Korean J Hematol.
2010 Dec;45(4):253-259. 10.5045/kjh.2010.45.4.253.
Additional rituximab-CHOP (R-CHOP) versus involved-field radiotherapy after a brief course of R-CHOP in limited, non-bulky diffuse large B-cell lymphoma: a retrospective analysis
- Affiliations
-
- 1Department of Internal Medicine, Gachon University Gil Hospital, Gachon University of Medicine and Science, Graduate School of Medicine, Incheon, Korea. jhagnes@gilhospital.com
- 2Department of Therapeutic Radiology & Oncology, Gachon University Gil Hospital, Gachon University of Medicine and Science, Graduate School of Medicine, Incheon, Korea.
- KMID: 2252036
- DOI: http://doi.org/10.5045/kjh.2010.45.4.253
Abstract
- BACKGROUND
Standard treatment for stage I or non-bulky stage II diffuse large B-cell lymphoma (DLBCL) has been either a brief course of chemotherapy plus involved-field radiotherapy (IFRT) or prolonged cycles of chemotherapy. The introduction of rituximab has necessitated re-evaluation of the treatment for limited disease (LD) DLBCL.
METHODS
Thirty-nine LD DLBCL patients (median age, 52 years; range, 24-85) treated with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisolone (R-CHOP) were retrospectively analyzed. Treatment outcomes were evaluated, and toxicity, event-free survival (EFS), and overall survival (OS) were compared according to the treatment and risk factors.
RESULTS
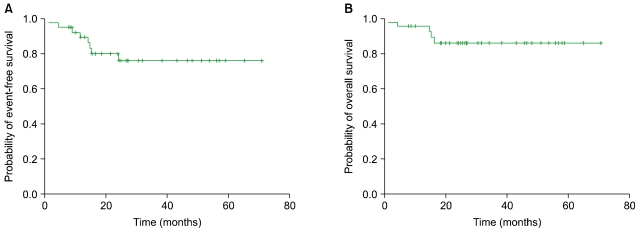
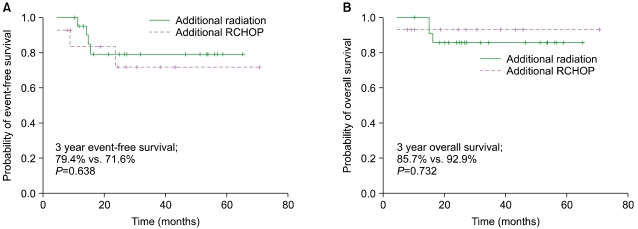
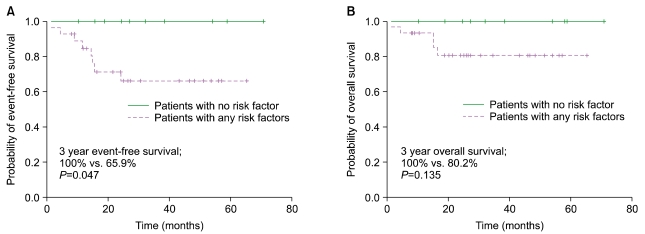
The median follow-up duration was 34.6 months (range, 9.1-65.4). The 3-year EFS and OS were 76.0% and 86.0%, respectively. Among the 36 patients who underwent either 3-4 cycles of R-CHOP followed by IFRT (N=22) or 6-8 cycles of R-CHOP (N=14), there was no difference in the 3-year EFS (79.4% vs. 71.6%, P=0.638) and 3-year OS (85.7% vs. 92.9%, P=0.732). Severe neutropenia and neutropenic fever were more frequent in patients treated with R-CHOP alone, with 1 treatment-related mortality. Among the IFRT patients, 1 required hospital admission for IFRT-related complications. No events or deaths were reported among patients without adverse risk factors.
CONCLUSION
The difference in outcomes between the 2 treatment options was not significant. Analysis of treatment outcomes suggested that baseline characteristics and expected toxicities should be considered in LD DLBCL treatment. Further studies are needed to define the optimal treatment in the rituximab era.
MeSH Terms
Figure
Cited by 2 articles
-
A therapeutic dilemma between the two "R"s: additional rituximab or radiotherapy for limited, non-bulky diffuse large B-cell lymphoma
Seok Jin Kim
Korean J Hematol. 2011;46(1):57-58. doi: 10.5045/kjh.2011.46.1.57.Abbreviated chemotherapy for limited-stage diffuse large B-cell lymphoma after complete resection
Jungmin Jo, Dok Hyun Yoon, Sang Wook Lee, Chan-Sik Park, Jooryung Huh, Kyoungmin Lee, Eun Hee Kang, Shin Kim, Cheolwon Suh
Blood Res. 2014;49(2):115-119. doi: 10.5045/br.2014.49.2.115.
