J Korean Med Sci.
2012 Apr;27(4):363-369. 10.3346/jkms.2012.27.4.363.
Iron Overload during Follow-up after Tandem High-Dose Chemotherapy and Autologous Stem Cell Transplantation in Patients with High-Risk Neuroblastoma
- Affiliations
-
- 1Faculty of Human Medicine, Hannover Medical School, Hannover, Germany.
- 2Faculty of Medicine, University of British Columbia, Vancouver, Canada.
- 3Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. kwsped@skku.edu
- 4Department of Pediatrics, Dong-A Medical Center, Dong-A University College of Medicine, Busan, Korea.
- KMID: 2157895
- DOI: http://doi.org/10.3346/jkms.2012.27.4.363
Abstract
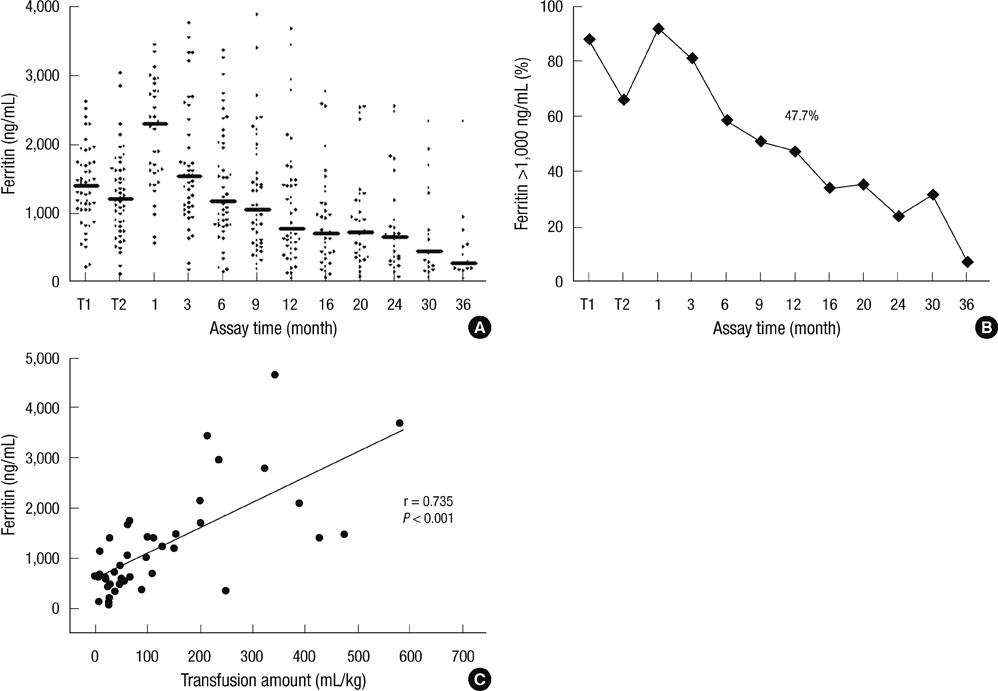
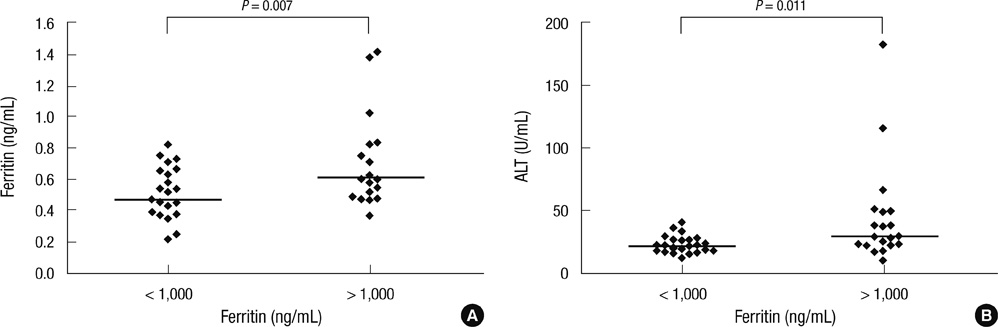
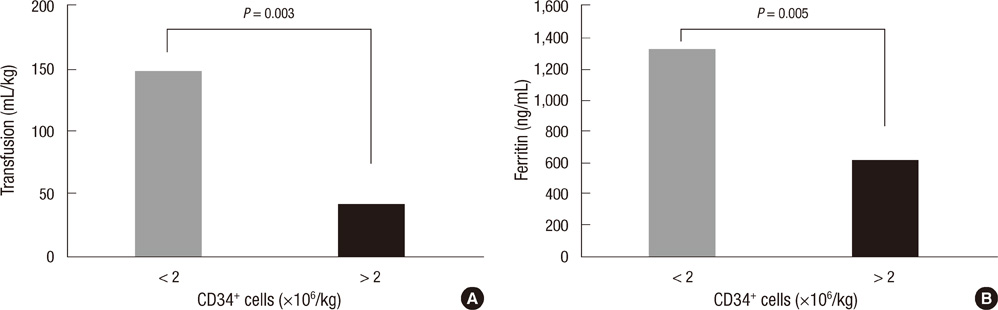
- Multiple RBC transfusions inevitably lead to a state of iron overload before and after high-dose chemotherapy and autologous stem cell transplantation (HDCT/autoSCT). Nonetheless, iron status during post-SCT follow-up remains unknown. Therefore, we investigated post-SCT ferritin levels, factors contributing to its sustained levels, and organ functions affected by iron overload in 49 children with high-risk neuroblastoma who underwent tandem HDCT/autoSCT. Although serum ferritin levels gradually decreased during post-SCT follow-up, 47.7% of the patients maintained ferritin levels above 1,000 ng/mL at 1 yr after the second HDCT/autoSCT. These patients had higher serum creatinine (0.62 vs 0.47 mg/mL, P = 0.007) than their counterparts (< 1,000 ng/mL). Post-SCT transfusion amount corresponded to increased ferritin levels at 1 yr after the second HDCT/autoSCT (P < 0.001). A lower CD34+ cell count was associated with a greater need of RBC transfusion, which in turn led to a higher serum ferritin level at 1 yr after HDCT/autoSCT. The number of CD34+ cells transplanted was an independent factor for ferritin levels at 1 yr after the second HDCT/autoSCT (P = 0.019). Consequently, CD34+ cells should be transplanted as many as possible to prevent the sustained iron overload after tandem HDCT/autoSCT and consequent adverse effects.
Keyword
MeSH Terms
-
Antigens, CD34/metabolism
Antineoplastic Combined Chemotherapy Protocols/*therapeutic use
Benzoic Acids/therapeutic use
Blood Transfusion/*adverse effects
Child
Child, Preschool
Creatinine/blood
Ferritins/blood
Follow-Up Studies
Humans
Infant
Iron Chelating Agents/therapeutic use
Iron Overload/*etiology
Neuroblastoma/drug therapy/*therapy
Retrospective Studies
Risk Factors
*Stem Cell Transplantation
Transplantation, Autologous
Triazoles/therapeutic use
Figure
Reference
-
