Korean J Urol.
2010 Apr;51(4):281-286.
Changes in Aquaporin 1 Expression in Rat Urinary Bladder after Partial Bladder Outlet Obstruction: Preliminary Report
- Affiliations
-
- 1Department of Urology, Sexual Medical Research Center, Chonnam National University Medical School, Gwangju, Korea. uropark@gmail.com
- 2Department of Anatomy, Sexual Medical Research Center, Chonnam National University Medical School, Gwangju, Korea.
Abstract
- PURPOSE
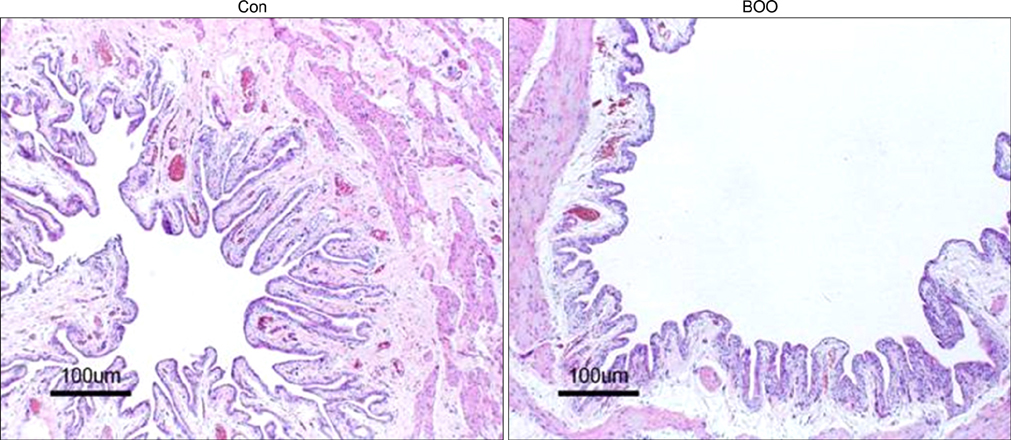
Aquaporins (AQPs) are membrane proteins that facilitate water movement across biological membranes. AQPs are also called water channels, and they have recently been reported to be expressed in rat and human urothelium. The purposes of this study were to investigate the effect of bladder outlet obstruction (BOO) on the rat urothelium and AQP1 expression in rat urothelium. MATERIALS AND METHODS: Female Sprague-Dawley rats (230-240 g each, n=20) were divided into 2 groups: the sham group (the Con group, n=10) and the partial BOO group (the BOO group, n=10). The BOO group underwent a partial BOO. The expression and cellular localization of AQP1 were determined by performing Western blotting and immunohistochemistry on the rat urinary bladder. RESULTS: AQP1 immunoreactivity in both the control and the BOO groups was localized in the capillaries, arterioles, and venules of the lamina propria of the urinary bladder. The protein expression of AQP1 was significantly increased in the BOO group. CONCLUSIONS: This study showed that BOO causes a significant increase in the expression of AQP1. This may imply that AQP1 has a functional role in the detrusor instability that occurs in association with BOO.
Keyword
MeSH Terms
-
Animals
Aquaporin 1
Aquaporins
Arterioles
Blotting, Western
Capillaries
Female
Humans
Immunohistochemistry
Membrane Proteins
Membranes
Mucous Membrane
Rats
Rats, Sprague-Dawley
Salicylamides
Urethral Obstruction
Urinary Bladder
Urinary Bladder Neck Obstruction
Urothelium
Venules
Water Movements
Aquaporin 1
Aquaporins
Membrane Proteins
Salicylamides
Figure
Reference
-
1. de Groat WC. The urothelium in overactive bladder: passive bystander or active participant? Urology. 2004. 64:7–11.2. Johnson JA, Cavert HM, Lifson N, Visscher MB. Permeability of the bladder to water studied by means of isotropes. Am J Physiol. 1951. 165:87–92.3. Levinsky NG, Berliner RW. Changes in composition of the urine in ureter and bladder at low urine flow. Am J Physiol. 1959. 196:549–553.4. Nelson RA, Jones JD, Wahner HW, McGill DB, Code CF. Nitrogen metabolism in bears: urea metabolism in summer starvation and in winter sleep and role of urinary bladder in water and nitrogen conservation. Mayo Clin Proc. 1975. 50:141–146.5. Negrete HO, Lavelle JP, Berg J, Lewis SA, Zeidel ML. Permeability properties of the intact mammalian bladder epithelium. Am J Physiol. 1996. 271:–F886. –F894.6. Rapoport A, Nicholson TF, Yendt ER. Movement of electrolytes across the wall of the urinary bladder in dogs. Am J Physiol. 1960. 198:191–194.7. Turnbull GJ, Fellows GJ. Permeability of the urinary bladder of the rabbit. Rev Eur Etud Clin Biol. 1972. 17:745–749.8. Cross WR, Eardley I, Leese HJ, Southgate J. A biomimetic tissue from cultured normal human urothelial cells: analysis of physiological function. Am J Physiol Renal Physiol. 2005. 289:F459–F468.9. Butler RJ, Holland P, Gasson S, Norfolk S, Houghton L, Penney M. Exploring potential mechanisms in alarm treatment for primary nocturnal enuresis. Scand J Urol Nephrol. 2007. 41:407–413.10. Eaton DC. Intracellular sodium ion activity and sodium transport in rabbit urinary bladder. J Physiol. 1981. 