J Korean Radiol Soc.
1998 Mar;38(3):385-390. 10.3348/jkrs.1998.38.3.385.
1H MR Spectroscopy of the Normal Human Brains: Comparison of Automated Prescan Method with Manual Method
- Affiliations
-
- 1Department of Radiology, Inha University Hospital College of Medicine.
- 2Department of Radiological Sciences, Asan Institute for Life Sciences.
- 3Department of Diagnostic Radiology, Asan Medical Center, University of Ulsan College of Medicine.
- KMID: 2201390
- DOI: http://doi.org/10.3348/jkrs.1998.38.3.385
Abstract
-
PURPOSE: To evaluate regional differences in relative metabolite ratios in the normal human brain by 1H MRspectroscopy (MRS), and compare the spectral quality obtained by the automated prescan method (PROBE) and themanual method.
MATERIALS AND METHODS
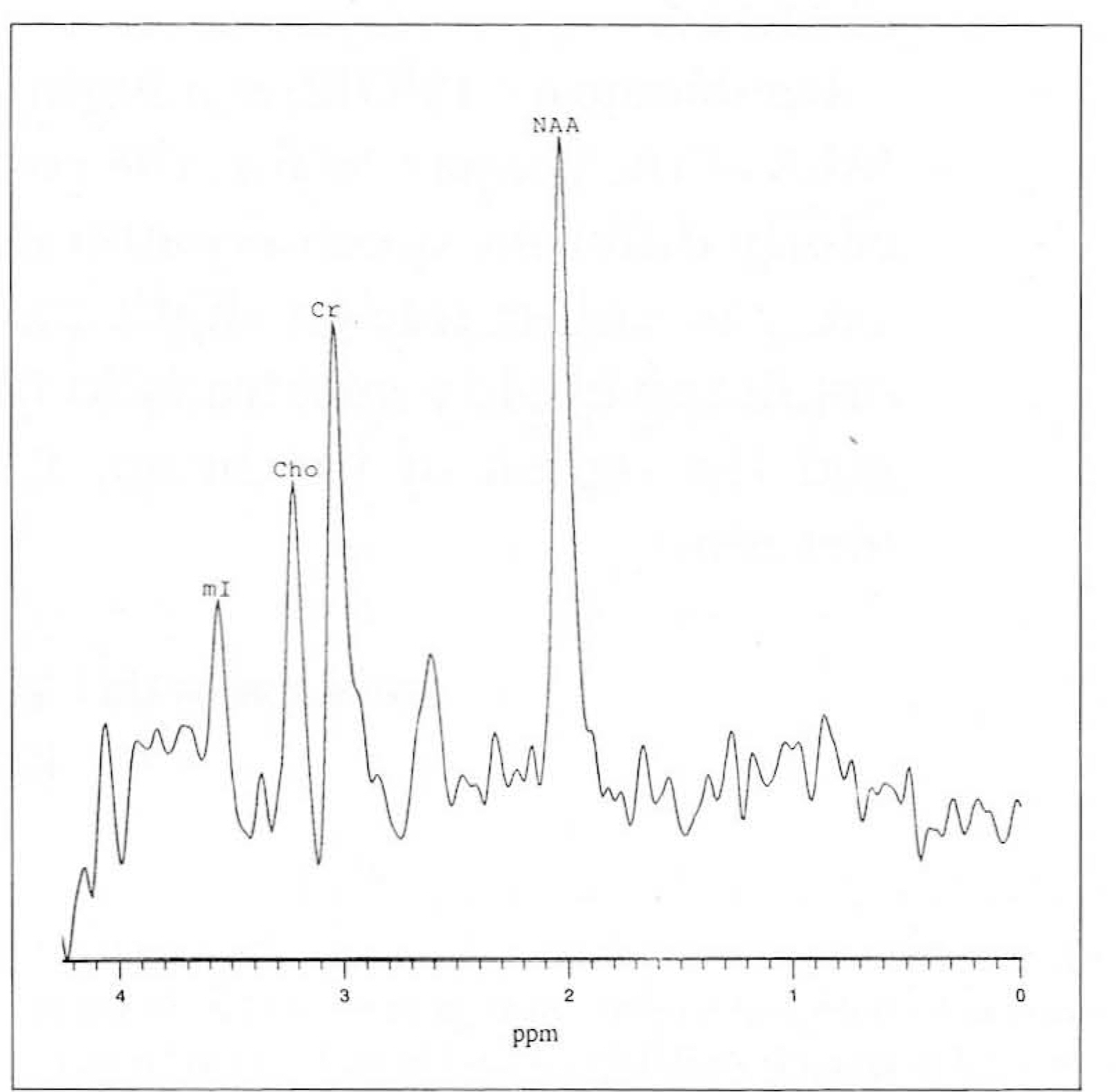
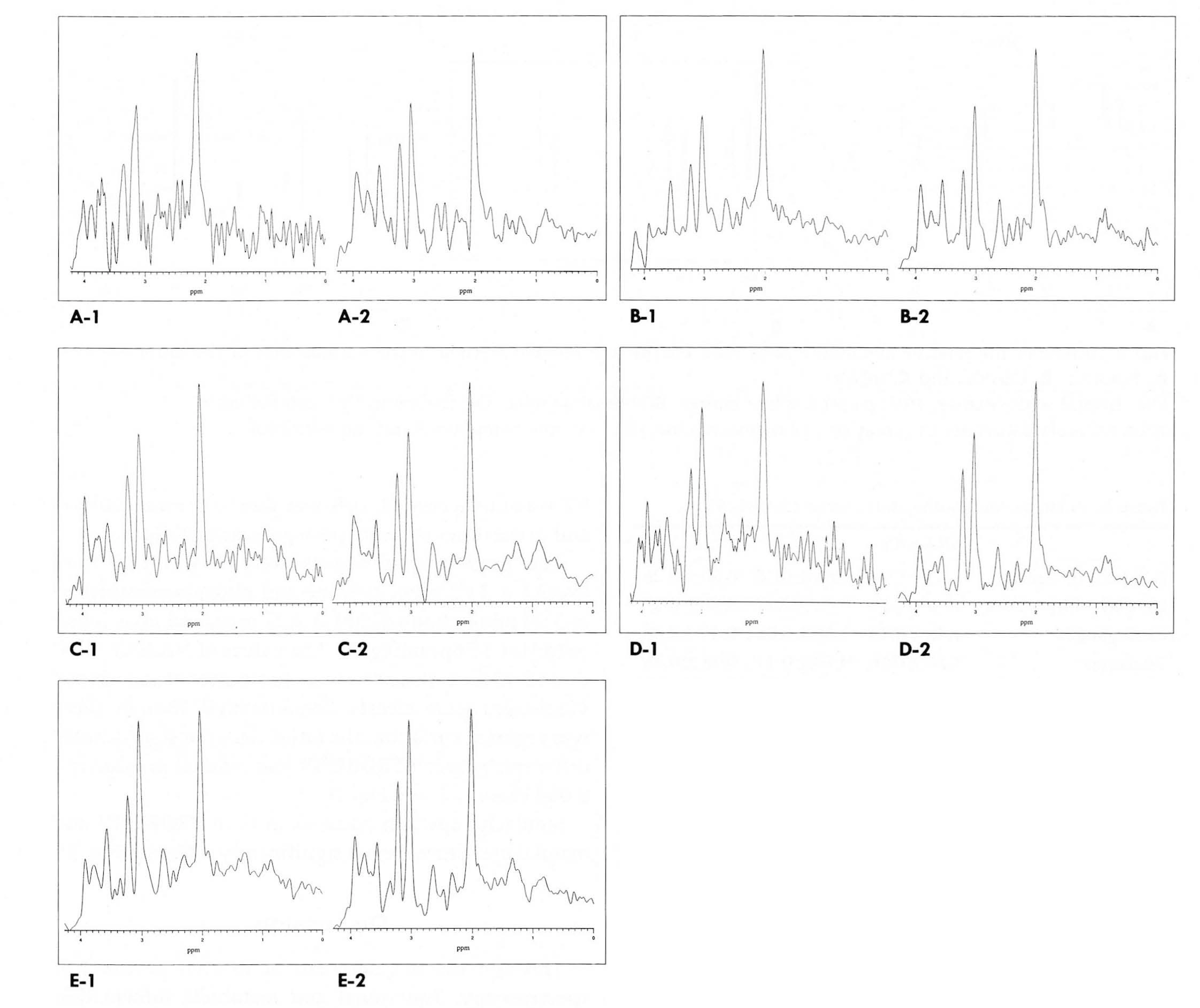
Localized 1H MRS was performed on a GE 1.5T SIGNA MRI/MRS system (version5.5) with active shielded gradients. For 20 normal volunteers aged 8-47 years, spectral parameters were adjustedby the auto-prescan routine provided by a PROBE package(N=34)and manually (N=33). Five regions of the human brainwere examined (N=PROBE,manual): frontal white matter(N=6,10), parietal white matter(N=8,9), basal ganglia(N=6,5),thalamus(N=4,5), and cerebellum(N=4,4). For all spectra, a STEAM localization sequence with three-pulse CHESS H2Osuppression was used, with the following acquisition parameters: TR=3.0 sec, TE=30 msec, TM=13,7 msec, SW=2500Hz,SI=2048 pts, AVG=48, and NEX=2.
