Korean J Physiol Pharmacol.
2016 Jan;20(1):83-89. 10.4196/kjpp.2016.20.1.83.
Effect of D-glucose feeding on mortality induced by sepsis
- Affiliations
-
- 1Department of Pharmacology, Institute of Natural Medicine, College of Medicine Hallym University, Chuncheon 24252, Korea. hwsuh@hallym.ac.kr
- 2Adult Stem Cell Research Center in Kangstem Biotech, #81, Seoul National University, Seoul 08826, Korea.
- KMID: 2150477
- DOI: http://doi.org/10.4196/kjpp.2016.20.1.83
Abstract
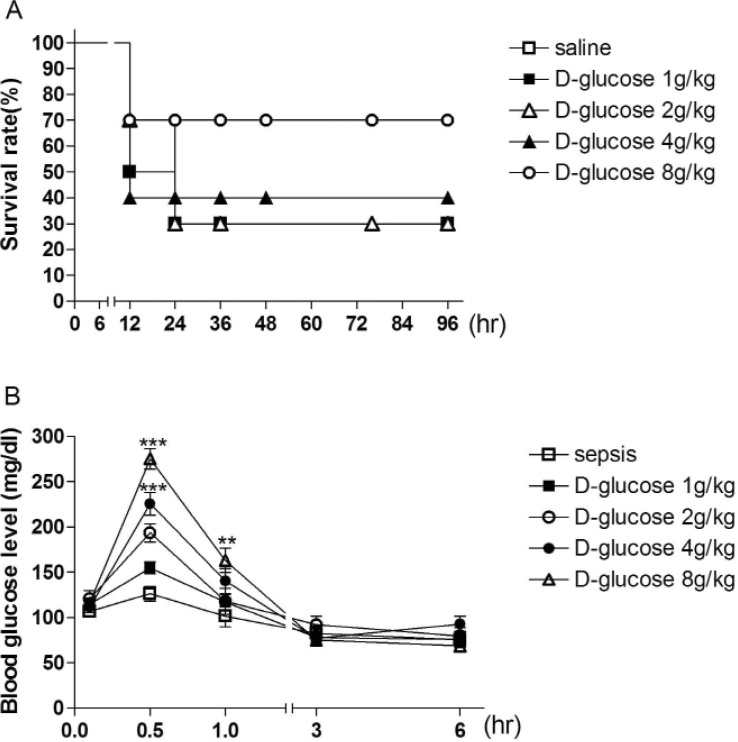
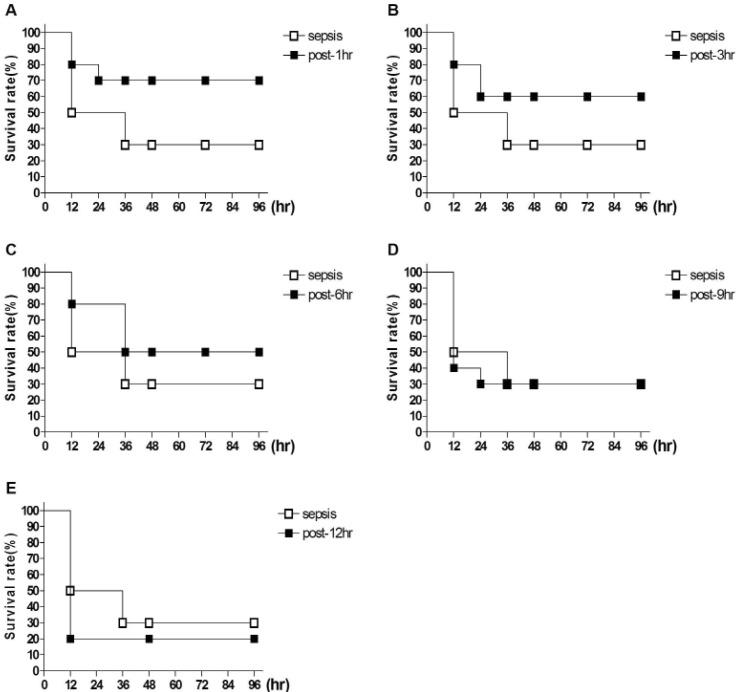
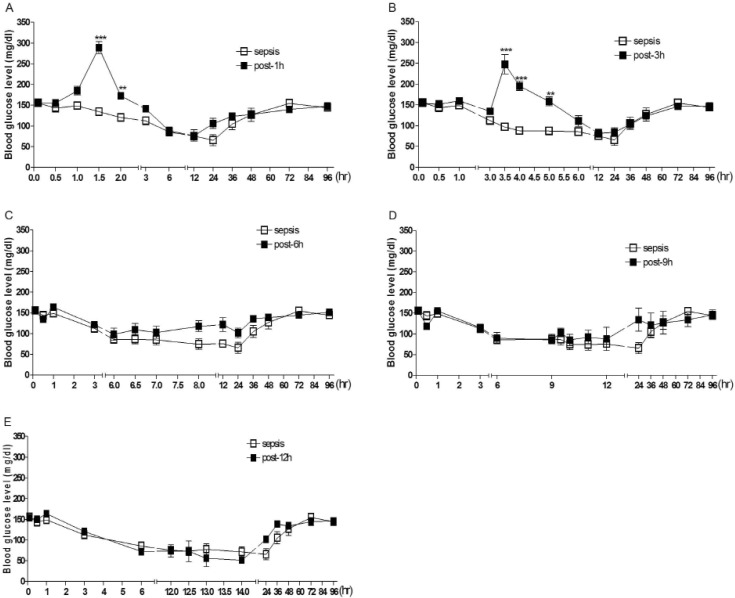
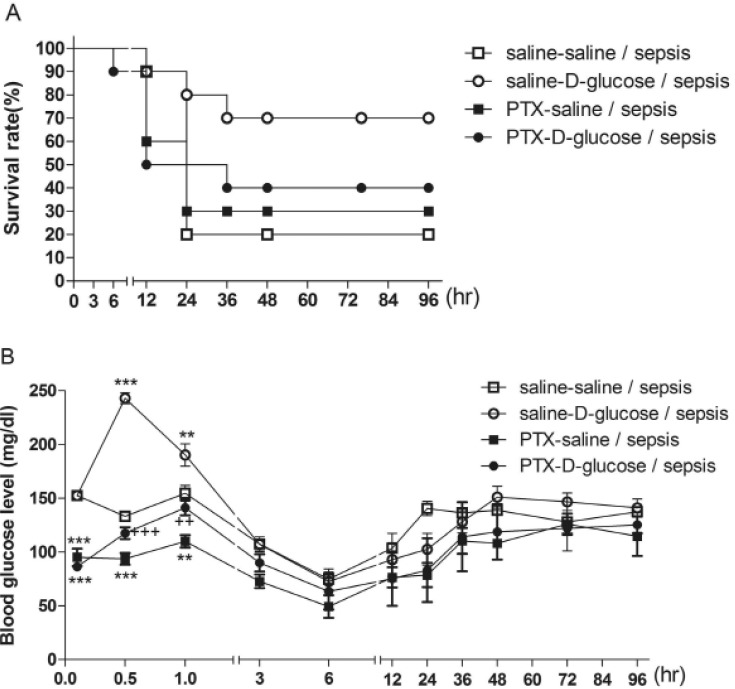
- Sepsis is the life-threatening response to infection which can lead to tissue damage, organ failure, and death. In the current study, the effect of orally administered D-glucose on the mortality and the blood glucose level induced by D-Galactosamine (GaLN)/lipopolysaccharide (LPS)-induced sepsis was examined in ICR mice. After various amounts of D-glucose (from 1 to 8 g/kg) were orally fed, sepsis was induced by injecting intraperitoneally (i.p.) the mixture of GaLN /LPS. Oral pre-treatment with D-glucose dose-dependently increased the blood glucose level and caused a reduction of sepsis-induced mortality. The oral post-treatment with D-glucose (8 g/kg) up to 3 h caused an elevation of the blood glucose level and protected the mortality observed in sepsis model. However, D-glucose post-treated at 6, 9, or 12 h after sepsis induction did not affect the mortality and the blood glucose level induced by sepsis. Furthermore, the intrathecal (i.t.) pretreatment once with pertussis toxin (PTX; 0.1 microg/5 ml) for 6 days caused a reduction of D-glucose-induced protection of mortality and hyperglycemia. Furthermore, once the hypoglycemic state is continued up to 6 h after sepsis initiated, sepsis-induced mortality could not be reversed by D-glucose fed orally. Based on these findings, it is assumed that the hypoglycemic duration between 3 and 6 h after the sepsis induction may be a critical time of period for the survival. D-glucose-induced protective effect against sepsis-induced mortality appears to be mediated via activating PTX-sensitive G-proteins in the spinal cord. Finally, the production of hyperglycemic state may be critical for the survival against the sepsis-induced mortality.