Reference
-
1. Anderson JR, Armitage JO, Weisenburger DD. Epidemiology of the non-Hodgkin's lymphomas: distributions of the major subtypes differ by geographic locations. Non-Hodgkin's Lymphoma Classification Project. Ann Oncol. 1998; 9:717–720. PMID: 9739436.2. Fisher RI, Miller TP, O'Connor OA. Diffuse aggressive lymphoma. Hematology Am Soc Hematol Educ Program. 2004; 221–236. PMID: 15561685.
Article3. Miller TP, Dahlberg S, Cassady JR, et al. Chemotherapy alone compared with chemotherapy plus radiotherapy for localized intermediate-and high-grade non-Hodgkin's lymphoma. N Engl J Med. 1998; 339:21–26. PMID: 9647875.4. Horning SJ, Weller E, Kim K, et al. Chemotherapy with or without radiotherapy in limited-stage diffuse aggressive non-Hodgkin's lymphoma: Eastern Cooperative Oncology Group study 1484. J Clin Oncol. 2004; 22:3032–3038. PMID: 15210738.
Article5. Shenkier TN, Voss N, Fairey R, et al. Brief chemotherapy and involved-region irradiation for limited-stage diffuse large-cell lymphoma: an 18-year experience from the British Columbia Cancer Agency. J Clin Oncol. 2002; 20:197–204. PMID: 11773170.
Article6. Miller TP. The limits of limited stage lymphoma. J Clin Oncol. 2004; 22:2982–2984. PMID: 15210737.
Article7. Reyes F, Lepage E, Ganem G, et al. ACVBP versus CHOP plus radiotherapy for localized aggressive lymphoma. N Engl J Med. 2005; 352:1197–1205. PMID: 15788496.
Article8. Cabanillas F, Bodey GP, Freireich EJ. Management with chemotherapy only of stage I and II malignant lymphoma of aggressive histologic types. Cancer. 1980; 46:2356–2359. PMID: 7002279.
Article9. Connors JM, Klimo P, Fairey RN, Voss N. Brief chemotherapy and involved field radiation therapy for limited-stage, histologically aggressive lymphoma. Ann Intern Med. 1987; 107:25–30. PMID: 3296898.
Article10. Longo DL, Glatstein E, Duffey PL, et al. Treatment of localized aggressive lymphomas with combination chemotherapy followed by involved-field radiation therapy. J Clin Oncol. 1989; 7:1295–1302. PMID: 2788716.
Article11. Nissen NI, Ersbøll J, Hansen HS, et al. A randomized study of radiotherapy versus radiotherapy plus chemotherapy in stage non-Hodgkin's lymphomas. Cancer. 1983; 52:1–7. PMID: 6406037.12. Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002; 346:235–242. PMID: 11807147.
Article13. Pfreundschuh M, Trümper L, Osterborg A, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006; 7:379–391. PMID: 16648042.14. Persky DO, Unger JM, Spier CM, et al. Phase II study of rituximab plus three cycles of CHOP and involved-field radiotherapy for patients with limited-stage aggressive B-cell lymphoma: Southwest Oncology Group study 0014. J Clin Oncol. 2008; 26:2258–2263. PMID: 18413640.
Article15. NCCN Clinical Practice Guidelines in Oncology: Non-Hodgkin's Lymphoma V.I.2010. 2010. Accessed November 8, 2010. Washington, DC: National Comprehensive Cancer Network;(at http://www.nccn.org/professionals/physician_gls/PDF/nhl.pdf).16. Cheson BD, Horning SJ, Coiffier B, et al. NCI Sponsored International Working Group. Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. J Clin Oncol. 1999; 17:1244. PMID: 10561185.
Article17. Bonnet C, Fillet G, Mounier N, et al. CHOP alone compared with CHOP plus radiotherapy for localized aggressive lymphoma in elderly patients: a study by the Groupe dEtude des Lymphomes de l'Adulte. J Clin Oncol. 2007; 25:787–792. PMID: 17228021.
Article18. Yhim HY, Kang HJ, Choi YH, et al. Clinical outcomes and prognostic factors in patients with breast diffuse large B cell lymphoma; Consortium for Improving Survival of Lymphoma (CISL) study. BMC Cancer. 2010; 10:321. PMID: 20569446.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment results of radiotherapy following CHOP or R-CHOP in limited-stage head-and-neck diffuse large B-cell lymphoma: a single institutional experience
- Real‑world data analysis of survival outcomes of patients with primary mediastinal large B‑cell lymphoma treated with immunochemotherapy: the role of consolidative radiation therapy
- Rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) chemotherapy for diffuse large B-cell lymphoma in pregnancy may be associated with preterm birth
- Addition of rituximab to the CHOP regimen has no benefit in patients with primary extranodal diffuse large B-cell lymphoma
- Rituximab Plus CHOP for the Treatment of Primary Mediastinal Large B Cell Lymphoma in a Pregnant Woman