J Bacteriol Virol.
2012 Dec;42(4):313-320. 10.4167/jbv.2012.42.4.313.
Characterization of Bacterial Community Contaminating Floor of A Hot and Dry Sauna
- Affiliations
-
- 1Department of Chemical and Biological Engineering, Seokyeong University, Seoul, Korea. baakdoo@skuniv.ac.kr
- KMID: 2168666
- DOI: http://doi.org/10.4167/jbv.2012.42.4.313
Abstract
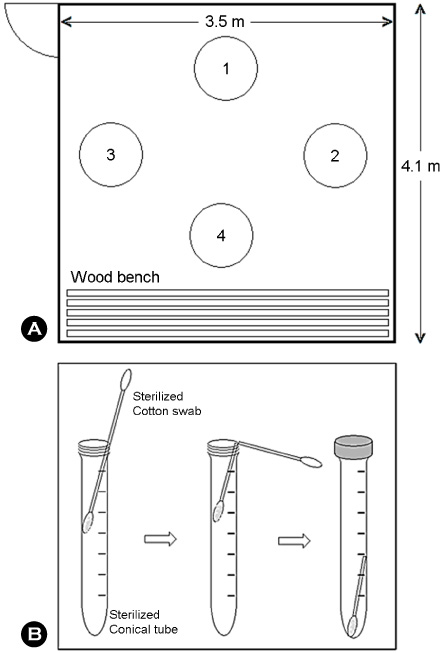
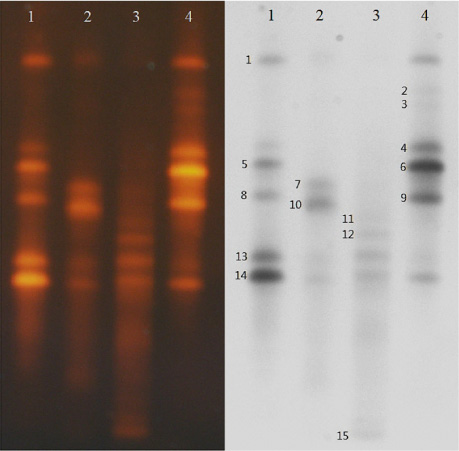
- The purpose of this study was to examine profile of bacteria contaminated in a dry sauna. Bacteria sampled from the floor of the dry sauna (75~80degrees C) were separated and identified by using a thermal gradient gel electrophoresis (TGGE) technique, and were cultivated using a defined medium at 50degrees C. Bacteria grown in the defined medium were identified based on 16S-rDNA sequence homology. The band number of DNA separated by TGGE was 15, indicating the species diversity of bacteria contaminating the dry sauna. Seven species of bacteria were successfully cultured on agar plate medium at 50degrees C, which represented a combination of thermophilic and thermoduric bacteria contaminating the dry sauna. The highest limit temperature for growth of the bacterial isolates was generally 50degrees C when cultivated in a defined medium, but was raised to 60degrees C when cultivated in a complex medium. Consequently, the bacteria grown at 50~60degrees C are thermoduric or thermophilic, but others may not be.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Variation and Characterization of Bacterial Communities Contaminating Two Saunas Operated at 64°C and 76°C
Bong Su Kim, Jae Ran Seo, Doo Hyun Park
J Bacteriol Virol. 2013;43(3):195-203. doi: 10.4167/jbv.2013.43.3.195.
Reference
-
1. Walsh C, Meade J, McGill K, Fanning S. The biodiversity of thermoduric bacteria isolated from whey. J Food Saf. 2012. 32:255–261.
Article2. Banykó J, Vyletelová M. Determining the source of Bacillus cereus and Bacillus licheniformis isolated from raw milk, pasteurised milk and yoghurt. Lett Appl Microbiol. 2009. 48:318–323.
Article3. Bott TL, Brock TD. Bacterial growth rates above 90 degrees C in Yellowstone hot springs. Science. 1969. 164:1411–1412.
Article4. Brock TD, Freeze H. Thermus aquaticus gen. n. and sp. n., a nonsporulating extreme thermophile. J Bacteriol. 1969. 98:289–297.
Article5. Brock TD, Boylen LK. Presence of thermophilic bacteria in laundry and domestic hot-water heaters. Appl Microbiol. 1973. 25:72–76.
Article6. Pask-Hughes R, Williama RA. Extremely thermophilic gram-negative bacteria from hot tap water. J Gen Microbiol. 1975. 88:321–328.
Article7. Oshima T, Imahori K. Description of Thermus thermophiles (Yoshida and Oshima) comb. Nov. A non-sporulating thermophilic bacterium from a Japanese thermal spa. Intern System Bacteriol. 1974. 24:102–112.
Article8. Ward J, Cockson A. Studies on a thermophilic bacillus: its isolation, properties, and temperature coefficient of growth. J Bacteriol. 1972. 112:1040–1042.
Article9. Metzger WJ, Patterson R, Fink J, Semerdjian R, Roberts M. Sauna-takers disease. Hypersensitivity pneumonitis due to contaminated water in a home sauna. JAMA. 1976. 236:2209–2211.