MeSH Terms
Figure
Reference
-
1. Levi M, Schouten M, van der Poll T. Sepsis, coagulation, and antithrombin: old lessons and new insights. Semin Thromb Hemost. 2008; 34:742–746. PMID: 19214912.
Article2. van Westerloo DJ, Giebelen IA, Florquin S, Daalhuisen J, Bruno MJ, de Vos AF, Tracey KJ, van der Poll T. The cholinergic antiinflammatory pathway regulates the host response during septic peritonitis. J Infect Dis. 2005; 191:2138–2148. PMID: 15898001.
Article3. Schouten M, Wiersinga WJ, Levi M, van der Poll T. Inflammation, endothelium, and coagulation in sepsis. J Leukoc Biol. 2008; 83:536–545. PMID: 18032692.
Article4. Dünser MW, Westphal M. Arginine vasopressin in vasodilatory shock: effects on metabolism and beyond. Curr Opin Anaesthesiol. 2008; 21:122–127. PMID: 18443477.
Article5. Lang CH. Sepsis-induced insulin resistance in rats is mediated by a beta-adrenergic mechanism. Am J Physiol. 1992; 263:E703–E711. PMID: 1329550.
Article6. Maitra SR, Wojnar MM, Lang CH. Alterations in tissue glucose uptake during the hyperglycemic and hypoglycemic phases of sepsis. Shock. 2000; 13:379–385. PMID: 10807013.
Article7. Lang CH, Bagby GJ, Spitzer JJ. Glucose kinetics and body temperature after lethal and nonlethal doses of endotoxin. Am J Physiol. 1985; 248:R471–R478. PMID: 3885759.
Article8. Marik PE, Raghavan M. Stress-hyperglycemia, insulin and immunomodulation in sepsis. Intensive Care Med. 2004; 30:748–756. PMID: 14991101.
Article9. Tiruvoipati R, Chiezey B, Lewis D, Ong K, Villanueva E, Haji K, Botha J. Stress hyperglycemia may not be harmful in critically ill patients with sepsis. J Crit Care. 2012; 27:153–158. PMID: 21855283.
Article10. Pulzi Júnior SA, Assunção MS, Mazza BF, Fernandes Hda S, Jackiu M, Freitas FG, Machado FR. Accuracy of different methods for blood glucose measurement in critically ill patients. Sao Paulo Med J. 2009; 127:259–265. PMID: 20169273.
Article11. Szrama J, Smuszkiewicz P, Trojanowska I. Glycemic profile and effectiveness and safety of insulin therapy in septic patients: is the blood glucose level sufficient? Pol Arch Med Wewn. 2009; 119:621–627. PMID: 19847137.
Article12. Ogetii GN, Akech S, Jemutai J, Boga M, Kivaya E, Fegan G, Maitland K. Hypoglycaemia in severe malaria, clinical associations and relationship to quinine dosage. BMC Infect Dis. 2010; 10:334. PMID: 21092224.
Article13. Clemens MG, Chaudry IH, McDermott PH, Baue AE. Regulation of glucose production from lactate in experimental sepsis. Am J Physiol. 1983; 244:R794–R800. PMID: 6407337.
Article14. Miller SI, Wallace RJ Jr, Musher DM, Septimus EJ, Kohl S, Baughn RE. Hypoglycemia as a manifestation of sepsis. Am J Med. 1980; 68:649–654. PMID: 6990758.
Article15. Chang L, Zhao J, Yang J, Zhang Z, Du J, Tang C. Therapeutic effects of ghrelin on endotoxic shock in rats. Eur J Pharmacol. 2003; 473:171–176. PMID: 12892835.
Article16. Chang L, Du JB, Gao LR, Pang YZ, Tang CS. Effect of ghrelin on septic shock in rats. Acta Pharmacol Sin. 2003; 24:45–49. PMID: 12511228.17. Sim YB, Park SH, Kim SS, Lim SM, Jung JS, Suh HW. Activation of spinal α2 adrenergic receptors induces hyperglycemia in mouse though activating sympathetic outflow. Eur J Pharmacol. 2014; 741:316–322. PMID: 25179570.