RESULTS
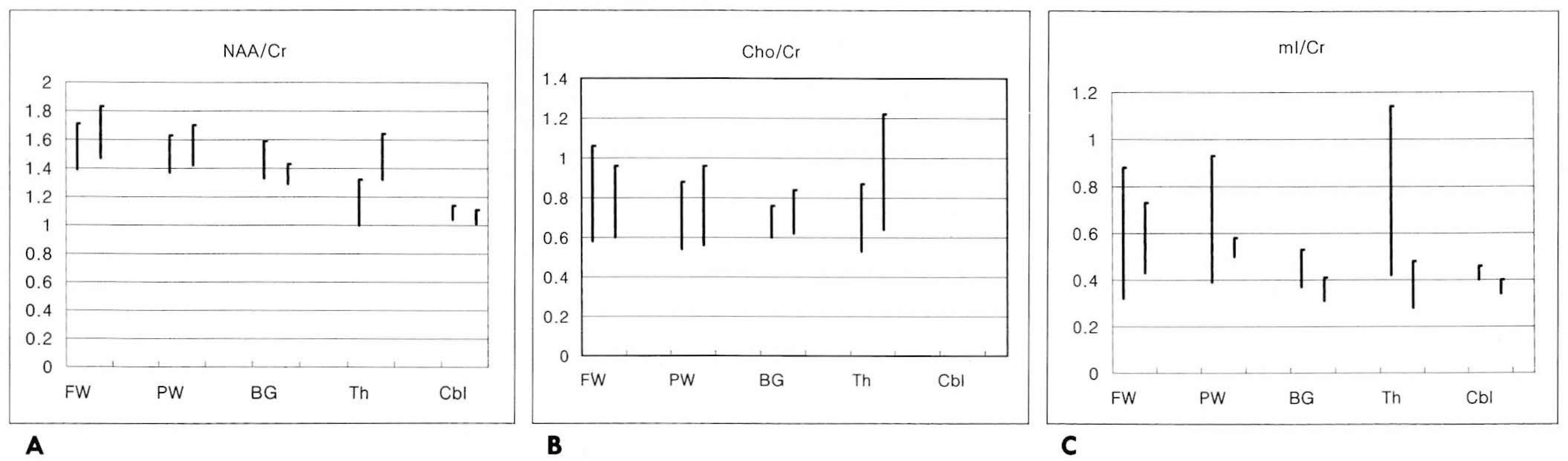
A total of 61 reliable spectra were obtained by PROBE (28/34=82%success) and by the manual method (33/33=100% success). Regional differences in the spectral patterns of the fiveregions were clearly demonstrated by both PROBE and the manual methods. For prescanning, the manual method tookslightly longer than PROBE (3-5 mins and 2 mins,respectively). There were no significant differences in spectralpatterns and relative metabolic ratios between the two methods. However, auto-prescan by PROBE seemed to be veryvulnerable to slight movement by patients, and in three cases, an acceptable spectrum was thus not obtained.
CONCLUSION
PROBE is a highly practical and reliable method for single voxel 1H MRS of the human brain; the twomethods of prescanning do not result in significantly different spectral patterns and the relative metaboliteratios. PROBE, however, is vulnerable to slight movement by patients, and if the success rate for obtainingquality spectra is to be increased, regardless of the patient's condition and the region of the brain, it must beused in conjunction with the manual method.
Figure
Reference
-
1.Wang Ζ., Zimmerman RA., Sauter R. Proton MR Spectroscopy of the brain: Clinically Usefull information obtained in assessing CNS diseases in children. AJR. 1996. 167:191–199.2.Tien RD., Lau PH., Smith JS., Lazeyras F. Singlevox el proton brain spectroscopy exam (PROBE/SV) in patients with primary brain tumors. AJR. 1996. 167:201–209.3.Miller BL., Moats RA., Shonk T., Ernest T., Woolley S., Ross BR. Alzheimer Disease: Depiction of increased cerebral myo-Inositol with proton MR spectroscopy. Radiology. 1993. 187:433–437.
