Allergy Asthma Immunol Res.
2015 May;7(3):205-220. 10.4168/aair.2015.7.3.205.
Mold Allergens in Respiratory Allergy: From Structure to Therapy
- Affiliations
-
- 1Division of Immunopathology, Department of Pathophysiology and Allergy Research, Center for Pathophysiology, Infectiology and Immunology, Medical University of Vienna, Vienna, Austria. rudolf.valenta@meduniwien.ac.at
- 2The Molecular Biotechnology Section, University of Applied Sciences, Campus Vienna Biocenter, Vienna, Austria.
- KMID: 2260457
- DOI: http://doi.org/10.4168/aair.2015.7.3.205
Abstract
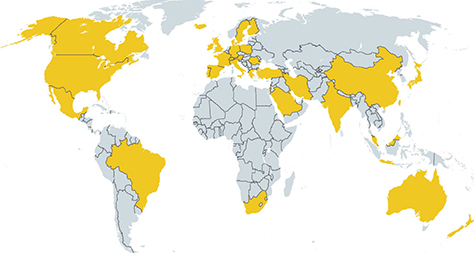
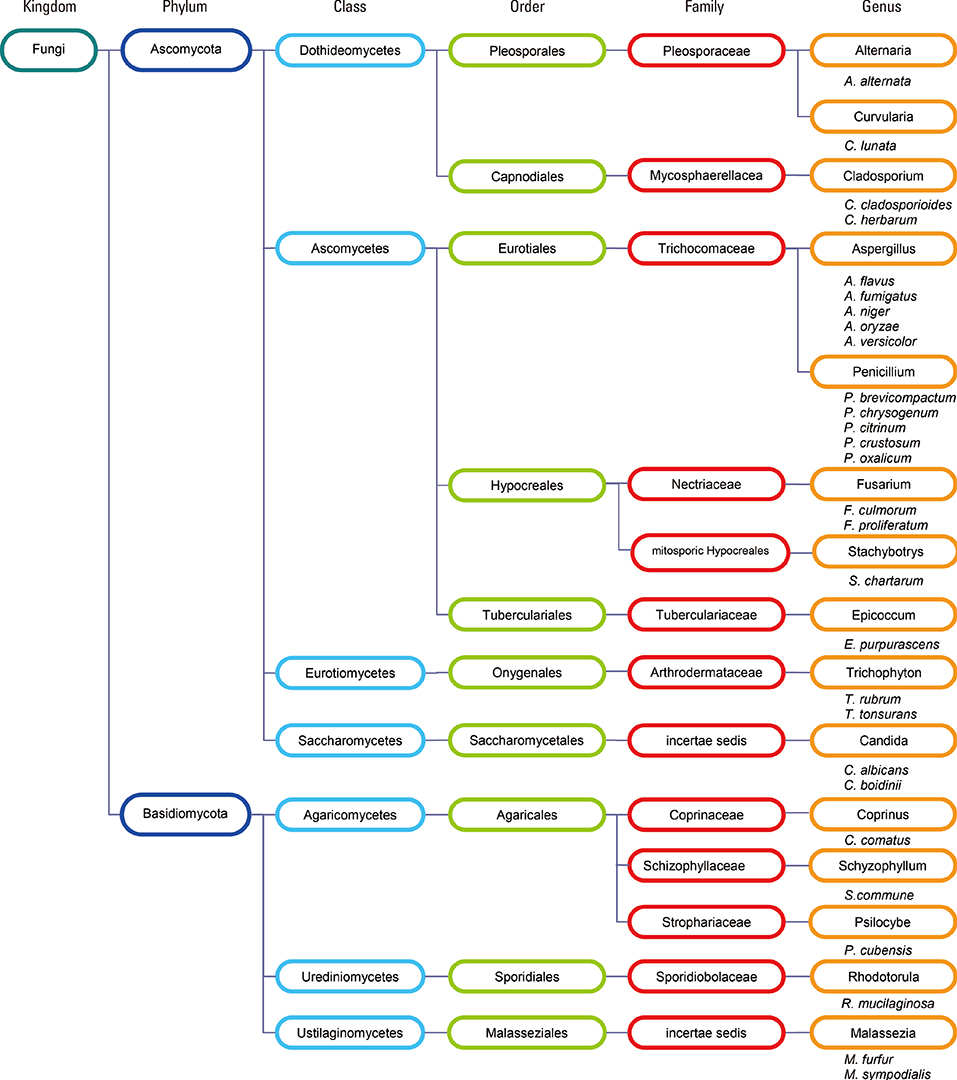
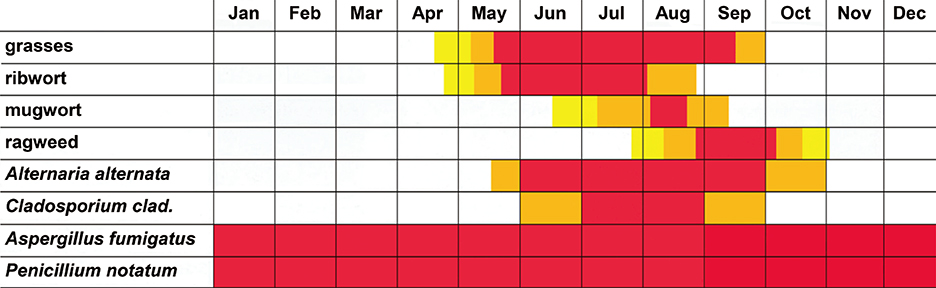
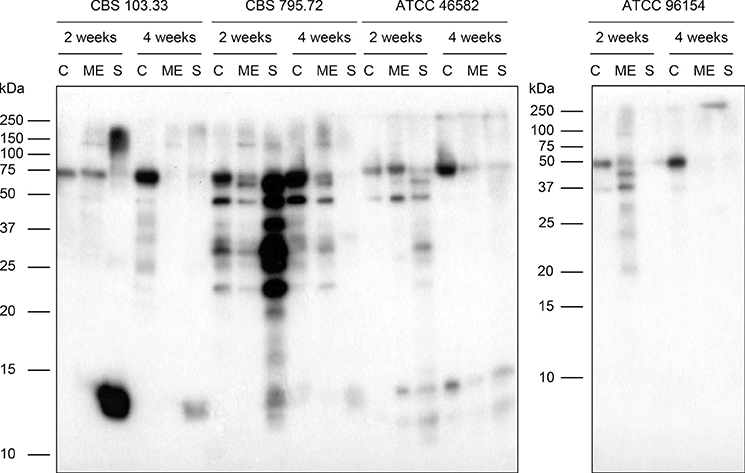
- Allergic reactions to fungi were described 300 years ago, but the importance of allergy to fungi has been underestimated for a long time. Allergens from fungi mainly cause respiratory and skin symptoms in sensitized patients. In this review, we will focus on fungi and fungal allergens involved in respiratory forms of allergy, such as allergic rhinitis and asthma. Fungi can act as indoor and outdoor respiratory allergen sources, and depending on climate conditions, the rates of sensitization in individuals attending allergy clinics range from 5% to 20%. Due to the poor quality of natural fungal allergen extracts, diagnosis of fungal allergy is hampered, and allergen-specific immunotherapy is rarely given. Several factors are responsible for the poor quality of natural fungal extracts, among which the influence of culture conditions on allergen contents. However, molecular cloning techniques have allowed us to isolate DNAs coding for fungal allergens and to produce a continuously growing panel of recombinant allergens for the diagnosis of fungal allergy. Moreover, technologies are now available for the preparation of recombinant and synthetic fungal allergen derivatives which can be used to develop safe vaccines for the treatment of fungal allergy.
MeSH Terms
Figure
Cited by 2 articles
-
Can Controlling Endoplasmic Reticulum Dysfunction Treat Allergic Inflammation in Severe Asthma With Fungal Sensitization?
Jae Seok Jeong, So Ri Kim, Yong Chul Lee
Allergy Asthma Immunol Res. 2018;10(2):106-120. doi: 10.4168/aair.2018.10.2.106.Association Between Sensitization to Mold and Impaired Pulmonary Function in Children With Asthma
Jung Hye Byeon, Soohyun Ri, Oyuntulga Amarsaikhan, Eunji Kim, So Hyun Ahn, Ic Sun Choi, Hyung Jin Kim, SungChul Seo, Wonsuck Yoon, Young Yoo
Allergy Asthma Immunol Res. 2017;9(6):509-516. doi: 10.4168/aair.2017.9.6.509.
Reference
-
1. von Pirquet C. Allergie. Münch Med Wochenschr. 1906; 53:1457–1458.2. Floyer J. Violent asthma after visiting a wine cellar. London: Innys and Parker;1745.3. Blackley C. Experimental research on the cause and nature of Catarrhus Aestivus (hay fever or hay asthma). London: Bailliere, Tindall and Cox;1873.4. Newson R, Strachan D, Corden J, Millington W. Fungal and other spore counts as predictors of admissions for asthma in the Trent region. Occup Environ Med. 2000; 57:786–792.5. Crameri R, Garbani M, Rhyner C, Huitema C. Fungi: the neglected allergenic sources. Allergy. 2014; 69:176–185.6. Simon-Nobbe B, Denk U, Pöll V, Rid R, Breitenbach M. The spectrum of fungal allergy. Int Arch Allergy Immunol. 2008; 145:58–86.7. Zureik M, Neukirch C, Leynaert B, Liard R, Bousquet J, Neukirch F. European Community Respiratory Health Survey. Sensitisation to airborne moulds and severity of asthma: cross sectional study from European Community respiratory health survey. Bmj. 2002; 325:411–414.8. Jo EJ, Kim MY, Lee SE, Lee SY, Kim MH, Song WJ, et al. Eosinophilic airway inflammation and airway hyperresponsiveness according to aeroallergen sensitization pattern in patients with lower airway symptoms. Allergy Asthma Immunol Res. 2014; 6:39–46.9. Iossifova YY, Reponen T, Ryan PH, Levin L, Bernstein DI, Lockey JE, et al. Mold exposure during infancy as a predictor of potential asthma development. Ann Allergy Asthma Immunol. 2009; 102:131–137.10. Garrett MH, Rayment PR, Hooper MA, Abramson MJ, Hooper BM. Indoor airborne fungal spores, house dampness and associations with environmental factors and respiratory health in children. Clin Exp Allergy. 