Article18. Kim CH, Park SH, Sim YB, Sharma N, Kim SS, Lim SM, Jung JS, Suh HW. Effect of pertussis and cholera toxins administered supraspinally on CA3 hippocampal neuronal cell death and the blood glucose level induced by kainic acid in mice. Neurosci Res. 2014; 89:31–36. PMID: 25218563.
Article19. Sim YB, Park SH, Kim SS, Lim SM, Jung JS, Lee JK, Suh HW. Pertussis toxin administered spinally induces a hypoglycemic effect on normal and diabetic mice. Pharmacology. 2014; 94:29–40. PMID: 25171426.
Article20. Suh HW, Sim YB, Choi YS, Song DK, Kim YH. Multiplicative interaction between intrathecally and intracerebroventricularly administered morphine for antinociception in the mouse: effects of spinally and supraspinally injected 3-isobutyl-1-methylxanthine, cholera toxin, and pertussis toxin. Gen Pharmacol. 1995; 26:1597–1602. PMID: 8690252.21. Hylden JL, Wilcox GL. Intrathecal substance P elicits a caudally-directed biting and scratching behavior in mice. Brain Res. 1981; 217:212–215. PMID: 6167328.
Article22. Hylden JL, Wilcox GL. Intrathecal morphine in mice: a new technique. Eur J Pharmacol. 1980; 67:313–316. PMID: 6893963.
Article23. Polito A, Brouland JP, Porcher R, Sonneville R, Siami S, Stevens RD, Guidoux C, Maxime V, de la Grandmaison GL, Chretien FC, Gray F, Annane D, Sharshar T. Hyperglycaemia and apoptosis of microglial cells in human septic shock. Crit Care. 2011; 15:R131. PMID: 21612615.
Article24. Hirasawa H, Oda S, Nakamura M. Blood glucose control in patients with severe sepsis and septic shock. World J Gastroenterol. 2009; 15:4132–4136. PMID: 19725146.
Article25. Thompson BT. Glucose control in sepsis. Clin Chest Med. 2008; 29:713–720. PMID: 18954705.
Article26. Igaki N, Matsuda T, Hirota Y, Kawaguchi T, Tamada F, Goto T. Streptococcal toxic shock syndrome presenting with spontaneous hypoglycemia in a chronic hemodialysis patient: pathophysiological mechanisms. Intern Med. 2003; 42:421–423. PMID: 12793713.
Article27. Kornum JB, Thomsen RW, Riis A, Lervang HH, Schonheyder HC, Sorensen HT. Type 2 diabetes and pneumonia outcomes: a population-based cohort study. Diabetes Care. 2007; 30:2251–2257. PMID: 17595354.28. Benfield T, Jensen JS, Nordestgaard BG. Influence of diabetes and hyperglycaemia on infectious disease hospitalisation and outcome. Diabetologia. 2007; 50:549–554. PMID: 17187246.
Article29. Thomsen RW, Hundborg HH, Lervang HH, Johnsen SP, Schønheyder HC, Sørensen HT. Diabetes mellitus as a risk and prognostic factor for community-acquired bacteremia due to enterobacteria: a 10-year, population-based study among adults. Clin Infect Dis. 2005; 40:628–631. PMID: 15712091.
Article30. Shah BR, Hux JE. Quantifying the risk of infectious diseases for people with diabetes. Diabetes Care. 2003; 26:510–513. PMID: 12547890.
Article31. Vincent JL, Preiser JC, Sprung CL, Moreno R, Sakr Y. Insulin-treated diabetes is not associated with increased mortality in critically ill patients. Crit Care. 2010; 14:R12. PMID: 20132545.
Article32. Stegenga ME, Vincent JL, Vail GM, Xie J, Haney DJ, Williams MD, Bernard GR, van der Poll T. Diabetes does not alter mortality or hemostatic and inflammatory responses in patients with severe sepsis. Crit Care Med. 2010; 38:539–545. PMID: 19851093.
Article33. Tsai CL, Lee CC, Ma MH, Fang CC, Chen SY, Chen WJ, Chang SC, Mehta SH. Impact of diabetes on mortality among patients with community-acquired bacteremia. J Infect. 2007; 55:27–33. PMID: 17349696.
Article34. McAlister FA, Majumdar SR, Blitz S, Rowe BH, Romney J, Marrie TJ. The relation between hyperglycemia and outcomes in 2,471 patients admitted to the hospital with community-acquired pneumonia. Diabetes Care. 2005; 28:810–815. PMID: 15793178.
