J Rheum Dis.
2015 Jun;22(3):140-145. 10.4078/jrd.2015.22.3.140.
Hydroxychloroquine Retinopathy
- Affiliations
-
- 1Department of Ophthalmology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. ophthalmo@amc.seoul.kr
- KMID: 2222879
- DOI: http://doi.org/10.4078/jrd.2015.22.3.140
Abstract
- Hydroxychloroquine (HCQ) has been widely used for treatment of various rheumatic and dermatologic diseases. However, under a high cumulative dose, HCQ may cause retinal toxicity. In this review, we summarize the underlying mechanisms, prevalence, risk factors, clinical characteristics, screening tests, treatments, and prognosis of HCQ retinopathy. Because HCQ retinopathy is rarely reversible, screening tests to determine the retinal toxicity prior to its development are important. The American Academy of Ophthalmology screening protocols for early detection were updated in 2013. However, as a different clinical type is found in Asian patients, predominantly the pericentral type of photoreceptor damage, rather than the traditional parafoveal type, we propose a modified screening protocol for detection of HCQ retinopathy in Korean and other Asian patients.
Keyword
MeSH Terms
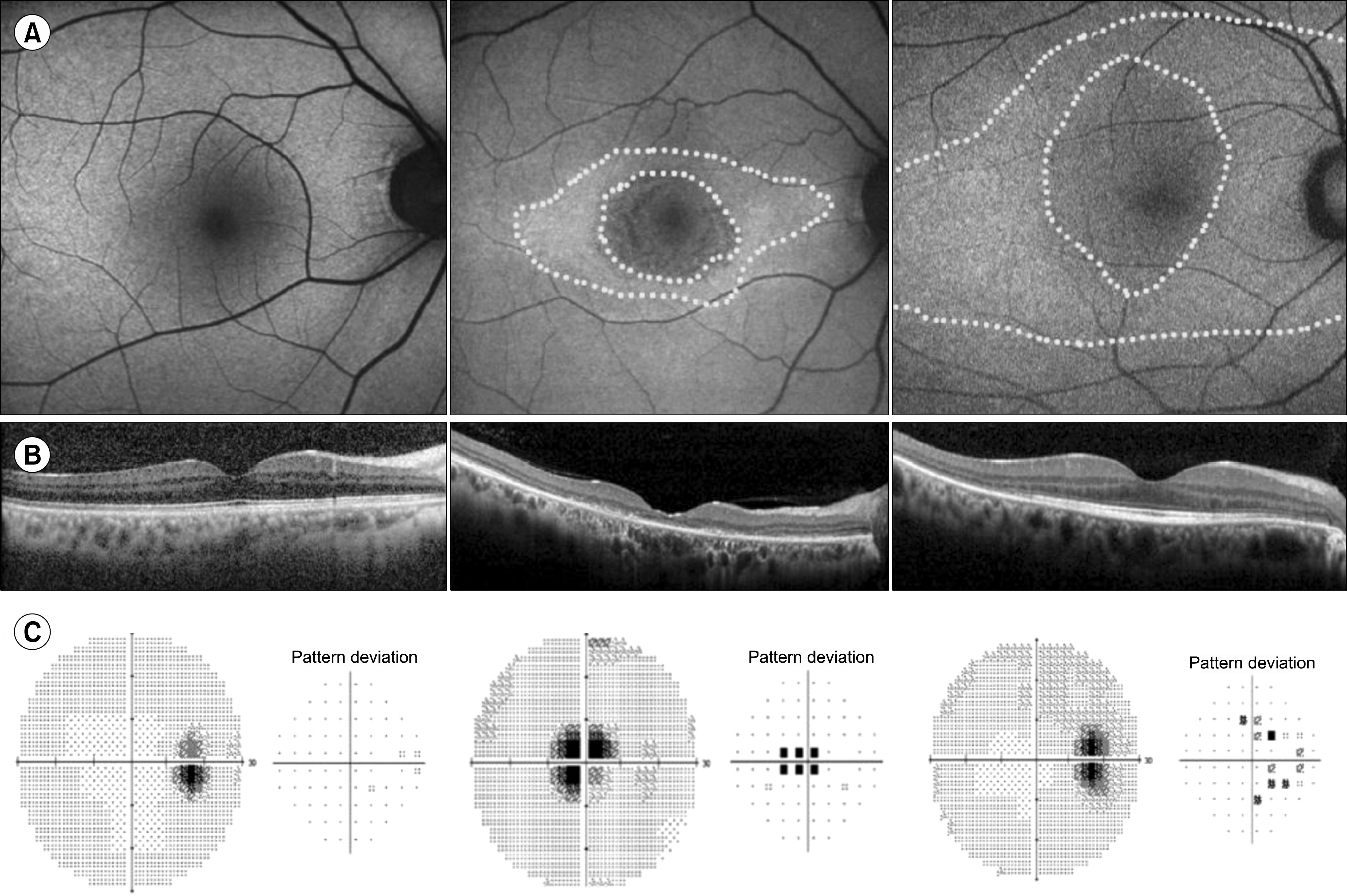
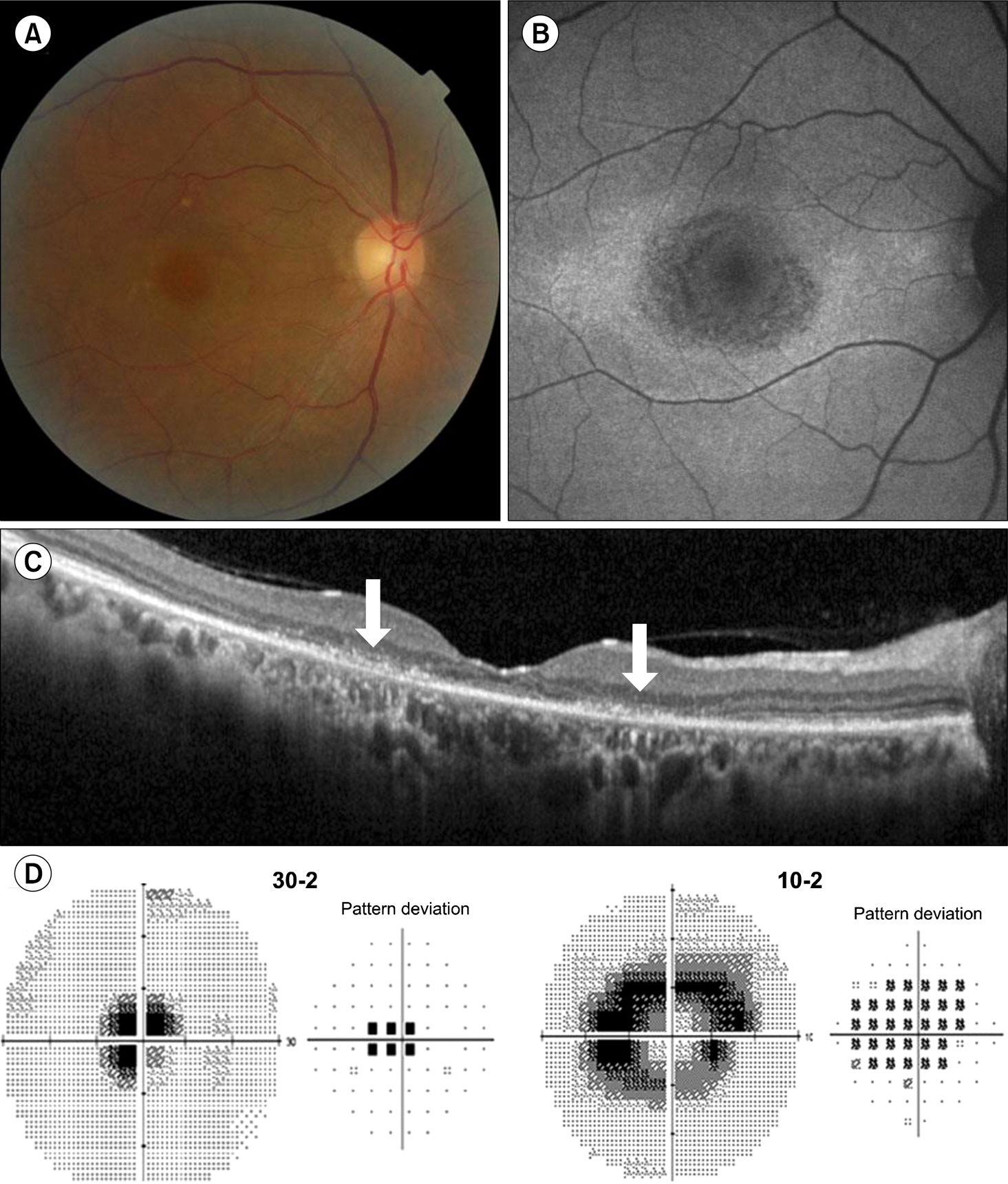
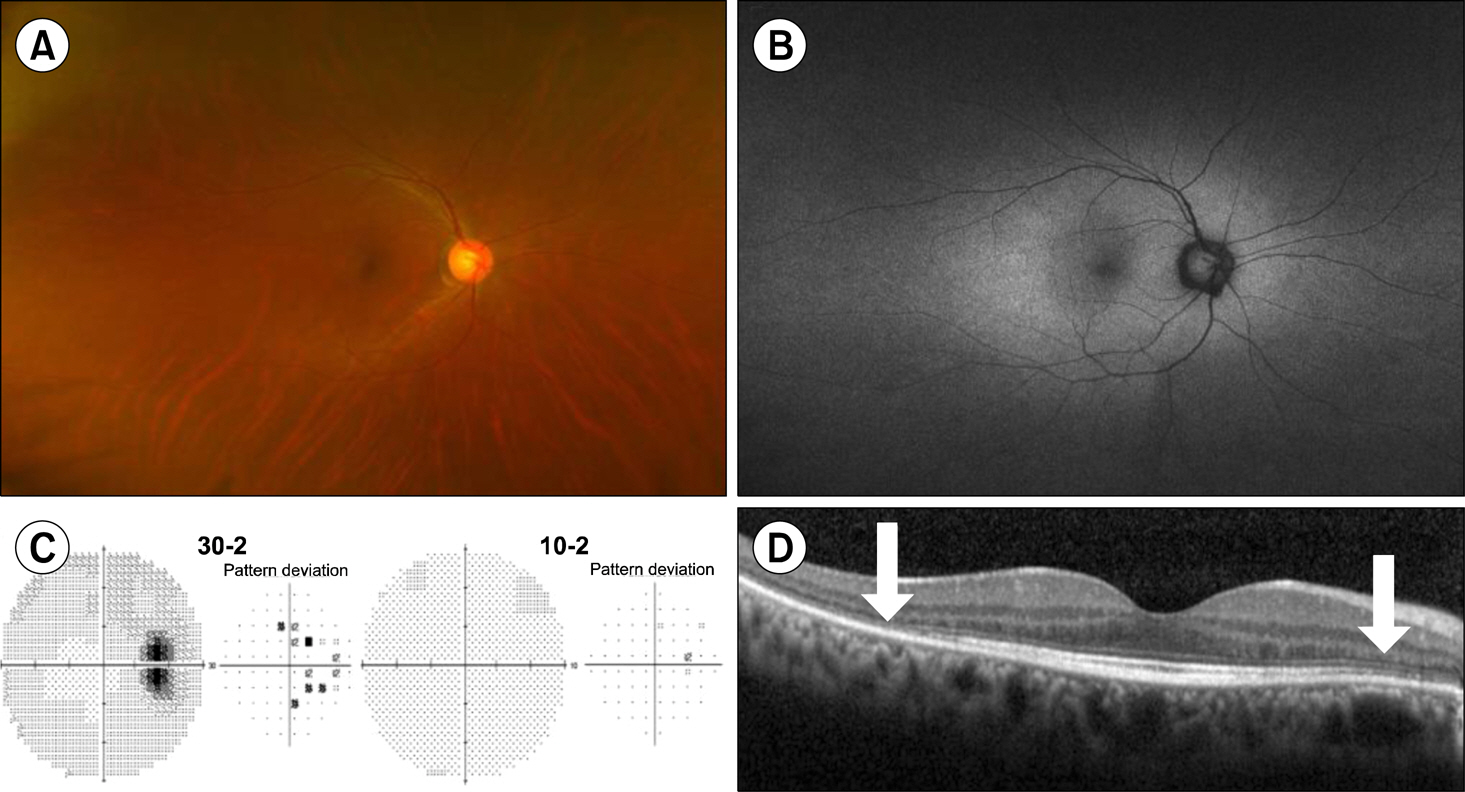
Figure
Reference
-
1. Tehrani R, Ostrowski RA, Hariman R, Jay WM. Ocular toxicity of hydroxychloroquine. Semin Ophthalmol. 2008; 23:201–9.
Article2. Fox RI. Mechanism of action of hydroxychloroquine as an antirheumatic drug. Semin Arthritis Rheum. 1993; 23(2 Suppl 1):82–91.