1. Gilman AL, Jacobsen C, Bunin N, Levine J, Goldman F, Bendel A, Joyce M, Anderson P, Rozans M, Wall DA, Macdonald TJ, Simon S, Kadota RP. Phase I study of tandem high-dose chemotherapy with autologous peripheral blood stem cell rescue for children with recurrent brain tumors: a Pediatric Blood and Marrow Consortium study. Pediatr Blood Cancer. 2011. 57:506–513.2. Chen AR, Civin CI. Blume KJ, Forman SJ, Appelbaum FR, editors. Hematopoietic cell transplantation for pediatric patients with solid tumors. Thomas' hematopoietic cell transplantation. 2004. Massachusetts: Blackwell;1354–1368.3. Koreth J, Antin JH. Iron overload in hematologic malignancies and outcome of allogeneic hematopoietic stem cell transplantation. Haematologica. 2010. 95:364–366.4. Lee SH, Yoo KH, Sung KW, Koo HH, Kwon YJ, Kwon MM, Park HJ, Park BK, Kim YY, Park JA, Im HJ, Seo JJ, Kang HJ, Shin HY, Ahn HS. Hepatic veno-occlusive disease in children after hematopoietic stem cell transplantation: incidence, risk factors, and outcome. Bone Marrow Transplant. 2009. 45:1287–1293.5. de Witte T. The role of iron in patients after bone marrow transplantation. Blood Rev. 2008. 22:S22–S28.6. Altès A, Remacha AF, Sureda A, Martino R, Briones J, Canals C, Brunet S, Sierra J, Gimferrer E. Iron overload might increase transplant-related mortality in haematopoietic stem cell transplantation. Bone Marrow Transplant. 2002. 29:987–989.7. Carmine TC, Evans P, Bruchelt G, Evans R, Handgretinger R, Niethammer D, Halliwell B. Presence of iron catalytic for free radical reactions in patients undergoing chemotherapy: implications for therapeutic management. Cancer Lett. 1995. 94:219–226.8. Halliwell B, Gutteridge JM, Cross CE. Free radicals, antitoxidants, and human disease: where are we now? J Lab Clin Med. 1992. 119:598–620.9. Jensen PD, Jensen FT, Christensen T, Nielsen JL, Ellegaard J. Relationship between hepatocellular injury and transfusional iron overload prior to and during iron chelation with desferrioxamine: a study in adult patients with acquired anemias. Blood. 2003. 101:91–96.10. Malcovati L, Porta MG, Pascutto C, Invernizzi R, Boni M, Travaglino E, Passamonti F, Arcaini L, Maffioli M, Bernasconi P, Lazzarino M, Cazzola M. Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J Clin Oncol. 2005. 23:7594–7603.11. Flanagan JM, Steward S, Hankins JS, Howard TM, Neale G, Ware RE. Microarray analysis of liver gene expression in iron overloaded patients with sickle cell anemia and beta-thalassemia. Am J Hematol. 2009. 84:328–334.12. Harmatz P, Butensky E, Quirolo K, Williams R, Ferrell L, Moyer T, Golden D, Neumayr L, Vichinsky E. Severity of iron overload in patients with sickle cell disease receiving chronic red blood cell transfusion therapy. Blood. 2000. 