J Periodontal Implant Sci.
2016 Apr;46(2):84-95. 10.5051/jpis.2016.46.2.84.
Assessment of MMP-1, MMP-8 and TIMP-2 in experimental periodontitis treated with kaempferol
- Affiliations
-
- 1Department of Periodontology, Bulent Ecevit University Faculty of Dentistry, Zonguldak, Turkey. umutballi@gmail.com
- 2Department of Periodontology, Ondokuzmayis University Faculty of Dentistry, Samsun, Turkey.
- 3Ondokuzmayis University Samsun High School of Health, Samsun, Turkey.
- 4Department of Histology and Embryology, Giresun University Faculty of Medicine, Giresun, Turkey.
- KMID: 2161985
- DOI: http://doi.org/10.5051/jpis.2016.46.2.84
Abstract
- PURPOSE
The objective of this study was to investigate the effect of a dietary flavonoid, kaempferol, which has been shown to possess antiallergic, anti-inflammatory, anticarcinogenic, and antioxidant activities on the periodontium by histomorphometric analysis and on gingival tissue matrix metalloproteinase-1 (MMP-1), MMP-8, and tissue inhibitor of metalloproteinase-2 (TIMP-2) by biochemical analysis of rats after experimental periodontitis induction.
METHODS
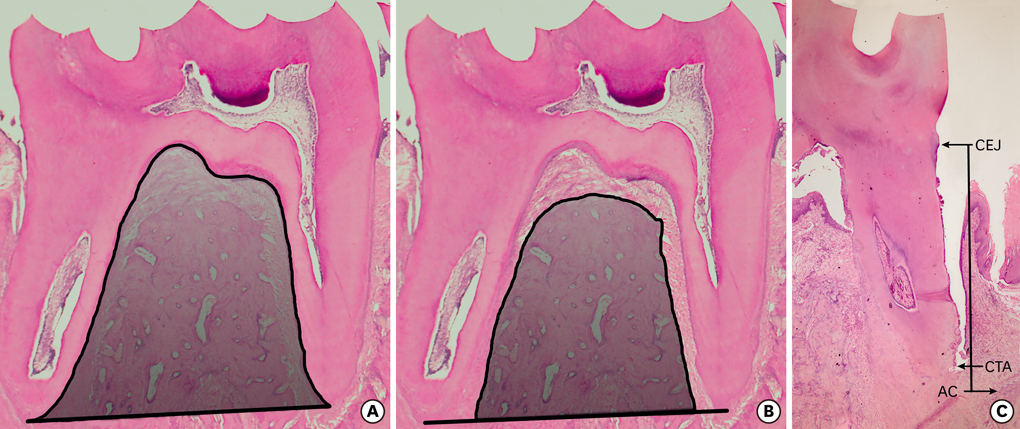
Sixty Wistar rats were randomly divided into six groups of ten rats each, and silk ligatures were placed around the cervical area of the mandibular first molars for 15 days, except in the healthy control rats. In the experimental periodontitis groups, systemic kaempferol (10 mg/kg/2d) and saline were administered by oral gavage at two different periods (with and without the presence of dental biofilm) to all rats except for the ten non-medicated rats. Alveolar bone area, alveolar bone level, and attachment level were determined by histomorphometric analysis, and gingival tissue levels of MMP-1, MMP-8, and TIMP-2 were detected by biochemical analysis.
RESULTS
Significantly greater bone area and significantly less alveolar bone and attachment loss were observed in the kaempferol application groups compared to the control groups (P<0.05). In addition, gingival tissue MMP-1 and -8 levels were significantly lower in the kaempferol application groups compared to the control groups and the periodontitis group (P<0.001). There were no statistically significant differences in TIMP-2 levels between the kaempferol and saline application groups (P>0.05).
CONCLUSIONS
Kaempferol application may be useful in decreasing alveolar bone resorption, attachment loss, and MMP-1 and -8 production in experimental periodontitis.
MeSH Terms
Figure
Reference
-
1. Giannobile WV. Host-response therapeutics for periodontal diseases. J Periodontol. 2008; 79:Suppl. 1592–1600.
Article2. Bascones-Martínez A, Muñoz-Corcuera M, Noronha S, Mota P, Bascones-Ilundain C, Campo-Trapero J. Host defence mechanisms against bacterial aggression in periodontal disease: Basic mechanisms. Med Oral Patol Oral Cir Bucal. 2009; 14:e680–5.3. Page RC, Kornman KS. The pathogenesis of human periodontitis: an introduction. Periodontol 2000. 1997; 14:9–11.
Article4. Page RC. Milestones in periodontal research and the remaining critical issues. J Periodontal Res. 1999; 34:331–339.
Article5. Nagase H, Woessner JF Jr. Matrix metalloproteinases. J Biol Chem. 1999; 274:21491–21494.
Article6. Sapna G, Gokul S, Bagri-Manjrekar K. Matrix metalloproteinases and periodontal diseases. Oral Dis. 2014; 20:538–550.
