J Bacteriol Virol.
2014 Mar;44(1):59-66. 10.4167/jbv.2014.44.1.59.
Identification of Leptospira species of Korean Isolates using Phylogenetic Analysis of Polymerase Chain Reaction-amplified 16S rDNA and LipL32 Genes
- Affiliations
-
- 1Department of Microbiology, College of Medicine, Konkuk University, Seoul, Korea. wjjang@kku.ac.kr
- 2Institute of Glocal Disease Control, College of Medicine, Konkuk University, Seoul, Korea.
- 3Department of Microbiology, College of Medicine, Hallym University, Chunchon, Kangwon-do, Korea.
- KMID: 2135358
- DOI: http://doi.org/10.4167/jbv.2014.44.1.59
Abstract
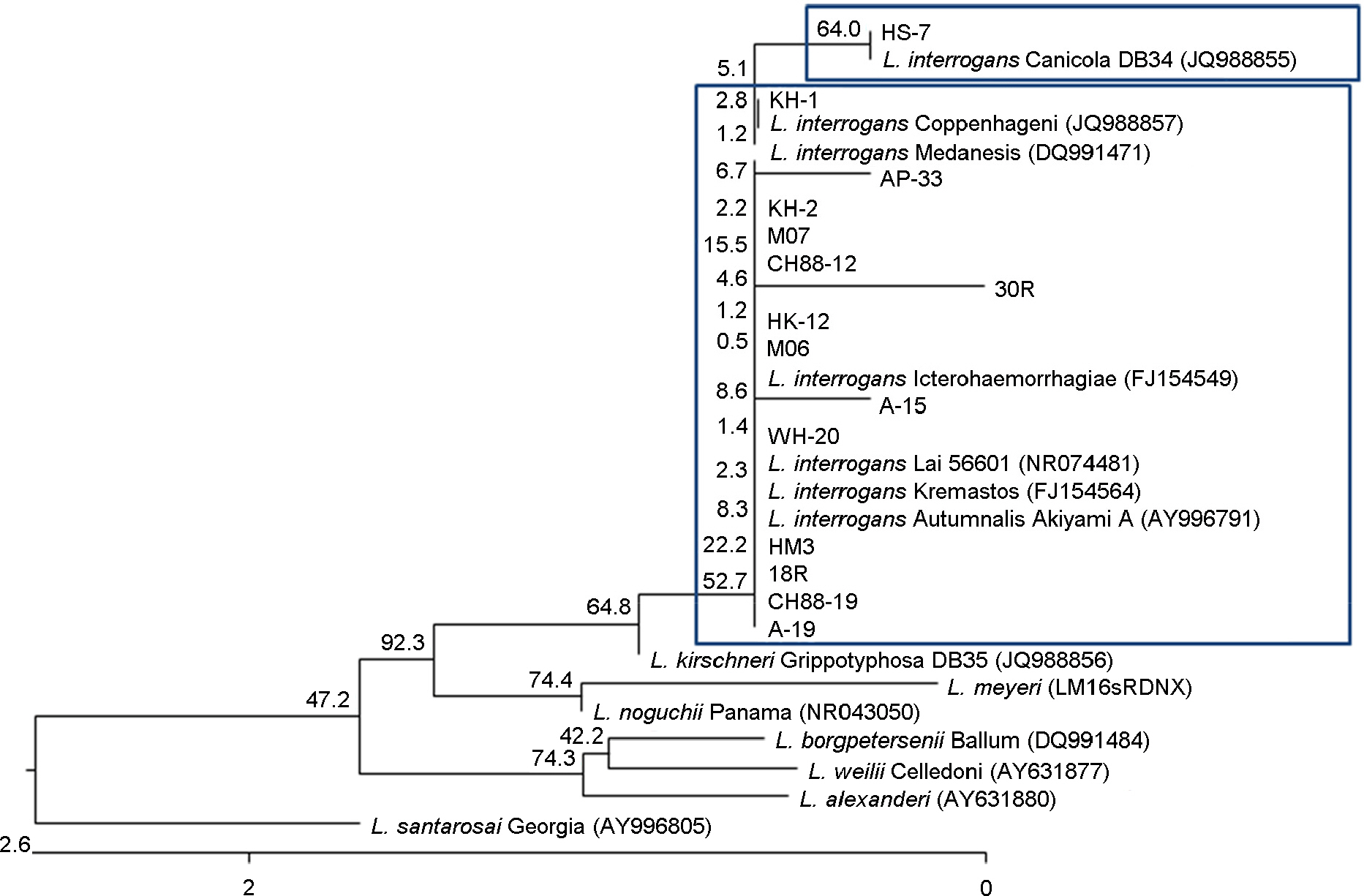
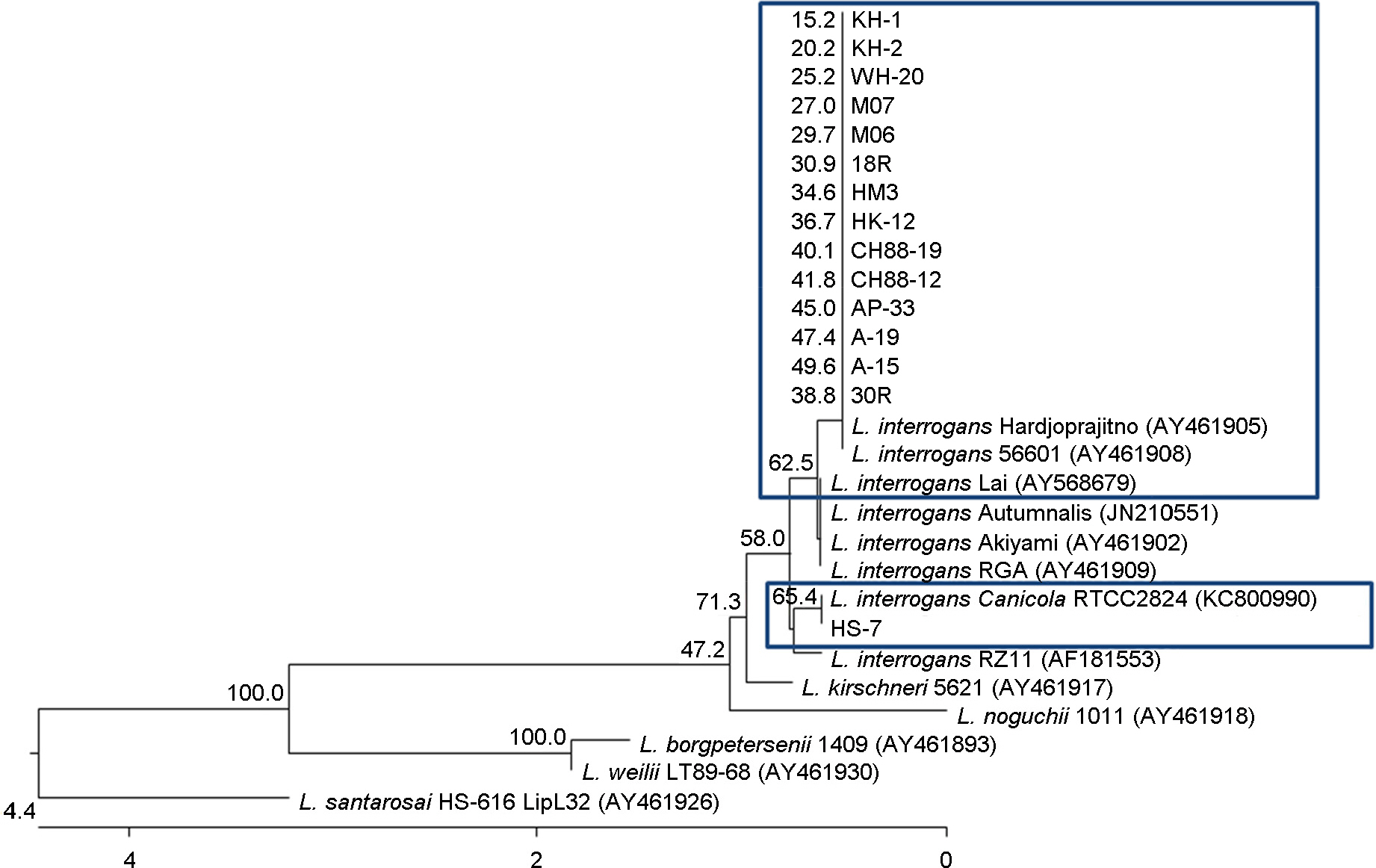
- In this study, we selected only serologically identified 15 Leptosira interrogans isolates in the past and analyzed and identified them by using molecular method. The partial 16S rDNA and LipL32 genes were amplified from the bacteria by polymerase chain reaction (PCR) and sequenced. Sizes of the PCR products were 529 bp and 819 bp respectively and analysis of the nucleotide sequence of 16S rDNA and LipL32 genes showed that 14 out the 15 Leptospira showed 99.4~100% and 99.2~99.9% similarity respectively to those of L. interrogans lai and one isolate named HS-7 showed 100% and 100% similarity to L. interrogans