1998; 28:459–467.11. Nolles G, Hoekstra MO, Schouten JP, Gerritsen J, Kauffman HF. Prevalence of immunoglobulin E for fungi in atopic children. Clin Exp Allergy. 2001; 31:1564–1570.12. Andriessen JW, Brunekreef B, Roemer W. Home dampness and respiratory health status in european children. Clin Exp Allergy. 1998; 28:1191–1200.13. Hagmolen of Ten Have W, van den Berg NJ, van der Palen J, van Aalderen WM, Bindels PJ. Residential exposure to mould and dampness is associated with adverse respiratory health. Clin Exp Allergy. 2007; 37:1827–1832.14. Meng J, Barnes CS, Rosenwasser LJ. Children's Mercy Center for Environmental Health. Identity of the fungal species present in the homes of asthmatic children. Clin Exp Allergy. 2012; 42:1448–1458.15. Targonski PV, Persky VW, Ramekrishnan V. Effect of environmental molds on risk of death from asthma during the pollen season. J Allergy Clin Immunol. 1995; 95:955–961.16. Rodriguez-Rajo FJ, Iglesias I, Jato V. Variation assessment of airborne Alternaria and Cladosporium spores at different bioclimatical conditions. Mycol Res. 2005; 109:497–507.17. Hamilton ED. Pollen and fungus spore counts. Proc R Soc Med. 1963; 56:220–221.18. Lacey J. Spore dispersal - its role in ecology and disease: the British contribution to fungal aerobiology. Mycol Res. 1996; 100:641–660.19. Pulimood TB, Corden JM, Bryden C, Sharples L, Nasser SM. Epidemic asthma and the role of the fungal mold Alternaria alternata. J Allergy Clin Immunol. 2007; 120:610–617.20. Hoffman DR, Kozak PP Jr, Gillman SA, Cummins LH, Gallup J. Isolation of spore specific allergens from Alternaria. Ann Allergy. 1981; 46:310–316.21. Wahl R, Oliver JD, Hauck PR, Winter HG, Maasch HJ. Comparison of Alternaria tenuis extracts prepared from different raw materials. Ann Allergy. 1989; 63:527–531.22. Paris S, Fitting C, Latgé JP, Herman D, Guinnepain MT, David B. Comparison of conidial and mycelial allergens of Alternaria alternata. Int Arch Allergy Appl Immunol. 1990; 92:1–8.23. Fadel R, David B, Paris S, Guesdon JL. Alternaria spore and mycelium sensitivity in allergic patients: in vivo and in vitro studies. Ann Allergy. 1992; 69:329–335.24. Licorish K, Novey HS, Kozak P, Fairshter RD, Wilson AF. Role of Alternaria and Penicillium spores in the pathogenesis of asthma. J Allergy Clin Immunol. 1985; 76:819–825.25. Matsuwaki Y, Uno K, Okushi T, Otori N, Moriyama H. Total and antigen- (fungi, mites and staphylococcal enterotoxins) specific IgEs in nasal polyps is related to local eosinophilic inflammation. Int Arch Allergy Immunol. 2013; 161:Suppl 2. 147–153.26. Niedoszytko M, Chełmińska M, Chełmiński K, Knopińska-Posłuszny W, Gruchała-Niedoszytko M, Jassem E. Late-phase allergic reaction in nasal provocation with fungal allergens. Allergy Asthma Proc. 2008; 29:35–39.27. Paris S, Debeaupuis JP, Prévost MC, Casotto M, Latgé JP. The 31 kd major allergen, Alt a I1563, of Alternaria alternata. J Allergy Clin Immunol. 1991; 88:902–908.28. Ibarrola I, Suárez-Cervera M, Arilla MC, Martínez A, Monteseirín J, Conde J, et al. Production profile of the major allergen Alt a 1 in Alternaria alternata cultures. Ann Allergy Asthma Immunol. 2004; 93:589–593.29. Twaroch TE, Arcalís E, Sterflinger K, Stöger E, Swoboda I, Valenta R. Predominant localization of the major Alternaria allergen Alt a 1 in the cell wall of airborne spores. J Allergy Clin Immunol. 2012; 129:1148–1149.30. Rivera-Mariani FE, Vysyaraju K, Negherbon J, Levetin E, Horner WE, Hartung T, et al. Comparison of the interleukin-1β-inducing potency of allergenic spores from higher fungi (basidiomycetes) in a cryopreserved human whole blood system. Int Arch Allergy Immunol. 2014; 163:154–162.31. Wada K, Kobayashi T, Matsuwaki Y, Moriyama H, Kita H. Alternaria inhibits double-stranded RNA-induced cytokine production through Toll-like receptor 3. Int Arch Allergy Immunol. 2013; 161:Suppl 2. 75–83.32. Kim HK, Lund S, Baum R, Rosenthal P, Khorram N, Doherty TA. Innate type 2 response to Alternaria extract enhances ryegrass-induced lung inflammation. Int Arch Allergy Immunol. 