Article3. Shearer RV, Dubois EL. Ocular changes induced by long-term hydroxychloroquine (plaquenil) therapy. Am J Ophthalmol. 1967; 64:245–52.
Article4. Levy GD, Munz SJ, Paschal J, Cohen HB, Pince KJ, Peterson T. Incidence of hydroxychloroquine retinopathy in 1,207 patients in a large multicenter outpatient practice. Arthritis Rheum. 1997; 40:1482–6.
Article5. Mäntyjärvi M. Hydroxychloroquine treatment and the eye. Scand J Rheumatol. 1985; 14:171–4.
Article6. Lee DH, Melles RB, Joe SG, Lee JY, Kim JG, Lee CK, et al. Pericentral hydroxychloroquine retinopathy in Korean patients. Ophthalmology. 2015; 122:1252–6.
Article7. Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology. 2015; 122:110–6.
Article8. Yam JC, Kwok AK. Ocular toxicity of hydroxychloroquine. Hong Kong Med J. 2006; 12:294–304.9. Marmor MF, Carr RE, Easterbrook M, Farjo AA, Mieler WF. American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy: a report by the American Academy of Ophthalmology. Ophthalmology. 2002; 109:1377–82.10. Michaelides M, Stover NB, Francis PJ, Weleber RG. Retinal toxicity associated with hydroxychloroquine and chloroquine: risk factors, screening, and progression despite cessation of therapy. Arch Ophthalmol. 2011; 129:30–9.11. Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 2014; 132:1453–60.
Article12. Johnson MW, Vine AK. Hydroxychloroquine therapy in massive total doses without retinal toxicity. Am J Ophthalmol. 1987; 104:139–44.
Article13. Mavrikakis M, Papazoglou S, Sfikakis PP, Vaiopoulos G, Rougas K. Retinal toxicity in long term hydroxychloroquine treatment. Ann Rheum Dis. 1996; 55:187–9.
Article14. Falcone PM, Paolini L, Lou PL. Hydroxychloroquine toxicity despite normal dose therapy. Ann Ophthalmol. 1993; 25:385–8.15. Shroyer NF, Lewis RA, Lupski JR. Analysis of the ABCR (ABCA4) gene in 4-aminoquinoline retinopathy: is retinal toxicity by chloroquine and hydroxychloroquine related to Stargardt disease? Am J Ophthalmol. 2001; 131:761–6.
Article16. Marmor MF. Efficient and effective screening for hydroxychloroquine toxicity. Am J Ophthalmol. 2013; 155:413–4.
Article17. Wei LC, Chen SN, Ho CL, Kuo YH, Ho JD. Progression of hydroxychloroquine retinopathy after discontinuation of therapy: case report. Chang Gung Med J. 2001; 24:329–34.18. Brinkley JR Jr, Dubois EL, Ryan SJ. Long-term course of chloroquine retinopathy after cessation of medication. Am J Ophthalmol. 1979; 88:1–11.
Article19. Easterbrook M. The ocular safety of hydroxychloroquine. Semin Arthritis Rheum. 1993; 23(2 Suppl 1):62–7.
Article20. Bienfang D, Coblyn JS, Liang MH, Corzillius M. Hydroxychloroquine retinopathy despite regular ophthalmologic evaluation: a consecutive series. J Rheumatol. 2000; 27:2703–6.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prevalence and Risk Factors of Hydroxychloroquine Retinopathy in Rheumatic Patients with Dry Eye Symptoms
- Frequency and Clinical Characteristics of Hydroxychloroquine Retinopathy in Korean Patients with Rheumatologic Diseases
- Hydroxychloroquine Retinopathy Update
- A Case of Hydroxychloroquine Retinopathy
- A Case of Recurrent Annular Erythema of Sjogren's Syndrome Treated with Hydroxychloroquine