Korean J Physiol Pharmacol.
2011 Dec;15(6):371-382. 10.4196/kjpp.2011.15.6.371.
A Computational Model of the Temperature-dependent Changes in Firing Patterns in Aplysia Neurons
- Affiliations
-
- 1Department of Physics, Jeju National University, Jeju 690-756, Korea. nhyun@jejunu.ac.kr
- 2Department of Biological Sciences, Korea Advanced Institute of Science and Technology, Daejeon 305-701, Korea.
- 3Department of Biological Sciences, KAIST Institute for the Bio Century (KIB), KAIST, Daejeon 305-701, Korea.
- 4Department of Anatomy, Graduate School of Medicine, Kyungpook National University, Daegu 700-422, Korea.
- 5National Creative Research Initiative Center for Memory, Departments of Biological Sciences and Brain and Cognitive Sciences, College of Natural Science, Seoul National University, Seoul 151-747, Korea. kaang@snu.ac.kr
- KMID: 2285424
- DOI: http://doi.org/10.4196/kjpp.2011.15.6.371
Abstract
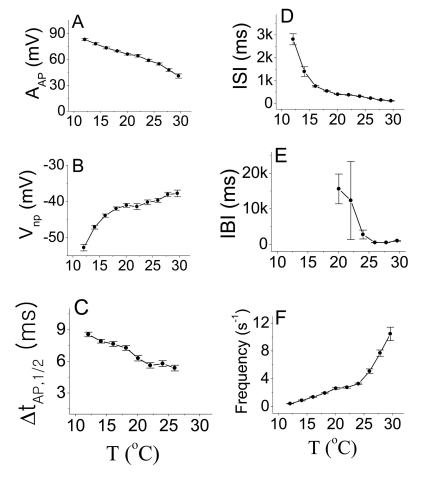
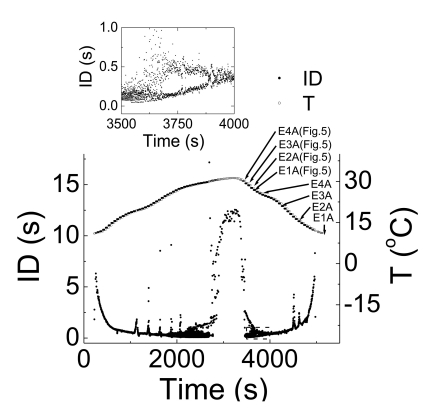
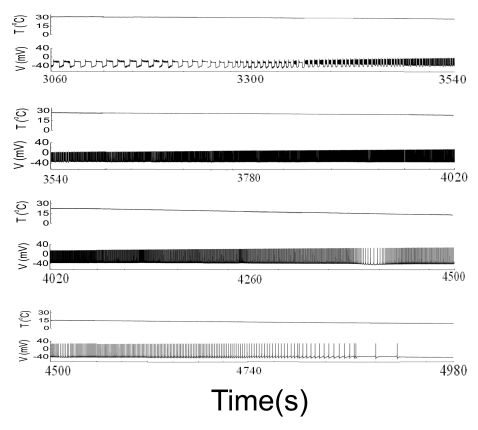
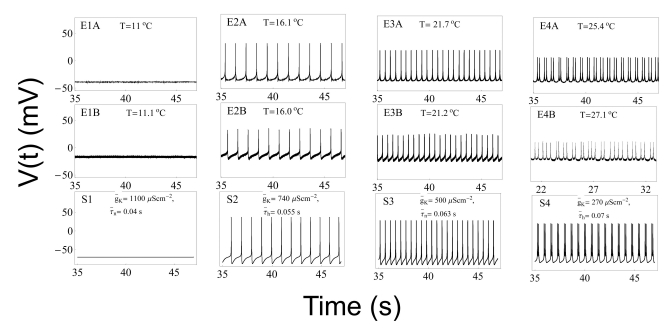
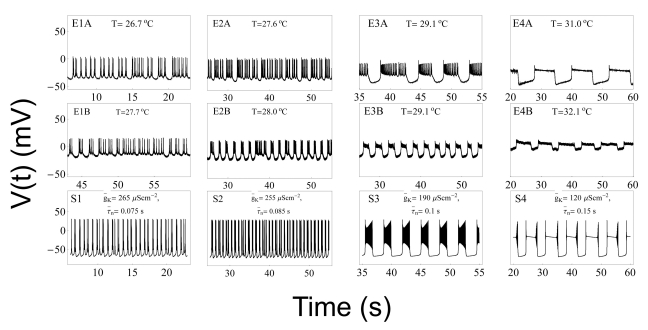
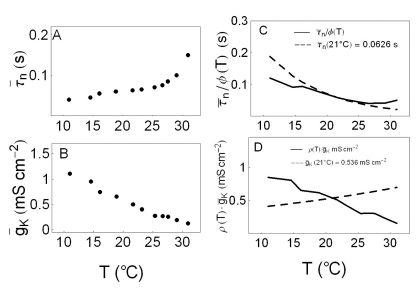
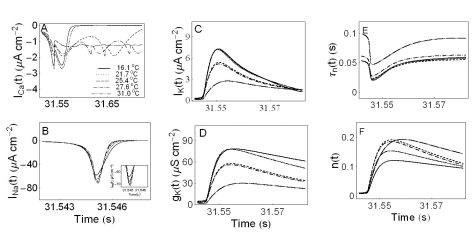
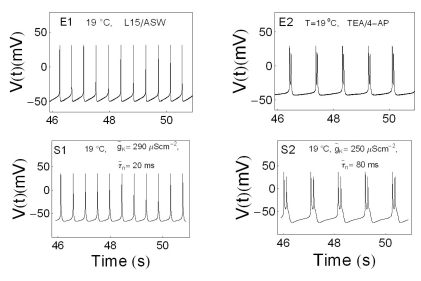
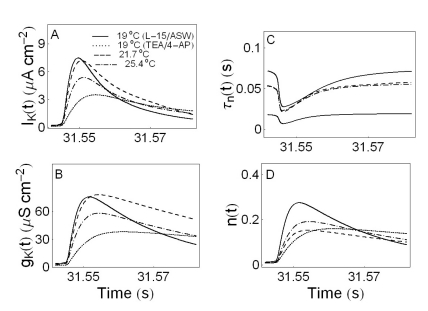
- We performed experiments using Aplysia neurons to identify the mechanism underlying the changes in the firing patterns in response to temperature changes. When the temperature was gradually increased from 11degrees C to 31degrees C the firing patterns changed sequentially from the silent state to beating, doublets, beating-chaos, bursting-chaos, square-wave