2014; 163:92–105.33. D'Amato G, Spieksma FT. Aerobiologic and clinical aspects of mould allergy in Europe. Allergy. 1995; 50:870–877.34. Oh SY, Fong JJ, Park MS, Chang L, Lim YW. Identifying airborne fungi in Seoul, Korea using metagenomics. J Microbiol. 2014; 52:465–472.35. Fröhlich-Nowoisky J, Pickersgill DA, Després VR, Pöschl U. High diversity of fungi in air particulate matter. Proc Natl Acad Sci U S A. 2009; 106:12814–12819.36. Kilic M, Ufuk Altintas D, Yilmaz M, Güneşer Kendirli S, Bingöl Karakoc G, Taskin E, et al. The effects of meteorological factors and Alternaria spore concentrations on children sensitised to Alternaria. Allergol Immunopathol (Madr). 2010; 38:122–128.37. Şakiyan N, Inceoglu Ö. Atmospheric concentrations of Cladosporium Link and Alternaria Nées spores in Ankara and the effects of meteorological factors. Turk J Bot. 2003; 27:77–81.38. Bowyer P, Fraczek M, Denning DW. Comparative genomics of fungal allergens and epitopes shows widespread distribution of closely related allergen and epitope orthologues. BMC Genomics. 2006; 7:251.39. Bernardis P, Agnoletto M, Puccinelli P, Parmiani S, Pozzan M. Injective versus sublingual immunotherapy in Alternaria tenuis allergic patients. J Investig Allergol Clin Immunol. 1996; 6:55–62.40. Green BJ, Yli-Panula E, Tovey ER. Halogen immunoassay, a new method for the detection of sensitization to fungal allergens; comparisons with conventional techniques. Allergol Int. 2006; 55:131–139.41. Cho SH, Seo SC, Schmechel D, Grinshpun SA, Reponen T. Aerodynamic characteristics and respiratory deposition of fungal fragments. Atmos Environ. 2005; 39:5454–5465.42. Barnes C, Schreiber K, Pacheco F, Landuyt J, Hu F, Portnoy J. Comparison of outdoor allergenic particles and allergen levels. Ann Allergy Asthma Immunol. 2000; 84:47–54.43. Feo Brito F, Alonso AM, Carnés J, Martín-Martín R, Fernández-Caldas E, Galindo PA, et al. Correlation between Alt a 1 levels and clinical symptoms in Alternaria alternata-monosensitized patients. J Investig Allergol Clin Immunol. 2012; 22:154–159.44. Mitakakis TZ, Barnes C, Tovey ER. Spore germination increases allergen release from Alternaria. J Allergy Clin Immunol. 2001; 107:388–390.45. Heinzerling L, Frew AJ, Bindslev-Jensen C, Bonini S, Bousquet J, Bresciani M, et al. Standard skin prick testing and sensitization to inhalant allergens across Europe--a survey from the GALEN network. Allergy. 2005; 60:1287–1300.46. Park HJ, Lee JH, Park KH, Ann HW, Jin MN, Choi SY, et al. A nationwide survey of inhalant allergens sensitization and levels of indoor major allergens in Korea. Allergy Asthma Immunol Res. 2014; 6:222–227.47. Lyons TW, Wakefield DB, Cloutier MM. Mold and Alternaria skin test reactivity and asthma in children in Connecticut. Ann Allergy Asthma Immunol. 2011; 106:301–307.48. Smit LA, Bouzigon E, Bousquet J, Le Moual N, Nadif R, Pin I, et al. Epidemiological Study on the Genetics and Environment of Asthma. Mold allergen sensitization in adult asthma according to integrin β3 polymorphisms and Toll-like receptor 2/+596 genotype. J Allergy Clin Immunol. 2011; 128:185–191.49. Wiszniewska M, Tymoszuk D, Nowakowska-Świrta E, Pałczyński C, Walusiak-Skorupa J. Mould sensitisation among bakers and farmers with work-related respiratory symptoms. Ind Health. 2013; 51:275–284.50. D'Amato G, Chatzigeorgiou G, Corsico R, Gioulekas D, Jäger L, Jäger S, et al. Evaluation of the prevalence of skin prick test positivity to Alternaria and Cladosporium in patients with suspected respiratory allergy. A European multicenter study promoted by the Subcommittee on Aerobiology and Environmental Aspects of Inhalant Allergens of the European Academy of Allergology and Clinical Immunology. Allergy. 1997; 52:711–716.51. de Benedictis FM, Franceschini F, Hill D, Naspitz C, Simons FE, Wahn U, et al. EPAAC Study Group. The allergic sensitization in infants with atopic eczema from different countries. Allergy. 2009; 64:295–303.52. Arbes SJ Jr, Gergen PJ, Elliott L, Zeldin DC. Prevalences of positive skin test responses to 10 common allergens in the US population: results from the third National Health and Nutrition Examination Survey. J Allergy Clin Immunol. 