canicola. The phylogenetic tree based on the 16S rDNA and LipL32 genes obtained the study revealed that 14 of the Leptospira composed a cluster distinct to that of L. interrogans lai and HS-7 composed to L. interrogans canicola.
Keyword
MeSH Terms
Figure
Reference
-
1). Yim EK, Kim YW, Lee JS, Chang IA, Baek LJ, Song JW, et al. Detection of leptospiral DNA from field rodents by PCR. J Bacteriol Virol. 2003; 33:177–81.2). Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003; 3:757–71.
Article3). Kim MJ. Leptospirosis in the Republic of Korea: Historical Perspectives, Current Status and Future Challenges. Infect Chemother. 2013; 45:137–44.
Article4). Adler B, de la Peña Moctezuma A. Leptospira and leptospirosis. Vet Microbiol. 2010; 27:287–96.5). Evangelista KV, Coburn J. Leptospira as an emerging pathogen: a review of its biology, pathogenesis and host immune responses. Future Microbiol. 2010; 5:1413–25.6). Chang WH, Kim YW, Oh HB, Cho MK, Kee SH, Kim HJ. Serovar identification and genetic characterization of Leptospira isolates by arbitrarily primed PCR and ribotyping. J Korean Soc Microbiol. 1999; 34:409–21.7). Cerqueira GM, Picardeau M. A century of Leptospira strain typing. Infect Genet Evol. 2009; 9:760–8.8). Levett PN. Leptospirosis. Clin Microbiol Rev. 2001; 14:296–326.