bursting, and bursting-oscillation patterns. When the temperature was decreased over the same temperature range, these sequential changes in the firing patterns reappeared in reverse order. To simulate this entire range of spiking patterns we modified nonlinear differential equations that Chay and Lee made using temperature-dependent scaling factors. To refine the equations, we also analyzed the spike pattern changes in the presence of potassium channel blockers. Based on the solutions of these equations and potassium channel blocker experiments, we found that, as temperature increases, the maximum value of the potassium channel relaxation time constant, taun(t) increases, but the maximum value of the probabilities of openings for activation of the potassium channels, n(t) decreases. Accordingly, the voltage-dependent potassium current is likely to play a leading role in the temperature-dependent changes in the firing patterns in Aplysia neurons.
MeSH Terms
Figure
Reference
-
1. Carpenter DO. Temperature effects on pacemaker generation, membrane potential, and critical firing threshold in Aplysia neurons. J Gen Physiol. 1967; 50:1469–1484. PMID: 6034753.2. Moffett S, Wachtel H. Correlations between temperature effects on behavior in Aplysia and firing patterns of identified neurons. Mar Behav Physiol. 1976; 4:61–74.3. Carpenter DO, Alving BO. A contribution of an electrogenic Na+ pump to membrane potential in Aplysia neurons. J Gen Physiol. 1968; 52:1–21. PMID: 5742832.4. Fletcher SD, Ram JL. High temperature induces reversible silence in Aplysia R15 bursting pacemaker neuron. Comp Biochem Physiol. 1991; 98A:399–405.
