J Bacteriol Virol.
2010 Dec;40(4):207-212. 10.4167/jbv.2010.40.4.207.
Detection of the Avian Influenza Viruses Nonstructural Protein 1 for Distinction between Vaccinated and Infected Chickens Using Synthetic Peptide-Based ELISA
- Affiliations
-
- 1Division of Molecular and Life Sciences, Hanyang University, Ansan, Korea. ygchai@hanyang.ac.kr
- 2Bionote, Inc., Suwon, Korea.
- KMID: 2168585
- DOI: http://doi.org/10.4167/jbv.2010.40.4.207
Abstract
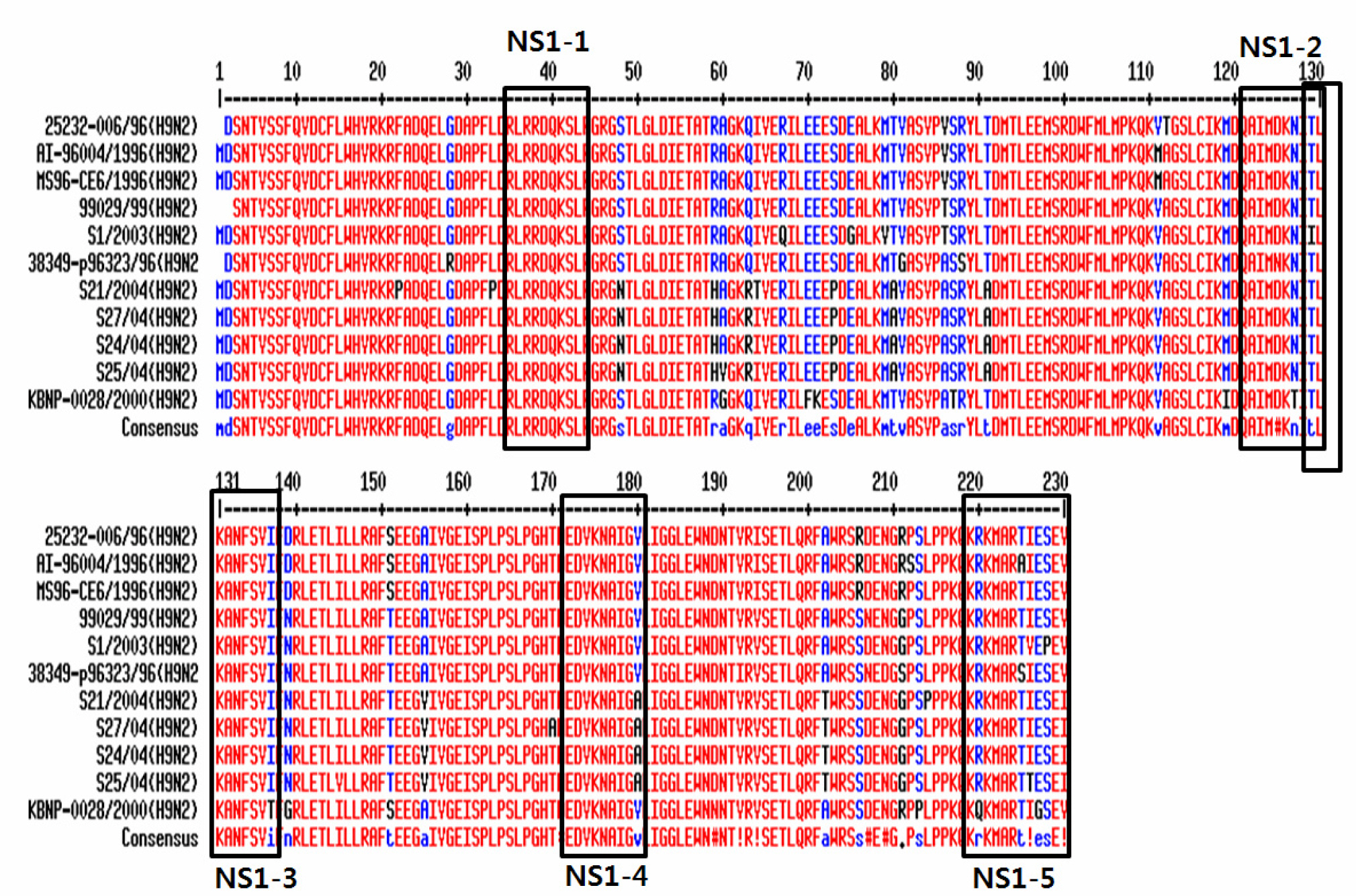
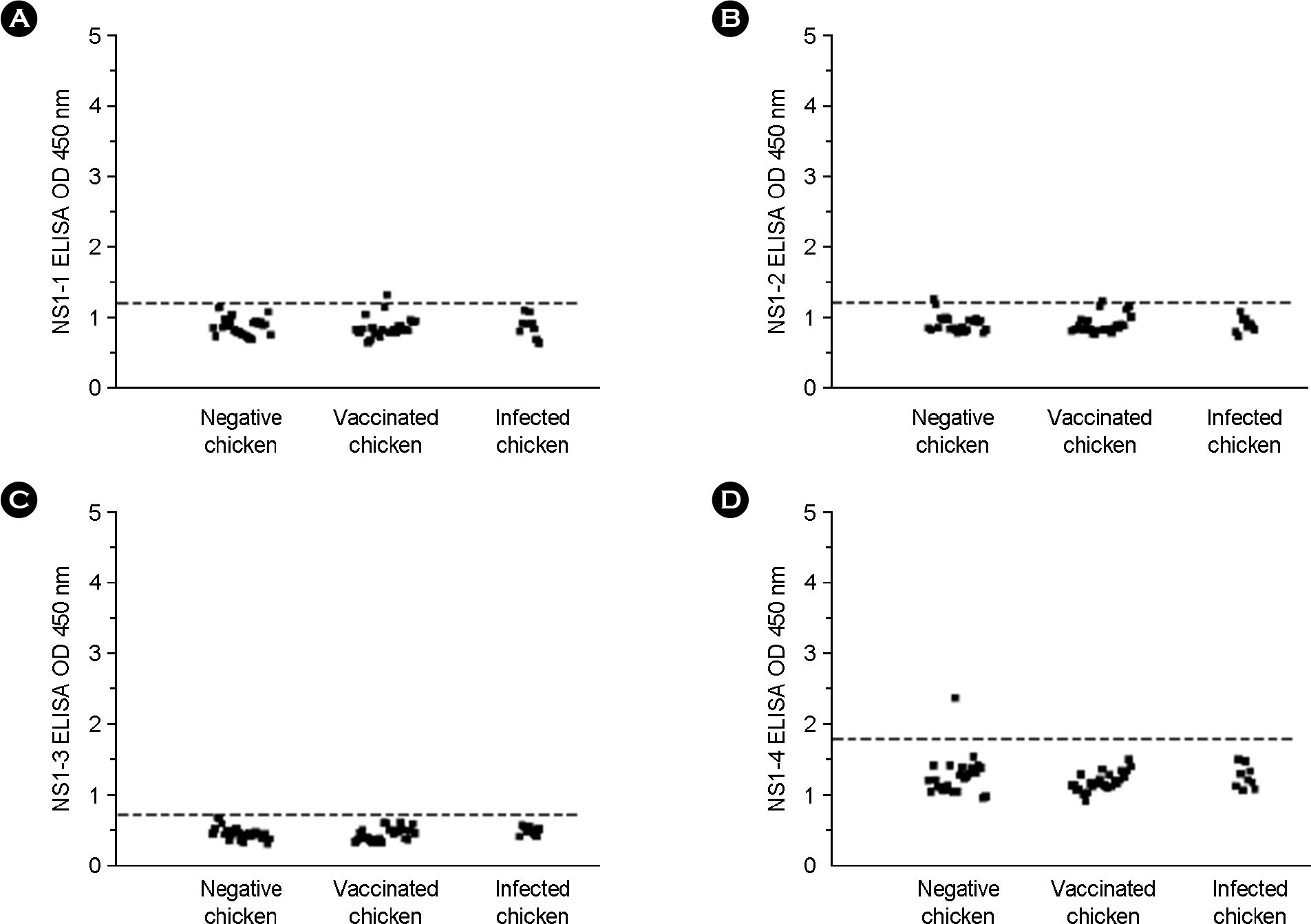
- Avian influenza (AI) virus infects both animal and human. Low pathogenic AI virus infections (some H7 and H9 subtypes) have been reported all over the world and pose a potential threat to the poultry industry. Vaccination is the most effective way to prevent virus infection. However, vaccination makes it difficult to differentiate between vaccinated chickens and infected chickens. In order to differentiate vaccinated chickens from naturally infected chickens, we adopted synthetic peptide-based enzyme-linked immunosorbent assay (ELISA) using the peptide sequences from nonstructural protein 1 (NS1) of H9N2. Five synthetic peptides were designed using Protein Variability Sever (http://imed.med.ucm.es/PVS/) and synthesized. NS1-1 ~ NS1-4 peptides failed to detect serum antibodies from both vaccinated and naturally infected chickens. NS1-5 peptide from the C-terminal NS1 protein detected serum antibody from naturally infected chickens but not vaccinated chickens. These results imply that NS1-5 peptide may be a useful tool to differentiate naturally infected chicken from vaccinated chicken as being used in the synthetic peptide-based ELISA.
MeSH Terms
Figure
Cited by 1 articles
-
Proteomic Analysis of the Serum from Chicken Infected by Avian Influenza Virus
Hyung Tae Lee, Kyoung Hwa Jung, Ji Hyun Park, Gun-Woo Ha, Youn-Kyoung Oh, Jin-Sik Oh, Young Gyu Chai
J Bacteriol Virol. 2011;41(3):195-204. doi: 10.4167/jbv.2011.41.3.195.