2005; 116:377–383.53. Rivera-Mariani FE, Nazario-Jiménez S, López-Malpica F, Bolaños-Rosero B. Skin test reactivity of allergic subjects to basidiomycetes' crude extracts in a tropical environment. Med Mycol. 2011; 49:887–891.54. Mari A, Schneider P, Wally V, Breitenbach M, Simon-Nobbe B. Sensitization to fungi: epidemiology, comparative skin tests, and IgE reactivity of fungal extracts. Clin Exp Allergy. 2003; 33:1429–1438.55. Niemeijer NR, de Monchy JG. Age-dependency of sensitization to aero-allergens in asthmatics. Allergy. 1992; 47:431–435.56. Ezeamuzie CI, Al-Ali S, Khan M, Hijazi Z, Dowaisan A, Thomson MS, et al. IgE-mediated sensitization to mould allergens among patients with allergic respiratory diseases in a desert environment. Int Arch Allergy Immunol. 2000; 121:300–307.57. Bartra J, Belmonte J, Torres-Rodriguez JM, Cistero-Bahima A. Sensitization to Alternaria in patients with respiratory allergy. Front Biosci. 2009; 14:3372–3379.58. Moral L, Roig M, Garde J, Alós A, Toral T, Fuentes MJ. Allergen sensitization in children with asthma and rhinitis: marked variations related to age and microgeographical factors. Allergol Immunopathol (Madr). 2008; 36:128–133.59. Cantani A, Ciaschi V. Epidemiology of alternaria alternata allergy: a prospective study in 6840 Italian asthmatic children. Eur Rev Med Pharmacol Sci. 2004; 8:289–294.60. Niederberger V, Stübner P, Spitzauer S, Kraft D, Valenta R, Ehrenberger K, et al. Skin test results but not serology reflect immediate type respiratory sensitivity: a study performed with recombinant allergen molecules. J Invest Dermatol. 2001; 117:848–851.61. Liang KL, Su MC, Jiang RS. Comparison of the skin test and ImmunoCAP system in the evaluation of mold allergy. J Chin Med Assoc. 2006; 69:3–6.62. Kespohl S, Maryska S, Zahradnik E, Sander I, Brüning T, Raulf-Heimsoth M. Biochemical and immunological analysis of mould skin prick test solution: current status of standardization. Clin Exp Allergy. 2013; 43:1286–1296.63. Soeria-Atmadja D, Onell A, Borgå A. IgE sensitization to fungi mirrors fungal phylogenetic systematics. J Allergy Clin Immunol. 2010; 125:1379–1386.64. Valenta R, Ferreira F, Focke-Tejkl M, Linhart B, Niederberger V, Swoboda I, et al. From allergen genes to allergy vaccines. Annu Rev Immunol. 2010; 28:211–241.65. Limacher A, Kloer DP, Flückiger S, Folkers G, Crameri R, Scapozza L. The crystal structure of Aspergillus fumigatus cyclophilin reveals 3D domain swapping of a central element. Structure. 2006; 14:185–195.66. Glaser AG, Limacher A, Flückiger S, Scheynius A, Scapozza L, Crameri R. Analysis of the cross-reactivity and of the 1.5 A crystal structure of the Malassezia sympodialis Mala s 6 allergen, a member of the cyclophilin pan-allergen family. Biochem J. 2006; 396:41–49.67. Limacher A, Glaser AG, Meier C, Schmid-Grendelmeier P, Zeller S, Scapozza L, et al. Cross-reactivity and 1.4-A crystal structure of Malassezia sympodialis thioredoxin (Mala s 13), a member of a new pan-allergen family. J Immunol. 2007; 178:389–396.68. Nüss D, Goettig P, Magler I, Denk U, Breitenbach M, Schneider PB, et al. Crystal structure of the NADP-dependent mannitol dehydrogenase from Cladosporium herbarum: Implications for oligomerisation and catalysis. Biochimie. 2010; 92:985–993.69. Chruszcz M, Chapman MD, Osinski T, Solberg R, Demas M, Porebski PJ, et al. Alternaria alternata allergen Alt a 1: a unique β-barrel protein dimer found exclusively in fungi. J Allergy Clin Immunol. 2012; 130:241–247.70. Wagner GE, Gutfreund S, Fauland K, Keller W, Valenta R, Zangger K. Backbone resonance assignment of Alt a 1, a unique β-barrel protein and the major allergen of Alternaria alternata. Biomol NMR Assign. 2014; 8:229–231.71. Chen JC, Chiu LL, Lee KL, Huang WN, Chuang JG, Liao HK, et al. Identification of critical amino acids in an immunodominant IgE epitope of Pen c 13, a major allergen from Penicillium citrinum. PLoS One. 2012; 7:e34627.72. Twaroch TE, Focke M, Fleischmann K, Balic N, Lupinek C, Blatt K, et al. Carrier-bound Alt a 1 peptides without allergenic activity for vaccination against Alternaria alternata allergy. Clin Exp Allergy. 