Korean J Hematol.
2011 Jun;46(2):103-110. 10.5045/kjh.2011.46.2.103.
Addition of rituximab to the CHOP regimen has no benefit in patients with primary extranodal diffuse large B-cell lymphoma
- Affiliations
-
- 1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. csuh@amc.seoul.kr
- 2Department of Radiation Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 3Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Hallym University Medical Center, Seoul, Korea.
- KMID: 1782774
- DOI: http://doi.org/10.5045/kjh.2011.46.2.103
Abstract
- BACKGROUND
The addition of rituximab to cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) chemotherapy (R-CHOP) has significantly improved clinical outcomes for patients with diffuse large B-cell lymphoma (DLBCL). However, new predictors of patient response to R-CHOP have not been established. We aimed to evaluate the impact of R-CHOP compared with CHOP in patients with DLBCL and to establish clinical predictors of better outcomes in these patients.
METHODS
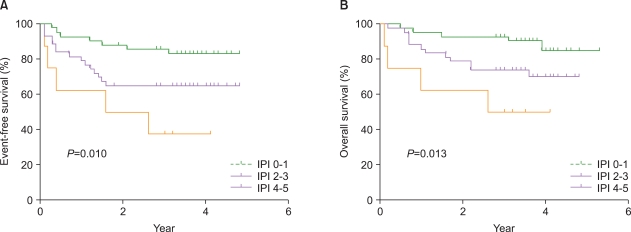
We retrospectively identified 177 patients diagnosed with CD20-positive DLBCL and treated with CHOP (N=82) or R-CHOP (N=95). The response rate, event-free survival (EFS), and overall survival (OS) rates were compared between the 2 treatment groups. All patients were classified into primary extranodal lymphoma (PENL) or nodal lymphoma (NL) subgroups, and the clinical parameters of each subgroup were analyzed.
RESULTS
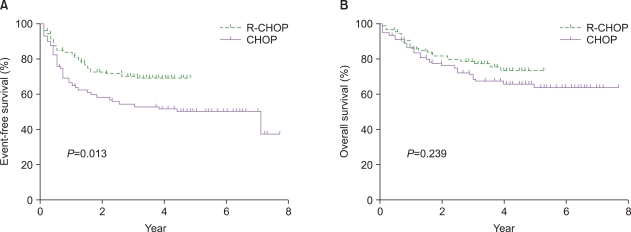
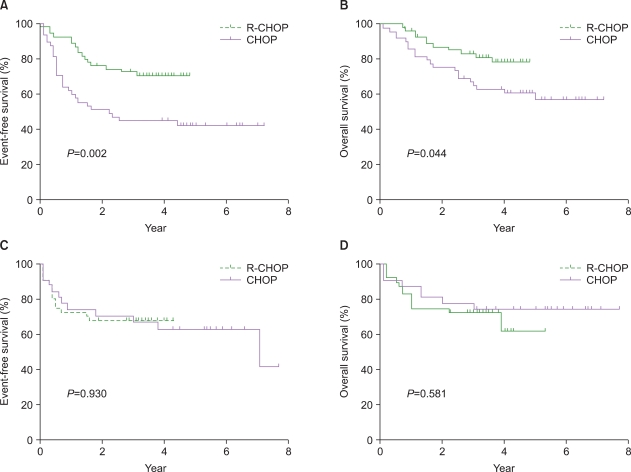
The overall response rate was higher in R-CHOP group (95% vs. 84%, P=0.07). The 3-year EFS rate was significantly higher in R-CHOP group (71% vs. 52%, P=0.013), but the OS rate was comparable between the 2 groups (79% vs. 69%, P=0.23). A significant survival benefit was seen with R-CHOP compared to CHOP therapy in NL patients (P=0.002 for EFS and 0.04 for OS). Multivariate analyses confirmed that R-CHOP therapy is an independent prognostic factor for EFS (hazard ratio of 0.32 [0.17-0.62], P=0.001) and OS (hazard ratio of 0.4 [0.18-0.87], P=0.02) in NL patients.