Article10. Scheldeman P, Pil A, Herman L, De Vos P, Heyndrickx M. Incidence and diversity of potentially highly heat-resistant spores isolated at dairy farms. Appl Environ Microbiol. 2005. 71:1480–1494.
Article11. Finley N, Fields ML. Heat activation and heat-induced dormancy of Bacillus stearothermophilus spores. Appl Microbiol. 1962. 10:231–236.
Article12. Hill WM, Fields ML. Factors affecting growth and interaction of rough and smooth variants of Bacillus stearothermophilus. I. Oxygen tension and temperature. J Food Sci. 1967. 32:458–462.
Article13. O'Brien RT, Campbell LL Jr. The nutritional requirements for germination and outgrowth of spores and vegetative cell growth of some aerobic spore forming bacteria. J Bacteriol. 1957. 73:522–525.14. Martin PA, Travers RS. Worldwide abundance and distribution of Bacillus thuringiensis isolates. Appl Environ Microbiol. 1989. 55:2437–2442.
Article15. Stefanic P, Mandic-Mulec I. Social interactions and distribution of Bacillus subtilis pherotypes at microscale. J Bacteriol. 2009. 191:1756–1764.
Article16. Lee SJ, Lee YW, Chung J, Lee JK, Lee JY, Jahng D, et al. Reuse of low concentrated electronic wastewater using selected microbe immobilised cell system. Water Sci Technol. 2008. 57:1191–1197.
Article17. Salkinoja-Salonen MS, Vuorio R, Andersson MA, Kämpfer P, Andersson MC, Honkanen-Buzalski T, et al. Toxigenic Strains of Bacillus licheniformis Related to Food Poisoning. Appl Environ Microbiol. 1999. 65:4637–4645.
Article18. An SY, Asahara M, Goto K, Kasai H, Yokota A. Virgibacillus halophilus sp. nov., spore-forming bacterial isolated from soil in Japan. Int J Syst Evol Microbiol. 2007. 57:1607–1611.
Article19. Earl AM, Losick R, Kolter R. Ecology and genomics of Bacillus subtilis. Trends Microbiol. 2008. 16:269–275.20. Dean DH. Biochemical genetics of the bacterial insectcontrol agent Bacillus thuringiensis: basic principles and prospects for genetic engineering. Biotechnol Genet Eng Rev. 1984. 2:341–363.
Article21. Moreira C, Rainey FA, Nobre MF, da Silva MT, da Costa MS. Tepidimonas ignava gen nov, sp nov, a new chemolithoheterotrophic and slightly thermophilic member of the beta-Proteobacteria. Int J Syst Evol Microbiol. 2000. 50:735–742.
Article22. Mountain BW, Benning LG, Boerema JA. Experimental studies on New Zealand hot spring sinters: rates of growth and textural development. Can J Earth Sci. 2003. 40:1643–1667.
Article23. Kämpfer P, Falsen E, Busse HJ. Reclassification of Pseudomonas mephitica claydon and hammer 1939 as a later heterotypic synonym of Janthinobacterium lividum (Eisenberg 1891) De Ley et al. 1978. Int J Syst Evol Microbiol. 2008. 58:136–138.24. Stead DE. Grouping of plant-pathogenic and some other Pseudomonas spp. by using cellular fatty acid profiles. IJSEM. 1992. 42:281–295.
Article25. Chen MY, Tsay SS, Chen KY, Shi YC, Lin YT, Lin GH. Pseudoxanthomonas taiwanensis sp. nov., a novel thermophilic, N2O-producing species isolated from hot springs. Int J Syst Evol Microbiol. 2002. 52:2155–2161.
Article26. Palleroni NJ, Bradbury JF. Stenotrophomonas, a new bacterial genus for Xanthomonas maltophilia (Hugh 1980) Swings et al. 1983. Int J Syst Bacteriol. 1993. 43:606–609.
Article27. Yokoya F, York GK. Effect of several environmental conditions on the "thermal death rate" of endospores of aerobic, thermophilic bacteria. Appl Microbiol. 1965. 13:993–999.
Article28. Nicholson WL, Munakata N, Horneck G, Melosh HJ, Setlow P. Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol Mol Biol Rev. 2000. 64:548–572.
Article29. Lowe SE, Jain MK, Zeikus JG. Biology, ecology, and biotechnological applications of anaerobic bacteria adapted to environmental stresses in temperature, pH, salinity, or substrates. Microbiol Rev. 1993. 57:451–509.
Article30. Heinen W. Growth conditions and temperature dependent substrate specificity of two extremely thermophilic bacteria. Arch Mikrobiol. 1971. 76:2–17.
Article31. Vogt R, Larue D, Parry MF, Brokopp CD, Klaucke D, Allen J. Pseudomonas aeruginosa skin infections in persons using a whirlpool in Vermont. J Clin Microbiol. 1982. 15:571–574.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Variation and Characterization of Bacterial Communities Contaminating Two Saunas Operated at 64degrees C and 76degrees C
- The Effect of Bath and Sauna on IOP, BP, PR, and Body Temperature
- Dry sauna therapy is beneficial for patients with low back pain
- The Effect of Air in a Sauna on Nasal Mucociliary Function
- Quantitative and Qualitative Estimation of Bacteria Contaminating Human Hairs