Korean Circ J.
2011 Nov;41(11):641-648. 10.4070/kcj.2011.41.11.641.
The Effects of Statin and Niacin on Plaque Stability, Plaque Regression, Inflammation and Oxidative Stress in Patients With Mild to Moderate Coronary Artery Stenosis
- Affiliations
-
- 1Department of Cardiology, Gil Hospital, Gachon University of Medicine and Science, Incheon, Korea. encore@gilhospital.com
- KMID: 2225071
- DOI: http://doi.org/10.4070/kcj.2011.41.11.641
Abstract
- BACKGROUND AND OBJECTIVES
The aim of this study was to compare the effects of a combination of niacin and simvastatin to simvastatin alone, on plaque regression and inflammatory makers.
SUBJECTS AND METHODS
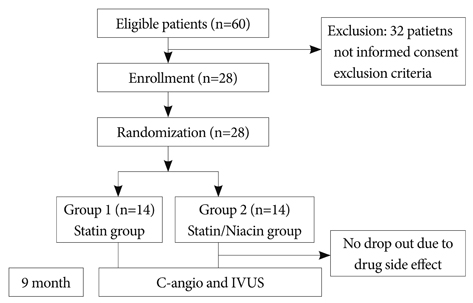
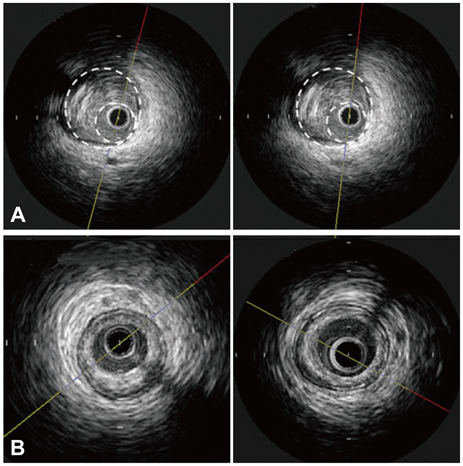
The study had a prospective, randomized design. Subjects were patients with intermediate coronary artery stenosis. A total of 28 patients received a combination of niacin 1,000 mg plus simvastatin 40 mg (N+S group, n=14); the other group received simvastatin 40 mg alone (S group, n=14). All patients had a baseline and a 9-month follow-up coronary angiogram and an intravascular ultrasound procedure. Parameters such as normalized total atheroma volume (nTAV) and percent atheroma volume (PAV) were analyzed before and after treatment as were inflammatory markers such as high sensitivity C-reactive protein (hs-CRP), Matrix me-talloproteinase-9 (MMP-9) and soluble CD40 ligand (sCD40L).
RESULTS
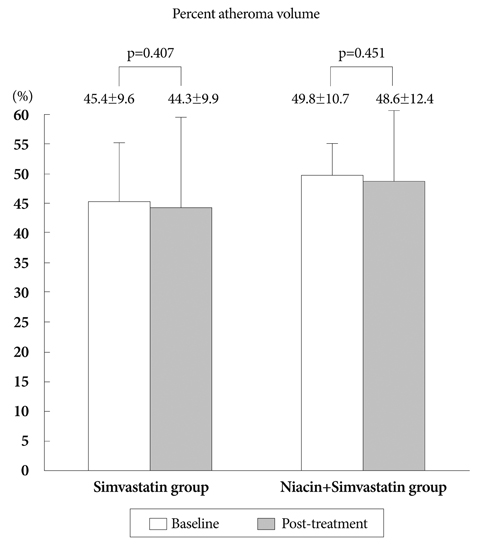
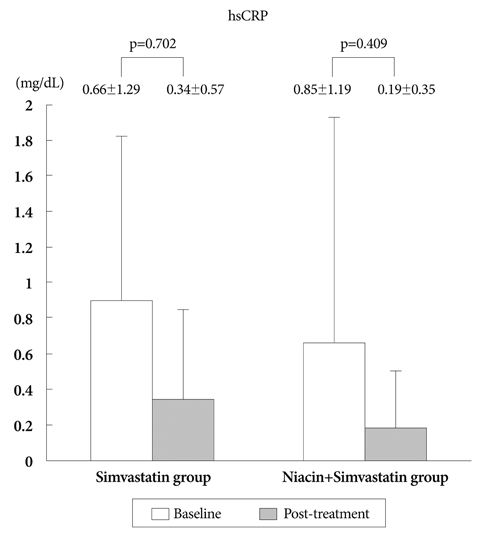
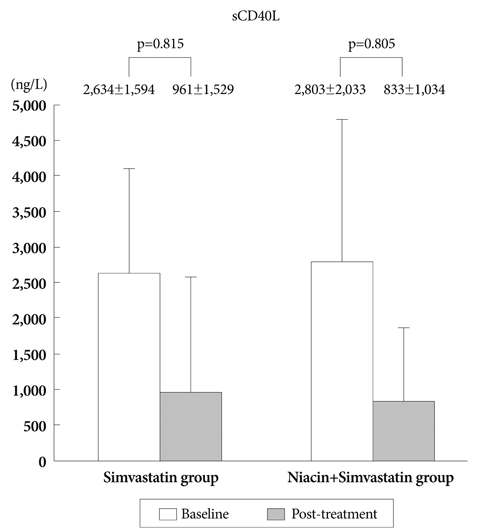
There was no difference in baseline characteristics between the two groups. The nTAV and PAV in the N+S group before and after treatment were not different than those in the S group. But the degree of changes (delta) in nTAV in the N+S group was greater than that in the S group (-21.6+/-10.68 vs. 5.25+/-42.19, respectively, p=0.024). Also, the change in PAV in the NS group was higher than that in the S group (-1.2+/-2.5 vs. -0.6+/-5, respectively, p=0.047. Changes in hs-CRP, MMP-9, and sCD40L in the NS group were significantly greater than those of the S group (-0.71+/-1.25, 73.5+/-64.9, -1,970+/-1,925 vs. -0.32+/-0.96, 62.5+/-30.6, -1,673+/-2,628, respectively).
CONCLUSION
The combination of niacin plus simvastatin decreases coronary plaque volume and attenuates the inflammatory response in patients with intermediate coronary artery stenosis.
MeSH Terms
-
C-Reactive Protein
CD40 Ligand
Coronary Stenosis
Coronary Vessels
Follow-Up Studies
Humans
Hydroxymethylglutaryl-CoA Reductase Inhibitors
Inflammation
Niacin
Oxidative Stress
Plaque, Atherosclerotic
Prospective Studies
Simvastatin
Ultrasonography, Interventional
C-Reactive Protein
CD40 Ligand
Hydroxymethylglutaryl-CoA Reductase Inhibitors
Niacin
Simvastatin
Figure
Reference
-