2012; 42:966–975.73. Nair S, Kukreja N, Singh BP, Arora N. Identification of B cell epitopes of alcohol dehydrogenase allergen of Curvularia lunata. PLoS One. 2011; 6:e20020.74. Herrera-Mozo I, Ferrer B, Luís Rodriguez-Sanchez J, Juarez C. Description of a novel panallergen of cross-reactivity between moulds and foods. Immunol Invest. 2006; 35:181–197.75. Simon-Nobbe B, Probst G, Kajava AV, Oberkofler H, Susani M, Crameri R, et al. IgE-binding epitopes of enolases, a class of highly conserved fungal allergens. J Allergy Clin Immunol. 2000; 106:887–895.76. Chang CY, Chou H, Tam M, Tang RB, Lai HY, Shen HD. Characterization of enolase allergen from Rhodotorula mucilaginosa. J Biomed Sci. 2002; 9:645–655.77. Ito K, Ishiguro A, Kanbe T, Tanaka K, Torii S. Detection of IgE antibody against Candida albicans enolase and its crossreactivity to Saccharomyces cerevisiae enolase. Clin Exp Allergy. 1995; 25:522–528.78. Flückiger S, Scapozza L, Mayer C, Blaser K, Folkers G, Crameri R. Immunological and structural analysis of IgE-mediated cross-reactivity between manganese superoxide dismutases. Int Arch Allergy Immunol. 2002; 128:292–303.79. Horner WE, Reese G, Lehrer SB. Identification of the allergen Psi c 2 from the basidiomycete Psilocybe cubensis as a fungal cyclophilin. Int Arch Allergy Immunol. 1995; 107:298–300.80. Flückiger S, Fijten H, Whitley P, Blaser K, Crameri R. Cyclophilins, a new family of cross-reactive allergens. Eur J Immunol. 2002; 32:10–17.81. Shankar J, Gupta PD, Sridhara S, Singh BP, Gaur SN, Arora N. Immunobiochemical analysis of cross-reactive glutathione-S-transferase allergen from different fungal sources. Immunol Invest. 2005; 34:37–51.82. Glaser AG, Menz G, Kirsch AI, Zeller S, Crameri R, Rhyner C. Auto- and cross-reactivity to thioredoxin allergens in allergic bronchopulmonary aspergillosis. Allergy. 2008; 63:1617–1623.83. Chou H, Wu KG, Yeh CC, Tai HY, Tam MF, Chen YS, et al. The Transaldolase, a Novel Allergen of Fusarium proliferatum, Demonstrates IgE Cross-Reactivity with Its Human Analogue. PLoS One. 2014; 9(7):e103488.84. van Ree R. Carbohydrate epitopes and their relevance for the diagnosis and treatment of allergic diseases. Int Arch Allergy Immunol. 2002; 129:189–197.85. Altmann F. The role of protein glycosylation in allergy. Int Arch Allergy Immunol. 2007; 142:99–115.86. Kobayashi T, Iijima K, Radhakrishnan S, Mehta V, Vassallo R, Lawrence CB, et al. Asthma-related environmental fungus, Alternaria, activates dendritic cells and produces potent Th2 adjuvant activity. J Immunol. 2009; 182:2502–2510.87. Tai HY, Tam MF, Chou H, Peng HJ, Su SN, Perng DW, et al. Pen ch 13 allergen induces secretion of mediators and degradation of occludin protein of human lung epithelial cells. Allergy. 2006; 61:382–388.88. Borger P, Koëter GH, Timmerman JA, Vellenga E, Tomee JF, Kauffman HF. Proteases from Aspergillus fumigatus induce interleukin (IL)-6 and IL-8 production in airway epithelial cell lines by transcriptional mechanisms. J Infect Dis. 1999; 180:1267–1274.89. Kauffman HF, Tomee JF, van de Riet MA, Timmerman AJ, Borger P. Protease-dependent activation of epithelial cells by fungal allergens leads to morphologic changes and cytokine production. J Allergy Clin Immunol. 2000; 105:1185–1193.90. Lamhamedi-Cherradi SE, Martin RE, Ito T, Kheradmand F, Corry DB, Liu YJ, et al. Fungal proteases induce Th2 polarization through limited dendritic cell maturation and reduced production of IL-12. J Immunol. 2008; 180:6000–6009.91. Boitano S, Flynn AN, Sherwood CL, Schulz SM, Hoffman J, Gruzinova I, et al. Alternaria alternata serine proteases induce lung inflammation and airway epithelial cell activation via PAR2. Am J Physiol Lung Cell Mol Physiol. 2011; 300:L605–L614.92. Snelgrove RJ, Gregory LG, Peiró T, Akthar S, Campbell GA, Walker SA, et al. Alternaria-derived serine protease activity drives IL-33 mediated asthma exacerbations. J Allergy Clin Immunol. 2014; 134:583–592.93. Chen JC, Chuang JG, Su YY, Chiang BL, Lin YS, Chow LP. The protease allergen Pen c 13 induces allergic airway inflammation and changes in epithelial barrier integrity and function in a murine model. J Biol Chem. 2011; 286:26667–26679.94. Malling HJ, Dreborg S, Weeke B. Diagnosis and immunotherapy of mould allergy. III. Diagnosis of Cladosporium allergy by means of symptom score, bronchial provocation test, skin prick test, RAST, CRIE and histamine release. Allergy. 