Article4.Kuhl CK., Layer G., Traber F., Zierz S., Block W., Reiser M. Mitochondrial encephalomyopathy: Correlation of P-31 exercise MR spectroscopy with clinical findings. Radiology. 1994. 192:223–230.
Article5.Song IC., Chang KH., Han MH, et al. In vivo single voxel Ή spectroscopy in cerebral glioma. J Korean Radiol Soc. 1996. 35:307–314.6.Lanfermann H., Kugel H., Heindel W., Herholz K., Heiss W-D., Lackner K. Metabolic changes in acute and subacute cerebral infarctions: Findings at proton MR spectroscopic imaging. Radiology. 1995. 196:203–210.
Article7.Ross RD., Jacobson S., Villamil F, et al. Subclinical hepatic encephalopathy: proton MR spectroscopic abnormaities. Radiology. 1994. 193:457–463.8.Rajanayagam V., Grad J., Krivit W, et al. Proton MR spectroscopy of childhood adrenoleukodystrophy. AJNR. 1996. 17:1013–1024.9.Chang KH., Jeon BS., Song IC, et al. Ή MR spectroscopy in Parkinson's disease and progressive supranuclear palsy: Preliminary stydy. J Korean Radiol Soc. 1996. 34:711–716.10.Chong WK., Sweeney B., Wilkinson ID, et al. Proton spectroscopy of the brain in HIV infection: Correlation with clinical, immunologic, and MR imaging findings, Radiology. 1993. 188:119–124.11.Shonk TK., Mosts RA., Gifford P, et al. Probable Alzheimer disease: Diagnosis with proton MR spectroscopy. Radiology. 1995. 195:65–72.
Article12.Tzika A A., Ball WS Jr., Vigneron DB., Dunn RS., Nelson SJ. Kirks D. Childhood adrenoleukodystropy: Assessment with proton MR spectroscopy. Radology. 1993. 189:467–480.13.Ott D., Hennig J., Ernst T. Human brain tumors: Assessment with in vivo proton MR spectroscopy. Radiology. 1993. 186:745–752.
Article14.Bizzi A., Movsas B., Tedeschi G, et al. Response of Non-Hodgkin lymphoma to radiation therapy: Early and long-term assessment with Ή MR spectroscopic imaging. Radiology. 1995. 194:271–276.15.Knaap MS., Grond J., Rijen PC., Faber JAJ., Valk J., Willemse K. Age-dependent changes in localized proton and phosphorus MR spectroscopy of the brain. Radiology. 1990. 176:509–515.
Article16.Kimura H., Fujii Y., Itoh S, et al. Metabolic alterations in neonate and infant brain during development: Evaluation with proton MR spectroscopy. Radiology. 1995. 194:483–489.17.Kreis R., Ernst T., Ross BD. Development of the human brain: In vivo quantification of metabolic and water content with proton magnetic resonance spectroscopy. MR Μ. 1993. 30:424–437.18.Webb PG., Sailasuta N., Kohler SJ., Raidy T., Moats RA., Hurd RE. Automated singlevoxel proton MRS: technical development and multisite verification. Magn Re son Med. 1994. 31:365–373.
Article19.Jung Hee Lee., Choong Gon Choi., Sang-Tae Kim, et al. Localized single voxel Ή MR spectroscopy toward routine clinical use. J Korean Radiol Soc. 1996. 34:185–191.20.In Chan Song., Kee Hyun Chang., Kwan Hong Min, et al. ΧΗMR spectroscopic patterns of normal adult brain. J Korean Radiol Soc. 1996. 35:435–440.21.Moats TA., Watson L., Shonl T, et al. Added value of automated clinical proton MR spectroscopy of the brain. J Comput Assist Tomogr. 1995. 19:480–491.
Article22.Negendank W. Studies of human tumors by MRS: a review. Ν MR Biomed. 1992. 5:303–324.
Article23.Βrun H., Michaelis T., Merboldt KD, et al. On the interpretation of proton NMR spectra from brain tumors in vivo and in vitro. NMR Biomed. 1992. 5:253–258.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- 1H MR Spectroscopy of the Normal Human Brains: Comparison between Signa and Echospeed 1.5 T system
- In vivo H MR spectroscopy of human brain in six normal volunteers
- Localized Single Voxel 1H MR Spectroscopy Toward Routine Clinical Use
- 1H MR Spectroscopic Patterns of Normal Adult Brain
- Localized, water-suppressed in vivo H MR spectroscopy of human brain tumors: Preliminary results