CONCLUSION
Patients in the PENL group did not benefit from R-CHOP chemotherapy.
MeSH Terms
-
Antibodies, Monoclonal, Murine-Derived
Antineoplastic Combined Chemotherapy Protocols
B-Lymphocytes
Cyclophosphamide
Disease-Free Survival
Doxorubicin
Humans
Lymphoma
Lymphoma, B-Cell
Multivariate Analysis
Prednisolone
Prednisone
Retrospective Studies
Vincristine
Antibodies, Monoclonal, Murine-Derived
Antineoplastic Combined Chemotherapy Protocols
Cyclophosphamide
Doxorubicin
Prednisolone
Prednisone
Vincristine
Figure
Cited by 1 articles
-
A controversial conclusion regarding primary extranodal diffuse large B-cell lymphoma
Seong Kyu Park, Jina Yun, Se Hyung Kim, Dae Sik Hong
Korean J Hematol. 2011;46(3):207-208. doi: 10.5045/kjh.2011.46.3.207.
Reference
-
1. Gatter KC, Warnke RA. Jaffe ES, Harris NL, Stein H, editors. Diffuse large B-cell lymphoma. World Health Organization Classification of Tumors: Pathology and genetics of tumours of haematopoietic and lymphoid tissues. 2001. 3rd ed. Lyon, France: IARC Press;p. 171–174.2. Coiffier B. Diffuse large cell lymphoma. Curr Opin Oncol. 2001; 13:325–334. PMID: 11555708.
Article3. Gordon LI, Harrington D, Andersen J, et al. Comparison of a second-generation combination chemotherapeutic regimen (m-BACOD) with a standard regimen (CHOP) for advanced diffuse non-Hodgkin's lymphoma. N Engl J Med. 1992; 327:1342–1349. PMID: 1383819.
Article4. Fisher RI, Gaynor ER, Dahlberg S, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin's lymphoma. N Engl J Med. 1993; 328:1002–1006. PMID: 7680764.
Article5. Coiffier B, Haioun C, Ketterer N, et al. Rituximab (anti-CD20 monoclonal antibody) for the treatment of patients with relapsing or refractory aggressive lymphoma: a multicenter phase II study. Blood. 1998; 92:1927–1932. PMID: 9731049.6. Vose JM, Link BK, Grossbard ML, et al. Phase II study of rituximab in combination with CHOP chemotherapy in patients with previously untreated, aggressive non-Hodgkin's lymphoma. J Clin Oncol. 2001; 19:389–397. PMID: 11208830.
Article7. Coiffier B, Lepage E, Briere J, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002; 346:235–242. PMID: 11807147.
Article8. Feugier P, VanF Hoof A, Sebban C, et al. Long-term results of the R-CHOP study in the treatment of elderly patients with diffuse large B-cell lymphoma: a study by the Groupe dEtudes des Lymphomes de lAdulte. J Clin Oncol. 2005; 23:4117–4126. PMID: 15867204.9. Sehn LH, Donaldson J, Chhanabhai M, et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol. 2005; 23:5027–5033. PMID: 15955905.
Article10. Pfreundschuh M, Trümper L, Osterborg A, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006; 7:379–391. PMID: 16648042.11. A predictive model for aggressive non-Hodgkin's lymphoma. The Iinternational Non-Hodgkin's Llymphoma prognostic factors project. N Engl J Med. 1993; 329:987–994. PMID: 8141877.12. Sehn LH, Berry B, Chhanabhai M, et al. The revised International Prognostic Index (R-IPI) is a better predictor ofr outcome than the standard IPI for patients with diffuse large B-cell lymphoma DLBCL treated with R-CHOP. Blood. 2007; 109:1857–1861. PMID: 17105812.13. Mounier N, Briere J, Gisselbrecht C, et al. Rituximab plus CHOP (R-CHOP) overcomes bcl-2-associated resistance to chemotherapy in elderly patients with diffuse large B-cell lymphoma (DLBCL). Blood. 2003; 101:4279–4284. PMID: 12576316.