Article9). Oh HB, Chang WH, Cho MK, Seong WK, Park KS. Identification of new serovar yeonchon and hongchon belonging to Leptospira interrogans Icterohaemorrhagiae serogroup. J Kor Soc Microbiol. 1991; 26:253–62.10). Ramadass P, Jarvis BD, Corner RJ, Penny D, Marshall RB. Genetic characterization of pathogenic Leptospira species by DNA hybridization. Int J Syst Bacteriol. 1992; 42:215–9.11). Turk N, Milas Z, Mojcec V, Ruzic-Sabljic E, Staresina V, Stritof Z, et al. Molecular analysis of Leptospira spp. isolated from humans by restriction fragment length polymorphism, real-time PCR and pulsed-field gel electrophoresis. FEMS Microbiol Lett. 2009; 300:174–9.12). Morey RE, Galloway RL, Bragg SL, Steigerwalt AG, Mayer LW, Levett PN. Species-specific identification of Leptospiraceae by 16S rRNA gene sequencing. J Clin Microbiol. 2006; 44:3510–6.13). La Scola B, Bui LT, Baranton G, Khamis A, Raoult D. Partial rpoB gene sequencing for identification of Leptospira species. FEMS Microbiol Lett. 2006; 263:142–7.14). Thaipadungpanit J, Chierakul W, Wuthiekanun V, Limmathurotsakul D, Amornchai P, Boonslip S, et al. Diagnostic accuracy of real-time PCR assays targeting 16S rRNA and lipL32 genes for human leptospirosis in Thailand: a case-control study. PLoS One. 2011; 6:e16236.