96:76–79.13. Files B, Brambilla D, Kutlar A, Miller S, Vichinsky E, Wang W, Granger S, Adams RJ. Longitudinal changes in ferritin during chronic transfusion: a report from the Stroke Prevention Trial in Sickle Cell Anemia (STOP). J Pediatr Hematol Oncol. 2002. 24:284–290.14. Majhail NS, Lazarus HM, Burns LJ. Iron overload in hematopoietic cell transplantation. Bone Marrow Transplant. 2008. 41:997–1003.15. Koreth J, Antin JH. Iron overload in hematologic malignancies and outcome of allogeneic hematopoietic stem cell transplantation. Haematologica. 2010. 95:364–366.16. Strasser SI, Kowdley KV, Sale GE, McDonald GB. Iron overload in bone marrow transplant recipients. Bone Marrow Transplant. 1998. 22:167–173.17. Bazuave GN, Buser A, Gerull S, Tichelli A, Stern M. Prognostic impact of iron parameters in patients undergoing allo-SCT. Bone Marrow Transplant. 2012. 47:60–64.18. Butt NM, Clark RE. Autografting as a risk factor for persisting iron overload in long-term survivors of acute myeloid leukaemia. Bone Marrow Transplant. 2003. 32:909–913.19. Chueh HW, Sung KW, Lee SH, Yoo KH, Koo HH, Kim JY, Cho EJ. Iron chelation treatment with deferasirox prior to high-dose chemotherapy and autologous stem cell transplantation may reduce the risk of hepatic veno-occlusive disease in children with high-risk solid tumors. Pediatr Blood Cancer. 2012. 58:441–447.20. Delforge M, Selleslag D, Triffet A, Mineur P, Bries G, Graux C, Trullemans F, MacDonald K, Abraham I, Pluymers W, Ravoet C. Iron status and treatment modalities in transfusion-dependent patients with myelodysplastic syndromes. Ann Hematol. 2011. 90:655–666.21. Cappellini MD, Cohen A, Piga A, Bejaoui M, Perrotta S, Agaoglu L, Aydinok Y, Kattamis A, Kilinc Y, Porter J, Capra M, Galanello R, Fattoum S, Drelichman G, Magnano C, Verissimo M, Athanassiou-Metaxa M, Giardina P, Kourakli-Symeonidis A, Janka-Schaub G, Coates T, Vermylen C, Olivieri N, Thuret I, Opitz H, Ressayre-Djaffer C, Marks P, Alberti D. A phase 3 study of deferasirox (ICL670), a once-daily oral iron chelator in patients with beta-thalassemia. Blood. 2006. 107:3455–3462.22. Kwiatkowski JL. Oral iron chelators. Hematol Oncol Clin N Am. 2010. 24:229–248.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment of high-risk neuroblastoma
- Hematologic Recovery after Tandem High-Dose Chemotherapy and Autologous Stem Cell Transplantation in Children with High-Risk Solid Tumors
- A Case of Autologous Cord Blood Stem Cell Transplantationin Stage IV Neuroblastoma
- High Dose Chemotherapy and Autologous Peripheral Blood Stem Cell Transplantation in Pediatric Patients with High-risk Neuroblastoma
- High-dose Chemotherapy and Autologous Stem Cell Rescue in Patients with High-risk Stage 3 Neuroblastoma: 10-Year Experience at a Single Center