Article14. Winter JN, Weller EA, Horning SJ, et al. Prognostic significance of Bcl-6 protein expression in DLBCL treated with CHOP or R-CHOP: a prospective correlative study. Blood. 2006; 107:4207–4213. PMID: 16449523.
Article15. Zucca E, Roggero E, Bertoni F, Cavalli F. Primary extranodal non-Hodgkin's lymphomas. Part 1: Gastrointestinal, cutaneous and genitourinary lymphomas. Ann Oncol. 1997; 8:727–737. PMID: 9332679.
Article16. Newton R, Ferlay J, Beral V, et al. The epidemiology of non-Hodgkin's lymphoma: cComparison of nodal and extra- nodal sites. Int J Cancer. 1997; 72:923–930. PMID: 9378552.17. Otter R, Gerrits WB, vd Sandt MM, Hermans J, Willemze R. Primary extranodal and nodal non-Hodgkin's lymphoma. A survey of a population-based registry. Eur J Cancer Clin Oncol. 1989; 25:1203–1210. PMID: 2767109.18. Shih LY, Liang DC. Non-Hodgkin's lymphomas in Asia. Hematol Oncol Clin North Am. 1991; 5:983–1001. PMID: 1938764.
Article19. Aozasa K, Tsujimoto M, Sakurai M, et al. Non-Hodgkin's lymphomas in Osaka, Japan. Eur J Cancer Clin Oncol. 1985; 21:487–492. PMID: 4007017.
Article20. Kim CW, Kim I, Ko YH, et al. Clinicopathologic and immunophenotypic study of non-Hodgkin's lymphoma in Korea. Lymphoreticular Study Group of the Korean Society of Pathologists. J Korean Med Sci. 1992; 7:193–198. PMID: 1285918.21. Kramer MH, Hermans J, Parker J, et al. Clinical significance of bcl2 and p53 protein expression in diffuse large B-cell lymphoma: a population-based study. J Clin Oncol. 1996; 14:2131–2138. PMID: 8683246.
Article22. Houldsworth J, Mathew S, Rao PH, et al. REL proto-oncogene is frequently amplified in extranodal diffuse large cell lymphoma. Blood. 1996; 87:25–29. PMID: 8547649.
Article23. Grønbaek K, Straten PT, Ralfkiaer E, et al. Somatic Fas mutations in non-Hodgkin's lymphoma: association with extranodal disease and autoimmunity. Blood. 1998; 92:3018–3024. PMID: 9787134.24. Rao PH, Houldsworth J, Dyomina K, et al. Chromosomal and gene amplification in diffuse large B-cell lymphoma. Blood. 1998; 92:234–240. PMID: 9639522.
Article25. Cheson BD, Pfistner B, Juweid ME, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007; 25:579–586. PMID: 17242396.26. Park YH, Lee JJ, Ryue MH, et al. Improved therapeutic outcomes of DLBCL after introduction of rituximab in Korean patients. Ann Hematol. 2006; 85:257–262. PMID: 16416337.
Article27. Kramer MH, Hermans J, Parker J, et al. Clinical significance of bcl2 and p53 protein expression in diffuse large B-cell lymphoma: a population-based study. J Clin Oncol. 1996; 14:2131–2138. PMID: 8683246.
Article28. Lopez-Guillermo A, Colomo L, Jimenez M, et al. Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin. J Clin Oncol. 2005; 23:2797–2804. PMID: 15728226.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Primary Adrenal Diffuse Large B-cell Lymphoma Achieving Complete Remission with Rituximab-CHOP Chemotherapy
- Primary Diffuse Large B Cell Lymphoma of the Bladder
- Rituximab Plus CHOP for the Treatment of Primary Mediastinal Large B Cell Lymphoma in a Pregnant Woman
- Clinicopathologic significance of tumor microenvironment CD11c, and FOXP3 expression in diffuse large B-cell lymphoma patients receiving rituximab, cyclophosphamide, anthracycline, vincristine, and prednisone (R-CHOP) combination chemotherapy
- A Case of Mantle Cell Lymphoma Treated with Autologous Stem Cell Transplantation and Rituximab