Immune Netw.
2014 Aug;14(4):201-206. 10.4110/in.2014.14.4.201.
IL-33 Priming Enhances Peritoneal Macrophage Activity in Response to Candida albicans
- Affiliations
-
- 1School of Biological Sciences, University of Ulsan, Ulsan 680-749, Korea. bkwon@mail.ulsan.ac.kr
- 2Biomedical Research Center, Ulsan University Hospital, School of Medicine, University of Ulsan, Ulsan 682-714, Korea. hrcho@uuh.ulsan.kr
- 3Department of Surgery, Ulsan University Hospital, School of Medicine, University of Ulsan, Ulsan 682-714, Korea.
- KMID: 2150808
- DOI: http://doi.org/10.4110/in.2014.14.4.201
Abstract
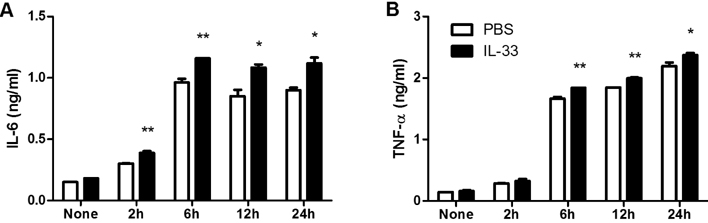
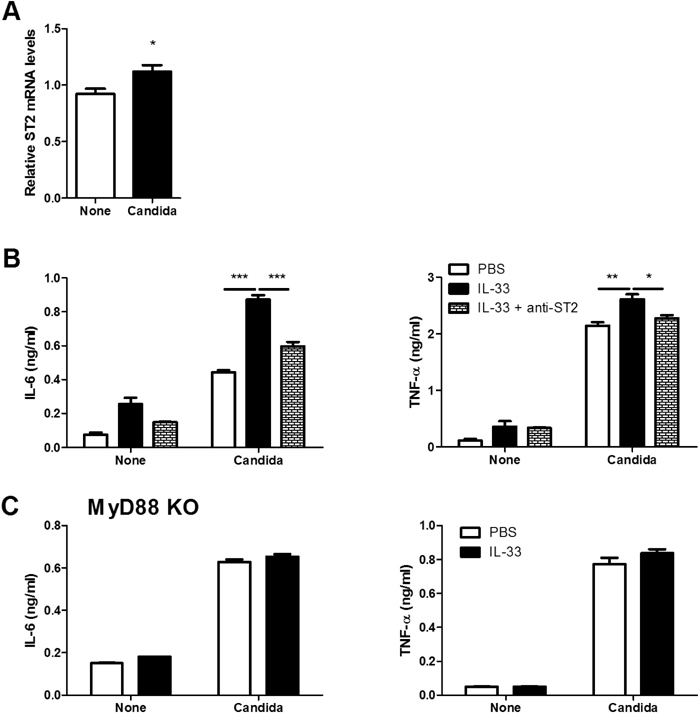
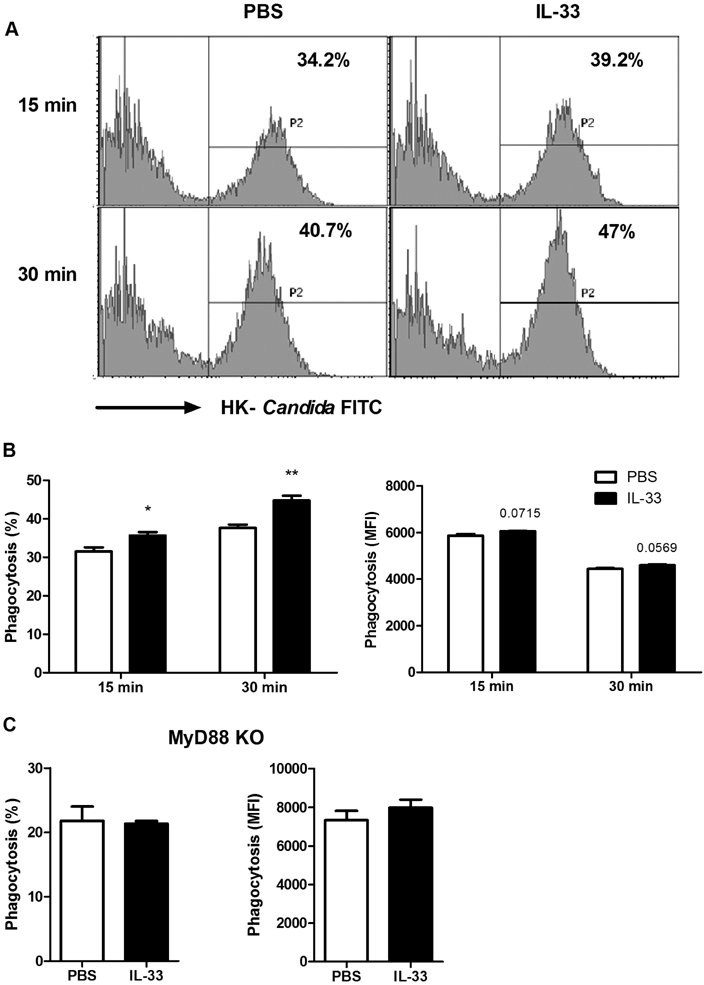
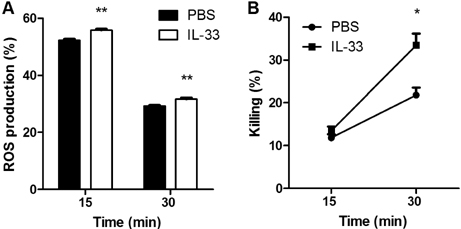
- IL-33 is a member of the IL-1 cytokine family and plays a role in the host defense against bacteria, viruses, and fungi. In this study, we investigated the function of IL-33 and its receptor in in vitro macrophage responses to Candida albicans. Our results demonstrate that pre-sensitization of isolated peritoneal macrophages with IL-33 enhanced their pro-inflammatory cytokine production and phagocytic activity in response to C. albicans. These macrophage activities were entirely dependent on the ST2-MyD88 signaling pathway. In addition, pre-sensitization with IL-33 also increased ROS production and the subsequent killing ability of macrophages following C. albicans challenge. These results indicate that IL-33 may increase anti-fungal activity against Candida through macrophage-mediated resistance mechanisms.