1. The Scandinavian Simvastatin Survival Study (4S). Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet. 1994. 344:1383–1389.2. Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N Engl J Med. 1995. 333:1301–1307.3. Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial Investigators. N Engl J Med. 1996. 335:1001–1009.4. The Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group. Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease and a broad range of initial cholesterol levels. The Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group. N Engl J Med. 1998. 339:1349–1357.5. Gordon DJ, Probstfield JL, Garrison RJ, et al. High-density lipoprotein cholesterol and cardiovascular disease. Four Prospective American Studies. Circulation. 1989. 79:8–15.6. Kannel WB. Range of serum cholesterol values in the population developing coronary artery disease. Am J Cardiol. 1995. 76:69C–77C.7. The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002. 106:3143–3421.8. Meyers CD, Kamanna VS, Kashyap ML. Niacin therapy in atherosclerosis. Curr Opin Lipidol. 2004. 15:659–665.9. Carlson LA. Nicotinic acid: the broad-spectrum lipid drug: a 50th anniversary review. J Intern Med. 2005. 258:94–114.10. Sakai T, Kamanna VS, Kashyap ML. Niacin, but not gemfibrozil, selectively increases LP-AI, a cardioprotective subfraction of HDL, in patients with low HDL cholesterol. Arterioscler Thromb Vasc Biol. 2001. 21:1783–1789.11. Taylor AJ, Zhu D, Sullenberger LE, Lee HJ, Lee JK, Grace KA. Relationship between glycemic status and progression of carotid intima-media thickness during treatment with combined statin and extended-release niacin in ARBITER 2. Vasc Health Risk Manag. 2007. 3:159–164.12. Taylor AJ, Lee HJ, Sullenberger LE. The effect of 24 months of combination statin and extended-release niacin on carotid intima-media thickness: ARBITER 3. Curr Med Res Opin. 2006. 22:2243–2250.13. Mintz GS, Nissen SE, Anderson WD, et al. A report of the American College of Cardiology Task Force on clinical expert consensus documents. American College of Cardiology Clinical Expert Consensus Document on Standards for Acquisition, Measurement and Reporting of Intravascular Ultrasound Studies (IVUS). J Am Coll Cardiol. 2001. 37:1478–1492.14. Park SY, Kwak JJ, Park SH. Dose dependent changes of lipid profiles, IL-6 and CRP in unstable angina patients after simvastatin therapy. Korean Circ J. 2003. 33:663–670.15. Son JW, Koh KK. Effects of statins on endothelium: vasomotor function, inflammation, and hemostasis. Korean Circ J. 1999. 29:1016–1031.16. Hong YJ, Jeong MH, Ahn YK, et al. Effect of conventional dose of simvastatin on plaque regression and vascular remodeling in the peristent reference segments of normocholesterolemic patients: a serial intravascular ultrasound assessment. Korean Circ J. 2007. 37:483–488.17. Hong YJ, Jeong MH, Lim JH, et al. The prognostic significance of statin therapy according to the level of C-reactive protein in acute myocardial infarction patients who underwent percutaneous coronary intervention. Korean Circ J. 2003. 33:891–900.18. Nissen SE, Nicholls SJ, Sipahi I, et al. Effect of very high-intensity statin therapy on regression of coronary atherosclerosis: the ASTEROID trial. JAMA. 2006. 