1986; 41:57–67.95. Malling HJ, Agrell B, Croner S, Dreborg S, Foucard T, Kjellman M, et al. Diagnosis and immunotherapy of mould allergy. I. Screening for mould allergy. Allergy. 1985; 40:108–114.96. Palma-Carlos ML, Palma-Carlos AG. "In vivo" and "in vitro": tests in the diagnosis of trichophyton allergy. Eur Ann Allergy Clin Immunol. 2007; 39:328–332.97. Schumacher MJ, Jeffrey SE. Variability of Alternaria alternata: biochemical and immunological characteristics of culture filtrates from seven isolates. J Allergy Clin Immunol. 1976; 58:263–277.98. Steringer I, Aukrust L, Einarsson R. Variability of antigenicity/allergenicity in different strains of Alternaria alternata. Int Arch Allergy Appl Immunol. 1987; 84:190–197.99. Vijay HM, Young NM, Jackson GE, White GP, Bernstein IL. Studies on Alternaria allergens. V. Comparative biochemical and immunological studies of three isolates of Alternaria tenuis cultured on synthetic media. Int Arch Allergy Appl Immunol. 1985; 78:37–42.100. Portnoy J, Pacheco F, Barnes C, Upadrashta B, Crenshaw R, Esch R. Selection of representative Alternaria strain groups on the basis of morphology, enzyme profile, and allergen content. J Allergy Clin Immunol. 1993; 91:773–782.101. Martínez J, Gutiérrez A, Postigo I, Cardona G, Guisantes J. Variability of Alt a 1 expression by different strains of Alternaria alternata. J Investig Allergol Clin Immunol. 2006; 16:279–282.102. Sáenz-de-Santamaría M, Guisantes JA, Martínez J. Enzymatic activities of Alternaria alternata allergenic extracts and its major allergen (Alt a 1). Mycoses. 2006; 49:288–292.103. Marquardt H. Spontaneous mutations in fungi. Humangenetik. 1972; 16:7–18.104. Portnoy J, Pacheco F, Ballam Y, Barnes C. The effect of time and extraction buffers on residual protein and allergen content of extracts derived from four strains of Alternaria. J Allergy Clin Immunol. 1993; 91:930–938.105. Bisht V, Singh BP, Arora N, Sridhara S, Gaur SN. Allergens of Epicoccum nigrum grown in different media for quality source material. Allergy. 2000; 55:274–280.106. Vailes L, Sridhara S, Cromwell O, Weber B, Breitenbach M, Chapman M. Quantitation of the major fungal allergens, Alt a 1 and Asp f 1, in commercial allergenic products. J Allergy Clin Immunol. 2001; 107:641–646.107. Brander KA, Borbély P, Crameri R, Pichler WJ, Helbling A. IgE-binding proliferative responses and skin test reactivity to Cop c 1, the first recombinant allergen from the basidiomycete Coprinus comatus. J Allergy Clin Immunol. 1999; 104:630–636.108. Hemmann S, Menz G, Ismail C, Blaser K, Crameri R. Skin test reactivity to 2 recombinant Aspergillus fumigatus allergens in A fumigatus-sensitized asthmatic subjects allows diagnostic separation of allergic bronchopulmonary aspergillosis from fungal sensitization. J Allergy Clin Immunol. 1999; 104:601–607.109. Unger A, Stöger P, Simon-Nobbe B, Susani M, Crameri R, Ebner C, Hintner H, Breitenbach M. Clinical testing of recombinant allergens of the mold Alternaria alternata. Int Arch Allergy Immunol. 1999; 118:220–221.110. Postigo I, Gutiérrez-Rodríguez A, Fernández J, Guisantes JA, Suñén E, Martínez J. Diagnostic value of Alt a 1, fungal enolase and manganese-dependent superoxide dismutase in the component-resolved diagnosis of allergy to Pleosporaceae. Clin Exp Allergy. 2011; 41:443–451.111. Katotomichelakis M, Anastassakis K, Gouveris H, Tripsianis G, Paraskakis E, Maroudias N, et al. Clinical significance of Alternaria alternata sensitization in patients with allergic rhinitis. Am J Otolaryngol. 2012; 33:232–238.112. Bousquet J, Lockey R, Malling HJ, Alvarez-Cuesta E, Canonica GW, Chapman MD, et al. World Health Organization. American academy of Allergy, Asthma and Immunology. Allergen immunotherapy: therapeutic vaccines for allergic diseases. Ann Allergy Asthma Immunol. 1998; 81:401–405.113. Vermani M, Vijayan VK, Kausar MA, Agarwal MK. Quantification of airborne Aspergillus allergens: redefining the approach. J Asthma. 