Reference
-
1). Yuen KY., Chan PK., Peiris M., Tsang DN., Que TL., Shortridge KF, et al. Clinical features and rapid viral diagnosis of human disease associated with avian influenza A H5N1 virus. Lancet. 1998. 351:467–71.
Article2). Swayne DE., Suarez DL. Highly pathogenic avian influenza. Rev Sci Tech. 2000. 19:463–82.
Article3). Tumpey TM., Alvarez R., Swayne DE., Suarez DL. Diagnostic approach for differentiating infected from vaccinated poultry on the basis of antibodies to NS1, the nonstructural protein of influenza A virus. J Clin Microbiol. 2005. 43:676–83.
Article4). Capua I., Alexander DJ. Avian influenza: recent developments. Avian Pathol. 2004. 33:393–404.
Article5). Dundon WG., Milani A., Cattoli G., Capua I. Progressive truncation of the Non-Structural 1 gene of H7N1 avian influenza viruses following extensive circulation in poultry. Virus Res. 2006. 119:171–6.
Article6). Webster RG., Peiris M., Chen H., Guan Y. H5N1 outbreaks and enzootic influenza. Emerg Infect Dis. 2006. 12:3–8.
Article7). Zhao S., Jin M., Li H., Tan Y., Wang G., Zhang R, et al. Detection of antibodies to the nonstructural protein (NS1) of avian influenza viruses allows distinction between vaccinated and infected chickens. Avian Dis. 2005. 49:488–93.
Article8). Banks J., Speidel EC., Harris PA., Alexander DJ. Phylogenetic analysis of influenza A viruses of H9 haemagglutinin subtype. Avian Pathol. 2000. 29:353–9.
Article9). Alexander DJ. Report on avian influenza in the Eastern Hemisphere during 1997~2002. Avian Dis. 2003. 47:792–7.
Article10). Guo YJ., Krauss S., Senne DA., Mo IP., Lo KS., Xiong XP, et al. Characterization of the pathogenicity of members of the newly established H9N2 influenza virus lineages in Asia. Virology. 2000. 267:279–88.
Article11). Lin YP., Shaw M., Gregory V., Cameron K., Lim W., Klimov A, et al. Avian-to-human transmission of H9N2 subtype influenza A viruses: relationship between H9N2 and H5N1 human isolates. Proc Natl Acad Sci U S A. 2000. 97:9654–8.
Article12). Horimoto T., Kawaoka Y. Influenza: lessons from past pandemics, warnings from current incidents. Nat Rev Microbiol. 2005. 3:591–600.
Article13). Lambrecht B., Steensels M., Van Borm S., Meulemans G., van den Berg T. Development of an M2e-specific enzyme-linked immunosorbent assay for differentiating infected from vaccinated animals. Avian Dis. 2007. 51:221–6.
Article14). Jeong OM., Kim MC., Kang HM., Ha GW., Oh JS., Yoo JE, et al. Validation of egg yolk antibody based C-ELISA for avian influenza surveillance in breeder duck. Vet Microbiol. 2010. 144:287–92.
Article15). Hale BG., Randall RE., Ortín J., Jackson D. The multifunctional NS1 protein of influenza A viruses. J Gen Virol. 2008. 89:2359–76.
Article16). Ghedin E., Sengamalay NA., Shumway M., Zaborsky J., Feldblyum T., Subbu V, et al. Large-scale sequencing of human influenza reveals the dynamic nature of viral genome evolution. Nature. 2005. 437:1162–6.
Article17). Wang W., Riedel K., Lynch P., Chien CY., Montelione GT., Krug RM. RNA binding by the novel helical domain of the influenza virus NS1 protein requires its dimer structure and a small number of specific basic amino acids. RNA. 1999. 5:195–205.
Article18). Basler CF., Aguilar PV. Progress in identifying virulence determinants of the 1918 H1N1 and the Southeast Asian H5N1 influenza A viruses. Antiviral Res. 2008. 79:166–78.
Article19). Krug RM., Etkind PR. Cytoplasmic and nuclear virus-specific proteins in influenza virus-infected MDCK cells. Virology. 1973. 56:334–48.
Article20). Corpet F. Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res. 1988. 16:10881–90.
Article21). Dundon WG., Maniero S., Toffan A., Capua I., Cattoli G. Appearance of serum antibodies against the avian influenza nonstructural 1 protein in experimentally infected chickens and turkeys. Avian Dis. 2007. 51:209–12.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- H5 and H9 subtypes of Avian Influenza Viruses are Real Threat To Humans
- Proteomic Analysis of the Serum from Chicken Infected by Avian Influenza Virus
- Development of synthetic peptide ELISA based on nonstructural protein 2C of foot and mouth disease virus
- Development of Competitive ELISA for Detection of Avian Metapneumovirus Antibodies in Chicken
- Pathogenicity of H5N8 virus in chickens from Korea in 2014