Article5. Hakozaki S, Matsumoto M, Sasaki K. Temperature-sensitive activation of G-protein regulating the resting membrane conductance of Aplysia neurons. Jpn J Physiol. 1989; 39:115–130. PMID: 2498557.6. Chay TR. Bursting excitable cell models by a slow Ca2+ current. J Theor Biol. 1990; 142:305–315. PMID: 2160027.7. Chay TR, Lee YS. Bursting, beating, and chaos by two functionally distinct inward current inactivations in excitable cells. Ann N Y Acad Sci. 1990; 591:328–350. PMID: 2165373.
Article8. Hensel H, Huopaniemi T. Static and dynamic properties of warm fibres in the infraorbital nerve. Pflugers Arch. 1969; 309:1–10. PMID: 5815315.
Article9. Hensel H, Schäfer K. Static an dynamic activity of cold receptors in cats after long-term exposure to various temperatures. Pflugers Arch. 1982; 392:291–294. PMID: 7070961.10. Schäfer K, Braun HA, Kürten L. Analysis of cold and warm receptor activity in vampire bats and mice. Pflugers Arch. 1988; 412:188–194. PMID: 3174381.
Article11. Hensel H. Static and dynamic activity of warm receptors in Boa constrictor. Pflugers Arch. 1975; 353:191–199. PMID: 1168336.
Article12. Kim YD, Cho MH, Kwon SC. Myoplasmic [Ca2+], crossbridge phosphorylation and latch in rabbit bladder smooth muscle. Korean J Physiol Pharmacol. 2011; 15:171–177. PMID: 21860596.13. Kim KS, Shin DH, Nam JH, Park KS, Zhang YH, Kim WK, Kim SJ. Functional expression of TRPV4 cation channels in human mast cell line (HMC-1). Korean J Physiol Pharmacol. 2010; 14:419–425. PMID: 21311684.
Article14. Braun HA, Huber MT, Dewald M, Schäfer K, Voigt K. Computer simulations of neuronal signal transduction: the role of nonlinear dynamics and noise. Int J Bifurcation Chaos. 1998; 8:881–889.
Article15. Finke C, Freund JA, Rosa E Jr, Braun HA, Feudel U. On the role of subthreshold currents in the Huber-Braun cold receptor model. Chaos. 2010; 20:045107. PMID: 21198119.
Article16. Lim CS, Chung DY, Kaang BK. Partial anatomical and physiological characterization and dissociated cell culture of the nervous system of the marine mollusc Aplysia kurodai. Mol Cells. 1997; 7:399–407. PMID: 9264029.17. Hyun NG, Hyun KH, Lee K, Kaang BK. Temperature Dependence of Action Potential Parameters in Aplysia Neurons. Neurosignals. 2011; (in press).18. Hille B. Ion channels of excitable membranes. 2001. 3rd ed. Sunderland Mass: Sinauer;p. 366.19. Hyun NG. The brain and the mind. 2007. 1st ed. Jeju: Jeju Nat Univ;p. 321–338.20. Wang LY, Fedchyshyn MJ, Yang YM. Action potential evoked transmitter release in central synapses: insights from the developing calyx of Held. Mol Brain. 2009; 2:36. PMID: 19939269.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Analysis of temperature-dependent abnormal bursting patterns of neurons in Aplysia
- The Effect of Heat on the Spiking Patterns of the Cells in Aplysia
- A Computational Modeling Reveals That Strength of Inhibitory Input, E/I Balance, and Distance of Excitatory Input Modulate Thalamocortical Bursting Properties
- Modeling Long-term Spike Frequency Adaptation in SA-I Afferent Neurons Using an Izhikevich-based Biological Neuron Model
- Regional difference in spontaneous firing inhibition by GABA(A) and GABA(B) receptors in nigral dopamine neurons