Keyword
MeSH Terms
Figure
Reference
-
1. Moussion C, Ortega N, Girard JP. The IL-1-like cytokine IL-33 is constitutively expressed in the nucleus of endothelial cells and epithelial cells in vivo: a novel 'alarmin'? PLoS One. 2008; 3:e3331.2. Liew FY, Pitman NI, McInnes IB. Disease-associated functions of IL-33: the new kid in the IL-1 family. Nat Rev Immunol. 2010; 10:103–110.
Article3. Le H, Kim W, Kim J, Cho HR, Kwon B. Interleukin-33: a mediator of inflammation targeting hematopoietic stem and progenitor cells and their progenies. Front Immunol. 2013; 4:104.
Article4. Romani L. Immunity to fungal infections. Nat Rev Immunol. 2011; 11:275–288.
Article5. Lionakis MS, Netea MG. Candida and host determinants of susceptibility to invasive candidiasis. PLoS Pathog. 2013; 9:e1003079.6. Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC. Hidden killers: human fungal infections. Sci Transl Med. 2012; 4:165rv13.
Article7. Lionakis MS, Fischer BG, Lim JK, Swamydas M, Wan W, Richard Lee CC, Cohen JI, Scheinberg P, Gao JL, Murphy PM. Chemokine receptor Ccr1 drives neutrophil-mediated kidney immunopathology and mortality in invasive candidiasis. PLoS Pathog. 2012; 8:e1002865.
Article8. Majer O, Bourgeois C, Zwolanek F, Lassnig C, Kerjaschki D, Mack M, Müller M, Kuchler K. Type I interferons promote fatal immunopathology by regulating inflammatory monocytes and neutrophils during Candida infections. PLoS Pathog. 2012; 8:e1002811.9. Le HT, Tran VG, Kim W, Kim J, Cho HR, Kwon B. IL-33 priming regulates multiple steps of the neutrophil-mediated anti-Candida albicans response by modulating TLR and dectin-1 signals. J Immunol. 2012; 189:287–295.
Article10. Kim J, Kim W, Le HT, Moon UJ, Tran VG, Kim HJ, Jung S, Nguyen QT, Kim BS, Jun JB, Cho HR, Kwon B. IL-33-Induced Hematopoietic Stem and Progenitor Cell Mobilization Depends upon CCR2. J Immunol. 2014; doi: 10.4049/jimmunol.1400176.
Article11. Schmitz J, Owyang A, Oldham E, Song Y, Murphy E, McClanahan TK, Zurawski G, Moshrefi M, Qin J, Li X, Gorman DM, Bazan JF, Kastelein RA. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity. 2005; 23:479–490.
Article12. Brown GD. Innate antifungal immunity: the key role of phagocytes. Annu Rev Immunol. 2011; 29:1–21.
Article13. Cheng SC, Joosten LA, Kullberg BJ, Netea MG. Interplay between Candida albicans and the mammalian innate host defense. Infect Immun. 2012; 80:1304–1313.
Article14. Missall TA, Lodge JK, McEwen JE. Mechanisms of resistance to oxidative and nitrosative stress: implications for fungal survival in mammalian hosts. Eukaryot Cell. 2004; 3:835–846.
Article15. Wellington M, Dolan K, Krysan DJ. Live Candida albicans suppresses production of reactive oxygen species in phagocytes. Infect Immun. 2009; 77:405–413.
Article16. Lewis LE, Bain JM, Lowes C, Gillespie C, Rudkin FM, Gow NA, Erwig LP. Stage specific assessment of Candida albicans phagocytosis by macrophages identifies cell wall composition and morphogenesis as key determinants. PLoS Pathog. 2012; 8:e1002578.17. Marcil A, Harcus D, Thomas DY, Whiteway M. Candida albicans killing by RAW 2647 mouse macrophage cells: effects of Candida genotype, infection ratios, and gamma interferon treatment. Infect Immun. 2002; 70:6319–6329.
Article18. Galès A, Conduché A, Bernad J, Lefevre L, Olagnier D, Béraud M, Martin-Blondel G, Linas MD, Auwerx J, Coste A, Pipy B. PPARγ controls Dectin-1 expression required for host antifungal defense against Candida albicans. PLoS Pathog. 2010; 6:e1000714.19. Nelson MP, Christmann BS, Werner JL, Metz AE, Trevor JL, Lowell CA, Steele C. IL-33 and M2a alveolar macrophages promote lung defense against the atypical fungal pathogen Pneumocystis murina. J Immunol. 2011; 186:2372–2381.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Roles of IL-33 in Resistance and Tolerance to Systemic Candida albicans Infections
- Candida spp.- Induced Cytokine Gene Expression on Mouse Peritoneal Macrophages and NIH 3T3 Fibroblasts
- In vitro Evaluation of the Antifungal Activity of Propolis Extract on Cryptococcus neoformans and Candida albicans
- Phagocytosis and intracellular killing of candida albicans by peritoneal macrophages in CAPD patients
- Candida zeylanoides Peritonitis in a Continuous Ambulatory Peritoneal Dialysis Patient