295:1556–1565.19. Brown BG, Zhao XQ, Chait A, et al. Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med. 2001. 345:1583–1592.20. Taylor AJ, Sullenberger LE, Lee HJ, Lee JK, Grace KA. Arterial biology for the investigation of the treatment effects of reducing cholesterol (ARBITER) 2: a double-blind, placebo-controlled study of extended-release niacin on atherosclerosis progression in secondary prevention patients treated with statins. Circulation. 2004. 110:3512–3517.21. Cipollone F, Mezzetti A, Porreca E, et al. Association between enhanced soluble CD40L and prothrombotic state in hypercholesterolemia: effects of statin therapy. Circulation. 2002. 106:399–402.22. Tomai F, Crea F, Gaspardone A, et al. Unstable angina and elevated c-reactive protein levels predict enhanced vasoreactivity of the culprit lesion. Circulation. 2001. 104:1471–1476.23. Fichtlscherer S, Rosenberger G, Walter DH, Breuer S, Dimmeler S, Zeiher AM. Elevated C-reactive protein levels and impaired endothelial vasoreactivity in patients with coronary artery disease. Circulation. 2000. 102:1000–1006.24. Son JW, Koh KK, Ahn JY, et al. Effects of statin on plaque stability and thrombogenicity in hypercholesterolemic patients with coronary artery disease. Int J Cardiol. 2003. 88:77–82.25. Fernandez-Patron C, Martinez-Cuesta MA, Salas E, et al. Differential regulation of platelet aggregation by matrix metalloproteinases-9 and -2. Thromb Haemost. 1999. 82:1730–1735.26. Lelongt B, Bengatta S, Delauche M, Lund LR, Werb Z, Ronco PM. Matrix metalloproteinase 9 protects mice from anti-glomerular basement membrane nephritis through its fibrinolytic activity. J Exp Med. 2001. 193:793–802.27. Fontaine V, Jacob MP, Houard X, et al. Involvement of the mural thrombus as a site of protease release and activation in human aortic aneurysms. Am J Pathol. 2002. 161:1701–1710.28. Tang J, Liu J, Zhou C, et al. Mmp-9 deficiency enhances collagenase-induced intracerebral hemorrhage and brain injury in mutant mice. J Cereb Blood Flow Metab. 2004. 24:1133–1145.29. Johnson JL, George SJ, Newby AC, Jackson CL. Divergent effects of matrix metalloproteinases 3, 7, 9, and 12 on atherosclerotic plaque stability in mouse brachiocephalic arteries. Proc Natl Acad Sci U S A. 2005. 102:15575–15580.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Statin Therapy with Coronary Plaque Imaging
- Plaque Characteristics on Coronary CT Angiography: Effects on the Degree of Coronary Artery Stenosis
- Early Differential Changes in Coronary Plaque Composition According to Plaque Stability Following Statin Initiation in Acute Coronary Syndrome: Classification and Analysis by Intravascular Ultrasound-Virtual Histology
- Noninvasive Imaging of Atherosclerotic Plaques Using MRI and CT
- Assessment of Coronary Stenosis Using Coronary CT Angiography in Patients with High Calcium Scores: Current Limitations and Future Perspectives