Article15). Clarridge JE 3rd. Impact of 16S rRNA gene sequence analysis for identification of bacteria on clinical microbiology and infectious diseases. Clin Microbiol Rev. 2004; 17:840–62.
Article16). Boonyod D, Poovorawan Y, Bhattarakosol P, Chirathaworn C. LipL32, an outer membrane protein of Leptospira, as an antigen in a dipstick assay for diagnosis of leptospirosis. Asian Pac J Allergy Immunol. 2005; 23:133–41.17). Faine S. Guidelines for the control of leptospirosis. WHO;1982.18). Lane DJ, Pace B, Olsen GJ, Stahl DA, Sogin ML, Pace NR. Rapid determination of 16S rRNA sequences for phylogenetic analyses. Proc Natl Acad Sci U S A. 1985; 82:6955–9.19). Cho MK, Kee SH, Song HJ, Kim KH, Song KJ, Baek LJ, et al. Infection rate of Leptospira interrogans in the field rodent, Apodemus agrarius, in Korea. Epidemiol Infect. 1998; 3:685–90.20). Wuyts J, Van de Peer Y, Winkelmans T, De Wachter R. The European database on small subunit rRNA. Nucleic Acids Res. 2002; 30:183–5.21). Haake DA, Suchard MA, Kelley MM, Dundoo M, Alt DP, Zuerner RL. Molecular evolution and mosaicism of leptospiral outer membrane proteins involves horizontal DNA transfer. J Bacteriol. 2004; 186:2818–28.
Article22). Haake DA, Chao G, Zuerner RL, Barnett JK, Barnett D, Mazel M, et al. The leptospiral major outer membrane protein LipL32 is a lipoprotein expressed during mammalian infection. Infect Immun. 2000; 68:2276–85.
Article23). Shang ES, Summers TA, Haake DA. Molecular cloning and sequence analysis of the gene encoding LipL41, a surface-exposed lipoprotein of pathogenic Leptospira species. Infect Immun. 1996; 64:2322–30.24). Perez J, Goarant C. Rapid Leptospira identification by direct sequencing of the diagnostic PCR products in New Caledonia. BMC Microbiol. 2010; 10:325.25). Mgode GF, Machang'u RS, Goris MG, Engelbert M, Sondij S, Hartskeerl RA. New Leptospira serovar sokoine of serogroup Icterohaemorrhagiae from cattle in Tanzania. Int J Syst Evol Microbiol. 2006; 56:593–7.26). Yasuda PH, Steigerwalt AG, Sulzer KR, Kaufmann AF, Rogers F, Brenner DJ. Deoxyribonucleic acid relatedness between serogroups and serovars in the family Leptospiraceae with proposals for seven new Leptospira species. Int J Syst Evol Microbiol. 1987; 37:407–15.27). Chang WH, Kee SH, Park KH, Kim SY, Kim IS, Choi MS. Antigenic analysis of Leptospira interrogans isolated in Korea using monoclonal antibodies and cross-agglutinin absorption test. J Kor Soc Microbiol. 1989; 24:165–73.28). Park KH, Chang WH. Serovar identification of leptospiral strain HS-7 isolated in Korea by monoclonal antibodies. J Kor Soc Microbiol. 1988; 23:293–305.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Molecular Evolution and Identification of Yersinia Species by 16S rDNA Analysis
- Molecular Identification of Anginosus Group Streptococci Isolated from Korean Oral Cavities
- Genomic species identification of Acinetobacter calcoaceticus - Acinetobacter baumannii complex strains by amplified ribosomal DNA restriction analysis (ARDRA)
- Molecular Discrimination of Mitis Group Streptococci Isolated from Koreans using RpoB Nucleotide Sequences
- Comparison of the Utility of dnaJ and 16S rDNA Sequences for Identification of Clinical Isolates of Vibrio Species