316:527–544.11. Smith PR, Mackler SA, Weiser PC, Brooker DR, Ahn YJ, Harte BJ, et al. Expression and localization of epithelial sodium channel in mammalian urinary bladder. Am J Physiol. 1998. 274:F91–F96.12. Garty H, Palmer LG. Epithelial sodium channels: function, structure, and regulation. Physiol Rev. 1997. 77:359–396.13. Spector DA, Wade JB, Dillow R, Steplock DA, Weinman EJ. Expression, localization, and regulation of aquaporin-1 to -3 in rat urothelia. Am J Physiol Renal Physiol. 2002. 282:F1034–F1042.14. Rubenwolf PC, Georgopoulos NT, Clements LA, Feather S, Holland P, Thomas DF, et al. Expression and localization of aquaporin water channels in human urothelium in situ and in vitro. Eur Urol. 2009. 56:1013–1024.15. Yool AJ, Weinstein AM. New roles for old holes: ion channel function in aquaporin-1. News Physiol Sci. 2002. 17:68–72.16. Milsom I, Abrams P, Cardozo L, Roberts RG, Thüroff J, Wein AJ. How widespread are the symptoms of an overactive bladder and how are they managed? A population-based prevalence study. BJU Int. 2001. 87:760–766.17. Steers WD, Creedon DJ, Tuttle JB. Immunity of nerve growth factor prevents afferent plasticity following urinary bladder hypertrophy. J Urol. 1996. 155:379–385.18. Hohlbrugger G. The vesical blood-urine barrier: a relevant and dynamic interface between renal function and nervous bladder control. J Urol. 1995. 154:6–15.19. Schmidt-Nielsen B. Excretion in mammals: role of the renal pelvis in the modification of the urinary concentration and composition. Fed Proc. 1977. 36:2493–2503.20. Araki I, Du S, Kamiyama M, Mikami Y, Matsushita K, Komuro M, et al. Overexpression of epithelial sodium channels in epithelium of human urinary bladder with outlet obstruction. Urology. 2004. 64:1255–1260.21. Agre P, Preston GM, Smith BL, Jung JS, Raina S, Moon C, et al. Aquaporin CHIP: the archetypal molecular water channel. Am J Physiol. 1993. 265:F463–F476.22. Verkman AS. More than just water channels: unexpected cellular roles of aquaporins. J Cell Sci. 2005. 118:3225–3232.23. Maeda Y, Smith BL, Agre P, Knepper MA. Quantification of Aquaporin-CHIP water channel protein in microdissected renal tubules by fluorescence-based ELISA. J Clin Invest. 1995. 95:422–428.24. Gannon BJ, Carati CJ. Endothelial distribution of the membrane water channel molecule aquaporin-1: implications for tissue and lymph fluid physiology? Lymphat Res Biol. 2003. 1:55–66.25. Gannon BJ, Warnes GM, Carati CJ, Verco CJ. Aquaporin-1 expression in visceral smooth muscle cells of female rat reproductive tract. J Smooth Muscle Res. 2000. 36:155–167.26. Lu DY, Li Y, Bi ZW, Yu HM, Li XJ. Expression and immunohistochemical localization of aquaporin-1 in male reproductive organs of the mouse. Anat Histol Embryol. 2008. 37:1–8.27. Sellars ME, Sidhu PS. Pictorial review: ultrasound appearances of the rete testis. Eur J Ultrasound. 2001. 14:115–120.28. Zhou Q, Clarke L, Nie R, Carnes K, Lai LW, Lien YH, et al. Estrogen action and male fertility: roles of the sodium/hydrogen exchanger-3 and fluid reabsorption in reproductive tract function. Proc Natl Acad Sci USA. 2001. 98:14132–14137.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of Peripheral and Spinal alpha1-adrenoceptor in Bladder Overactivity Induced by Partial Bladder Outlet Obstruction in Rat
- Overexpression of Aquaporin-1 and Caveolin-1 in the Rat Urinary Bladder Urothelium Following Bladder Outlet Obstruction
- Changes in Detrusor and Urinary Growth Factors According to the Bladder Function after Partial Bladder Outlet Obstruction in Rat
- Effect of Bladder Outlet Obstruction on Blood Flow and Tissue Collagen in Rat Bladder
- Ultrastructural Changes of Detrusor Muscle by Partial Obstruction of the Bladder Outlet in the Rat