Article7. Pozo P, Valenzuela MA, Melej C, Zaldívar M, Puente J, Martínez B, et al. Longitudinal analysis of metalloproteinases, tissue inhibitors of metalloproteinases and clinical parameters in gingival crevicular fluid from periodontitis-affected patients. J Periodontal Res. 2005; 40:199–207.
Article8. Kubota T, Itagaki M, Hoshino C, Nagata M, Morozumi T, Kobayashi T, et al. Altered gene expression levels of matrix metalloproteinases and their inhibitors in periodontitis-affected gingival tissue. J Periodontol. 2008; 79:166–173.
Article9. Tüter G, Kurtiş B, Serdar M. Effects of phase I periodontal treatment on gingival crevicular fluid levels of matrix metalloproteinase-1 and tissue inhibitor of metalloproteinase-1. J Periodontol. 2002; 73:487–493.
Article10. Marcaccini AM, Novaes AB Jr, Meschiari CA, Souza SL, Palioto DB, Sorgi CA, et al. Circulating matrix metalloproteinase-8 (MMP-8) and MMP-9 are increased in chronic periodontal disease and decrease after non-surgical periodontal therapy. Clin Chim Acta. 2009; 409:117–122.
Article11. Marcaccini AM, Meschiari CA, Zuardi LR, de Sousa TS, Taba M Jr, Teofilo JM, et al. Gingival crevicular fluid levels of MMP-8, MMP-9, TIMP-2, and MPO decrease after periodontal therapy. J Clin Periodontol. 2010; 37:180–190.
Article12. Kuula H, Salo T, Pirilä E, Tuomainen AM, Jauhiainen M, Uitto VJ, et al. Local and systemic responses in matrix metalloproteinase 8-deficient mice during Porphyromonas gingivalis-induced periodontitis. Infect Immun. 2009; 77:850–859.
Article13. Hernández M, Gamonal J, Salo T, Tervahartiala T, Hukkanen M, Tjäderhane L, et al. Reduced expression of lipopolysaccharide-induced CXC chemokine in Porphyromonas gingivalis-induced experimental periodontitis in matrix metalloproteinase-8 null mice. J Periodontal Res. 2011; 46:58–66.
Article14. Butler GS, Butler MJ, Atkinson SJ, Will H, Tamura T, Schade van Westrum S, et al. The TIMP2 membrane type 1 metalloproteinase “receptor” regulates the concentration and efficient activation of progelatinase A. A kinetic study. J Biol Chem. 1998; 273:871–880.
Article15. Meschiari CA, Marcaccini AM, Santos Moura BC, Zuardi LR, Tanus-Santos JE, Gerlach RF. Salivary MMPs, TIMPs, and MPO levels in periodontal disease patients and controls. Clin Chim Acta. 2013; 421:140–146.
Article16. Kubota T, Matsuki Y, Nomura T, Hara K. In situ hybridization study on tissue inhibitors of metalloproteinases (TIMPs) mRNA-expressing cells in human inflamed gingival tissue. J Periodontal Res. 1997; 32:467–472.
Article17. Calderón-Montaño JM, Burgos-Morón E, Pérez-Guerrero C, López-Lázaro M. A review on the dietary flavonoid kaempferol. Mini Rev Med Chem. 2011; 11:298–344.
Article18. Choi IS, Choi EY, Jin JY, Park HR, Choi JI, Kim SJ. Kaempferol inhibits P. intermedia lipopolysaccharide-induced production of nitric oxide through translational regulation in murine macrophages: critical role of heme oxygenase-1-mediated ROS reduction. J Periodontol. 2013; 84:545–555.
Article19. Coimbra LS, Rossa C Jr, Guimarães MR, Gerlach RF, Muscará MN, Spolidorio DM, et al. Influence of antiplatelet drugs in the pathogenesis of experimental periodontitis and periodontal repair in rats. J Periodontol. 2011; 82:767–777.
Article20. Kim HK, Park HR, Lee JS, Chung TS, Chung HY, Chung J. Down-regulation of iNOS and TNF-alpha expression by kaempferol via NF-kappaB inactivation in aged rat gingival tissues. Biogerontology. 2007; 8:399–408.
Article21. Balli U, Keles GC, Cetinkaya BO, Mercan U, Ayas B, Erdogan D. Assessment of vascular endothelial growth factor and matrix metalloproteinase-9 in the periodontium of rats treated with atorvastatin. J Periodontol. 2014; 85:178–187.
Article22. Donatelli RE, Lee SJ. How to report reliability in orthodontic research: Part 1. Am J Orthod Dentofacial Orthop. 2013; 144:156–161.
Article23. Kim HY. Statistical notes for clinical researchers: Evaluation of measurement error 1: using intraclass correlation coefficients. Restor Dent Endod. 2013; 38:98–102.