2010; 47:754–761.114. Barnes C, Pacheco F, Dhar M, Portnoy J. Alternaria and Cladosporium Fungal Allergen Epitopes are Denatured by Sodium Hypochlorite. World Allergy Organ J. 2009; 2:296–302.115. Frew AJ. Injection immunotherapy. British Society for Allergy and Clinical Immunology Working Party. BMJ. 1993; 307:919–923.116. Kaad PH, Ostergaard PA. The hazard of mould hyposensitization in children with asthma. Clin Allergy. 1982; 12:317–320.117. Ostergaard PA, Kaad PH, Kristensen T. A prospective study on the safety of immunotherapy in children with severe asthma. Allergy. 1986; 41:588–593.118. Martorell A, Sole A, Diez LV, Belenguer J, Sanz J, Torro MI, Cerda JC, Álvarez V. Inmunoterapia en alergia a hongos. Rev Esp Alergol Inmunol Clin. 1986; 1:188–192.119. Tabar AI, Lizaso MT, García BE, Echechipía S, Olaguibel JM, Rodríguez A. Tolerance of immunotherapy with a standardized extract of Alternaria tenuis in patients with rhinitis and bronchial asthma. J Investig Allergol Clin Immunol. 2000; 10:327–333.120. Moreno C, Fernández Távora L, Justicia JL, Cabeza N, Vidal C. Investigation of an adequate immunotherapy induction schedule with an Alternaria tenuis extract. Alergol Inmunol Clin. 2001; 16:133–137.121. Martínez-Cañavate A, Eseverri JL, Ródenas R, Tabar AI, Gardee J, Torres J, et al. Evaluation of paediatric tolerance to an extract of Alternaria alternata under two treatment regimes. A multicentre study. Allergol Immunopathol (Madr). 2005; 33:138–141.122. Dreborg S, Agrell B, Foucard T, Kjellman NI, Koivikko A, Nilsson S. A double-blind, multicenter immunotherapy trial in children, using a purified and standardized Cladosporium herbarum preparation. I. Clinical results. Allergy. 1986; 41:131–140.123. Karlsson R, Agrell B, Dreborg S, Foucard T, Kjellman NI, Koivikko A, et al. A double-blind, multicenter immunotherapy trial in children, using a purified and standardized Cladosporium herbarum preparation. II. In vitro results. Allergy. 1986; 41:141–150.124. Malling HJ, Dreborg S, Weeke B. Diagnosis and immunotherapy of mould allergy. V. Clinical efficacy and side effects of immunotherapy with Cladosporium herbarum. Allergy. 1986; 41:507–519.125. Malling HJ, Dreborg S, Weeke B. Diagnosis and immunotherapy of mould allergy. VI. IgE-mediated parameters during a one-year placebo-controlled study of immunotherapy with Cladosporium. Allergy. 1987; 42:305–314.126. Malling HJ, Djurup R. Diagnosis and immunotherapy of mould allergy. VII. IgG subclass response and relation to the clinical efficacy of immunotherapy with Cladosporium. Allergy. 1988; 43:60–70.127. Malling HJ, Stahl Skov P. Diagnosis and immunotherapy of mould allergy. VIII. Qualitative and quantitative estimation of IgE in Cladosporium immunotherapy. Allergy. 1988; 43:228–238.128. Horst M, Hejjaoui A, Horst V, Michel FB, Bousquet J. Double-blind, placebo-controlled rush immunotherapy with a standardized Alternaria extract. J Allergy Clin Immunol. 1990; 85:460–472.129. Criado Molina A, Guerra Pasadas F, Daza Muñoz JC, Moreno Aguilar C, Almeda Llamas E, Muñoz Gomariz E, et al. Immunotherapy with an oral Alternaria extract in childhood asthma. Clinical safety and efficacy and effects on in vivo and in vitro parameters. Allergol Immunopathol (Madr). 2002; 30:319–330.130. Lizaso MT, Martínez A, Asturias JA, Algorta J, Madariaga B, Labarta N, et al. Biological standardization and maximum tolerated dose estimation of an Alternaria alternata allergenic extract. J Investig Allergol Clin Immunol. 2006; 16:94–103.131. Tabar AI, Lizaso MT, García BE, Gómez B, Echechipía S, Aldunate MT, et al. Double-blind, placebo-controlled study of Alternaria alternata immunotherapy: clinical efficacy and safety. Pediatr Allergy Immunol. 2008; 19:67–75.132. Lizaso MT, Tabar AI, García BE, Gómez B, Algorta J, Asturias JA, et al. Double-blind, placebo-controlled Alternaria alternata immunotherapy: in vivo and in vitro parameters. Pediatr Allergy Immunol. 2008; 19:76–81.133. Kuna P, Kaczmarek J, Kupczyk M. Efficacy and safety of immunotherapy for allergies to Alternaria alternata in children. J Allergy Clin Immunol. 