Article35. Graham BB, Keniston A, Gajic O, Trillo Alvarez CA, Medvedev S, Douglas IS. Diabetes mellitus does not adversely affect outcomes from a critical illness. Crit Care Med. 2010; 38:16–24. PMID: 19789450.
Article36. Esper AM, Moss M, Martin GS. The effect of diabetes mellitus on organ dysfunction with sepsis: an epidemiological study. Crit Care. 2009; 13:R18. PMID: 19216780.
Article37. Thomsen RW, Hundborg HH, Lervang HH, Johnsen SP, Sørensen HT, Schønheyder HC. Diabetes and outcome of community-acquired pneumococcal bacteremia: a 10-year population-based cohort study. Diabetes Care. 2004; 27:70–76. PMID: 14693969.38. Ali NA, O'Brien JM Jr, Blum W, Byrd JC, Klisovic RB, Marcucci G, Phillips G, Marsh CB, Lemeshow S, Grever MR. Hyperglycemia in patients with acute myeloid leukemia is associated with increased hospital mortality. Cancer. 2007; 110:96–10. PMID: 17534900.
Article39. Weiser MA, Cabanillas ME, Konopleva M, Thomas DA, Pierce SA, Escalante CP, Kantarjian HM, O'Brien SM. Relation between the duration of remission and hyperglycemia during induction chemotherapy for acute lymphocytic leukemia with a hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone/methotrexate-cytarabine regimen. Cancer. 2004; 100:1179–1185. PMID: 15022284.
Article40. Norhammar A, Tenerz A, Nilsson G, Hamsten A, Efendíc S, Rydén L, Malmberg K. Glucose metabolism in patients with acute myocardial infarction and no previous diagnosis of diabetes mellitus: a prospective study. Lancet. 2002; 359:2140–2144. PMID: 12090978.
Article41. Van den Berghe G, Wilmer A, Hermans G, Meersseman W, Wouters PJ, Milants I, Van Wijngaerden E, Bobbaers H, Bouillon R. Intensive insulin therapy in the medical ICU. N Engl J Med. 2006; 354:449–461. PMID: 16452557.
Article42. van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001; 345:1359–1367. PMID: 11794168.
Article43. Risso A, Mercuri F, Quagliaro L, Damante G, Ceriello A. Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am J Physiol Endocrinol Metab. 2001; 281:E924–E930. PMID: 11595647.
Article44. Pinto DS, Skolnick AH, Kirtane AJ, Murphy SA, Barron HV, Giugliano RP, Cannon CP, Braunwald E, Gibson CM. TIMI Study Group. U-shaped relationship of blood glucose with adverse outcomes among patients with ST-segment elevation myocardial infarction. J Am Coll Cardiol. 2005; 46:178–180. PMID: 15992655.
Article45. Mendoza A, Kim YN, Chernoff A. Hypoglycemia in hospitalized adult patients without diabetes. Endocr Pract. 2005; 11:91–96. PMID: 15901523.
Article46. Griesdale DE, de Souza RJ, van Dam RM, Heyland DK, Cook DJ, Malhotra A, Dhaliwal R, Henderson WR, Chittock DR, Finfer S, Talmor D. Intensive insulin therapy and mortality among critically ill patients: a meta-analysis including NICE-SUGAR study data. CMAJ. 2009; 180:821–827. PMID: 19318387.
Article47. Komatsu M, McDermott AM, Gillison SL, Sharp GW. Time course of action of pertussis toxin to block the inhibition of stimulated insulin release by norepinephrine. Endocrinology. 1995; 136:1857–1863. PMID: 7720630.
Article48. Garcia Hermida O, Fontela T, Ghiglione M, Uttenthal LO. Effect of pertussis pretreatment on plasma glucose and insulin responses to lithium in rats. Br J Pharmacol. 1991; 103:1309–1312. PMID: 1884093.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The activation of α₂-adrenergic receptor in the spinal cord lowers sepsis-induced mortality
- Treatment of Severe Sepsis-Based on Surviving Sepsis Campaign Guideline
- The mortality of patients with sepsis increases in the first month of a new academic year
- The Effects of Early Enteral Feeding in Extremely Low Birth-Weight Infants
- Comparison of Glucose Concentration of Tracheal Secretions by Measuring Times and Feeding Methods in Enterally Fed Patients