Article24. Klausen B. Microbiological and immunological aspects of experimental periodontal disease in rats: a review article. J Periodontol. 1991; 62:59–73.
Article25. Aiba T, Akeno N, Kawane T, Okamoto H, Horiuchi N. Matrix metalloproteinases-1 and -8 and TIMP-1 mRNA levels in normal and diseased human gingivae. Eur J Oral Sci. 1996; 104:562–569.
Article26. Kou Y, Inaba H, Kato T, Tagashira M, Honma D, Kanda T, et al. Inflammatory responses of gingival epithelial cells stimulated with Porphyromonas gingivalis vesicles are inhibited by hop-associated polyphenols. J Periodontol. 2008; 79:174–180.
Article27. Yoon HY, Lee EG, Lee H, Cho IJ, Choi YJ, Sung MS, et al. Kaempferol inhibits IL-1β-induced proliferation of rheumatoid arthritis synovial fibroblasts and the production of COX-2, PGE2 and MMPs. Int J Mol Med. 2013; 32:971–977.
Article28. Sim GS, Lee BC, Cho HS, Lee JW, Kim JH, Lee DH, et al. Structure activity relationship of antioxidative property of flavonoids and inhibitory effect on matrix metalloproteinase activity in UVA-irradiated human dermal fibroblast. Arch Pharm Res. 2007; 30:290–298.
Article29. Kowalski J, Samojedny A, Paul M, Pietsz G, Wilczok T. Effect of kaempferol on the production and gene expression of monocyte chemoattractant protein-1 in J774.2 macrophages. Pharmacol Rep. 2005; 57:107–112.30. Sin BY, Kim HP. Inhibition of collagenase by naturally-occurring flavonoids. Arch Pharm Res. 2005; 28:1152–1155.
Article31. Kubota T, Nomura T, Takahashi T, Hara K. Expression of mRNA for matrix metalloproteinases and tissue inhibitors of metalloproteinases in periodontitis-affected human gingival tissue. Arch Oral Biol. 1996; 41:253–262.
Article32. Lin CW, Chen PN, Chen MK, Yang WE, Tang CH, Yang SF, et al. Kaempferol reduces matrix metalloproteinase-2 expression by down-regulating ERK1/2 and the activator protein-1 signaling pathways in oral cancer cells. PLoS One. 2013; 8:e80883.
Article33. Ji L, Yin XX, Wu ZM, Wang JY, Lu Q, Gao YY. Ginkgo biloba extract prevents glucose-induced accumulation of ECM in rat mesangial cells. Phytother Res. 2009; 23:477–485.
Article34. Lu Q, Yin XX, Wang JY, Gao YY, Pan YM. Effects of Ginkgo biloba on prevention of development of experimental diabetic nephropathy in rats. Acta Pharmacol Sin. 2007; 28:818–828.
Article35. Reddy MS, Jeffcoat MK. Methods of assessing periodontal regeneration. Periodontol 2000. 1999; 19:87–103.
Article36. Pang JL, Ricupero DA, Huang S, Fatma N, Singh DP, Romero JR, et al. Differential activity of kaempferol and quercetin in attenuating tumor necrosis factor receptor family signaling in bone cells. Biochem Pharmacol. 2006; 71:818–826.
Article37. Trivedi R, Kumar S, Kumar A, Siddiqui JA, Swarnkar G, Gupta V, et al. Kaempferol has osteogenic effect in ovariectomized adult Sprague-Dawley rats. Mol Cell Endocrinol. 2008; 289:85–93.
Article38. Nepal M, Li L, Cho HK, Park JK, Soh Y. Kaempferol induces chondrogenesis in ATDC5 cells through activation of ERK/BMP-2 signaling pathway. Food Chem Toxicol. 2013; 62:238–245.
Article39. Wattel A, Kamel S, Mentaverri R, Lorget F, Prouillet C, Petit JP, et al. Potent inhibitory effect of naturally occurring flavonoids quercetin and kaempferol on in vitro osteoclastic bone resorption. Biochem Pharmacol. 2003; 65:35–42.
Article40. Yang L, Takai H, Utsunomiya T, Li X, Li Z, Wang Z, et al. Kaempferol stimulates bone sialoprotein gene transcription and new bone formation. J Cell Biochem. 2010; 110:1342–1355.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Influence of Diabetes on of PGE2, MMP-14 and TIMP Expressions in Human Chronic Periodontitis
- MMP-2 and MMP-9 Expressions in Breast Carcinomas and Relationship with Major Prognostic Factors
- Immunodetections of the Metalloproteinase (MMP-2 and MMP-9) and Tissue Inhibitor of Metalloproteinases (TIMP-2) in Prostatic Adenocarcinomas
- Expression of Matrix Metalloproteinase-2 (MMP-2) and Tissue Inhibitor of Metalloproteinase-2 (TIMP-2) in Pancreatic Ductal Adenocarcinoma
- Expression of MMP-2, MT1-MMP, and TIMP-2 mRNA in Breast Carcinomas