2011; 127:502–508.134. Cantani A, Businco E, Maglio A. Alternaria allergy: a three-year controlled study in children treated with immunotherapy. Allergol Immunopathol (Madr). 1988; 16:1–4.135. Kiliç M, Altintaş DU, Yilmaz M, Bingöl-Karakoç G, Burgut R, Güneşer-Kendirli S. Evaluation of efficacy of immunotherapy in children with asthma monosensitized to Alternaria. Turk J Pediatr. 2011; 53:285–294.136. Valenta R. The future of antigen-specific immunotherapy of allergy. Nat Rev Immunol. 2002; 2:446–453.137. Linhart B, Valenta R. Vaccines for allergy. Curr Opin Immunol. 2012; 24:354–360.138. Linhart B, Valenta R. Mechanisms underlying allergy vaccination with recombinant hypoallergenic allergen derivatives. Vaccine. 2012; 30:4328–4335.139. Haselden BM, Kay AB, Larché M. Immunoglobulin E-independent major histocompatibility complex-restricted T cell peptide epitope-induced late asthmatic reactions. J Exp Med. 1999; 189:1885–1894.140. Oldfield WL, Kay AB, Larché M. Allergen-derived T cell peptide-induced late asthmatic reactions precede the induction of antigen-specific hyporesponsiveness in atopic allergic asthmatic subjects. J Immunol. 2001; 167:1734–1739.141. Focke-Tejkl M, Valenta R. Safety of engineered allergen-specific immunotherapy vaccines. Curr Opin Allergy Clin Immunol. 2012; 12:555–563.142. Focke M, Mahler V, Ball T, Sperr WR, Majlesi Y, Valent P, et al. Nonanaphylactic synthetic peptides derived from B cell epitopes of the major grass pollen allergen, Phl p 1, for allergy vaccination. Faseb J. 2001; 15:2042–2044.143. Focke M, Linhart B, Hartl A, Wiedermann U, Sperr WR, Valent P, et al. Non-anaphylactic surface-exposed peptides of the major birch pollen allergen, Bet v 1, for preventive vaccination. Clin Exp Allergy. 2004; 34:1525–1533.144. Edlmayr J, Niespodziana K, Linhart B, Focke-Tejkl M, Westritschnig K, Scheiblhofer S, et al. A combination vaccine for allergy and rhinovirus infections based on rhinovirus-derived surface protein VP1 and a nonallergenic peptide of the major timothy grass pollen allergen Phl p 1. J Immunol. 2009; 182:6298–6306.145. Zhang L, Curran IH, Muradia G, De Vouge MW, Rode H, Vijay HM. N-terminus of a major allergen, Alt a I, of Alternaria alternata defined to be an epitope. Int Arch Allergy Immunol. 1995; 108:254–259.146. Kurup VP, Vijay HM, Kumar V, Castillo L, Elms N. IgE binding synthetic peptides of Alt a 1, a major allergen of Alternaria alternata. Peptides. 2003; 24:179–185.147. Lai HY, Tam MF, Chou H, Lee SS, Tai HY, Shen HD. Molecular and structural analysis of immunoglobulin E-binding epitopes of Pen ch 13, an alkaline serine protease major allergen from Penicillium chrysogenum. Clin Exp Allergy. 2004; 34:1926–1933.148. Yu CJ, Chen YM, Su SN, Forouhar F, Lee SH, Chow LP. Molecular and immunological characterization and IgE epitope mapping of Pen n 18, a major allergen of Penicillium notatum. Biochem J. 2002; 363:707–715.149. Banerjee B, Greenberger PA, Fink JN, Kurup VP. Conformational and linear B-cell epitopes of Asp f 2, a major allergen of Aspergillus fumigatus, bind differently to immunoglobulin E antibody in the sera of allergic bronchopulmonary aspergillosis patients. Infect Immun. 1999; 67:2284–2291.150. Vilhelmsson M, Glaser AG, Martinez DB, Schmidt M, Johansson C, Rhyner C, et al. Mutational analysis of amino acid residues involved in IgE-binding to the Malassezia sympodialis allergen Mala s 11. Mol Immunol. 2008; 46:294–303.151. Cheng TT, Tam MF, Chou H, Tai HY, Shen HD. Lys89, Lys90, and Phe91 are critical core amino acid residues of the Pen ch 18 major fungal allergen recognized by human IgE antibodies. Biochem Biophys Res Commun. 2008; 375:671–674.152. Shankar J, Singh BP, Gaur SN, Arora N. Engineered Alt a 13 fragment of Alternaria alternata abrogated IgE binding without affecting T-cell stimulation. J Clin Immunol. 2009; 29:63–70.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Early Exposure to Inhalant Allergens and Sensitization
- A Study on Allergenicity and Purification of Allergens from Alternaria
- Changes of aeroallergen sensitization in childhood
- Change of causative inhalant allergens in respiratory allergic patients in Chungbuk district
- Changing sensitization rate to common inhalant allergens in our environment