article23. Green K, Downs SJ. Ocular penetration of pilocarpine in rabbits. Arch Ophthalmol. 1975; 93:1165–8.
Article24. Yu AL, Fuchshofer R, Kampik A, Welge-Lű ssen U. Effects of oxidative stress in trabecular meshwork cells are reduced by prostaglandin analogues. Invest Ophthalmol Vis Sci. 2008; 49:4872–80.
Article25. Vasa M, Breitschopf K, Zeiher AM, Dimmeler S. Nitric oxide acti-vates telomerase and delays endothelial cell senescence. Circ Res. 2000; 87:540–2.
Article26. Scalera F, Borlak J, Beckmann B, et al. Endogenous nitric oxide synthesis inhibitor asymmetric dimethyl L-arginine accelerates endothelial cell senescence. Arterioscler Thromb Vasc Biol. 2004; 24:1816–22.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Age-related Changes of the Cellularity and Acid Mucopolysaccharides in the Trabecular Meshwork of the Normal Korean
- The Cytotoxicity of Truncated (Q368X) Myocilin in Trabecular Meshwork (TM) Cells
- Effect of Mitomycin C on the Proliferation and Nitric Oxide Production in the Cultured Trabecular Meshwork Cells
- Effect of beta-adrenergics on the Survival and Production of Nitric Oxide in the Cultured Trabecular Meshwork Cells
- Change of Intraocular Pressure in Trabecular Meshwork Rupture Associated with Traumatic Hyphema