Chonnam Med J.
2010 Dec;46(3):148-155. 10.4068/cmj.2010.46.3.148.
Antioxidant Activity of Rubus coreanus Fruit Extract: In Comparison to Green Tea Extract
- Affiliations
-
- 1Department of Biochemistry, Medical School and The Research Institute of Medical Sciences, Chonnam National University, Gwangju, Korea. syyang@jnu.ac.kr
- KMID: 2116768
- DOI: http://doi.org/10.4068/cmj.2010.46.3.148
Abstract
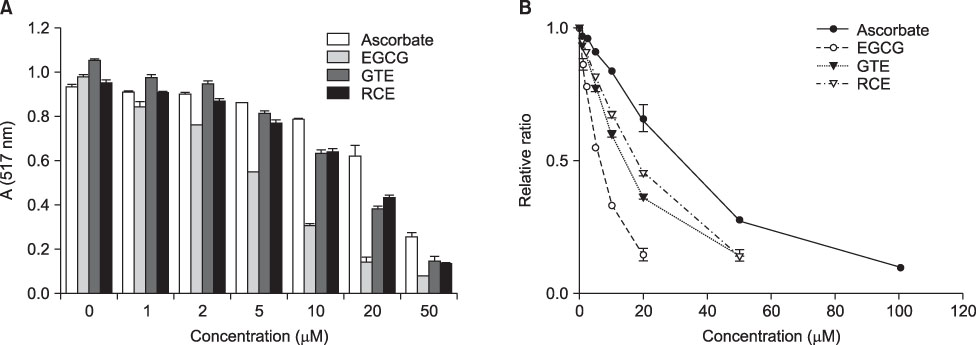
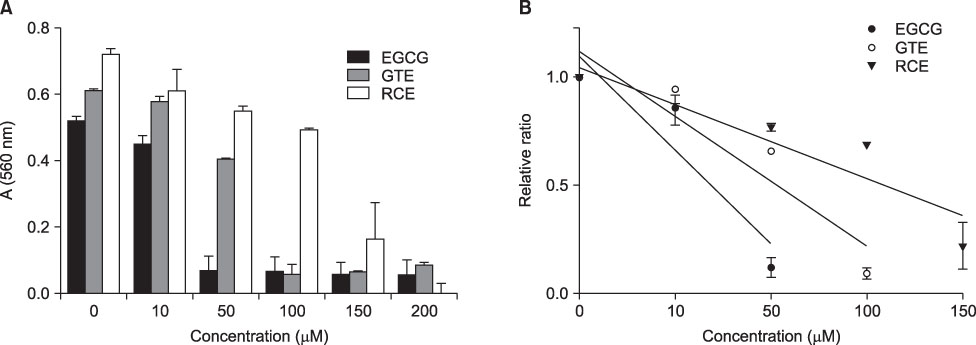
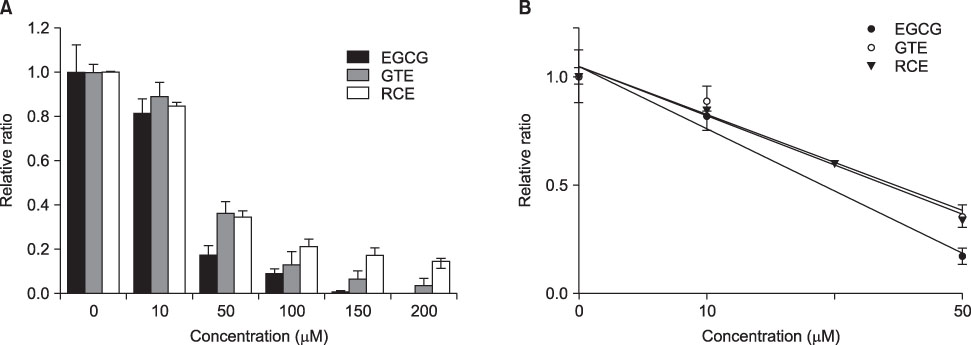
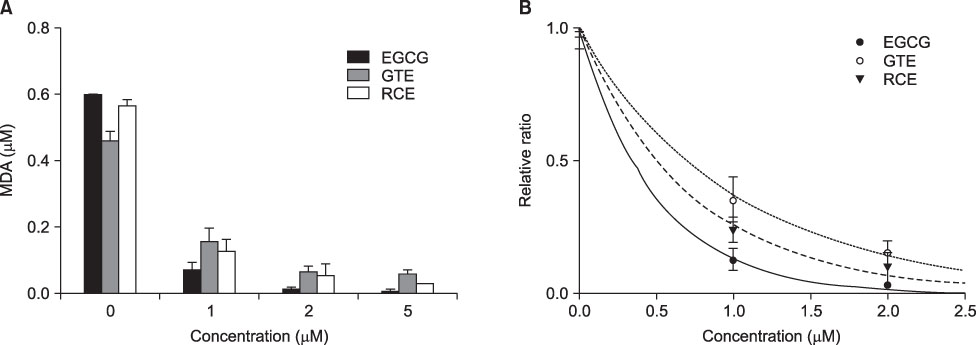
- This study aimed to evaluate the antioxidant activity of Korean raspberry (Rubus coreanus Miq.) fruits, which are grown widely in the southwest area of Korea. Freeze-dried Rubus coreanus (RC) fruits and green tea (GT) were used to obtain water extracts (RCE and GTE, respectively). The total phenolic contents of the dried RC and GT were 126micromol CE (catechin equivalent)/g and 360micromol CE/g, respectively. The free radical scavenging activity of RCE and GTE was expressed as SC50 (50% scavenging capacity) and compared. The scavenging activity of RC toward DPPH, superoxide, and hydroxyl radicals was lower than that of GT. The inhibitory activity of RCE and GTE on AAPH-induced lipid peroxidation was also analyzed. In contrast with the free radical scavenging activities, the lipid peroxidation-inhibiting activity of RC was superior to that of GT. These results suggest that RCE contains effective scavengers of peroxyl radicals. In summary, RC contains less phenolics than does GT. Although RC has lower activity for scavenging DPPH, superoxide, and hydroxyl radicals than does GT, its activity to inhibit peroxyl radical-lipid peroxidation was higher. These results suggest that RC could be used as a valuable natural source of antioxidants.
Keyword
MeSH Terms
Figure
Reference
-
1. Ames BM, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci USA. 1993. 90:7915–7922.
Article2. Dragsted LO, Strube M, Larsen JC. Cancer-protective factors in fruits and vegetables: biochemical and biological background. Pharmacol Toxicol. 1993. 72:Suppl 1. 116–135.
Article3. Cao G, Sofic E, Prior RL. Antioxidant capacity of tea and common vegetables. J Agric Food Chem. 1996. 44:3426–3431.
Article4. Velioglu YS, Mazza G, Gao L, Oomah BD. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J Agric Food Chem. 1998. 46:4113–4117.
Article5. Heber D. Vegetables, fruits and phytoestrogens in the prevention of diseases. J Postgrad Med. 2004. 50:145–149.6. Ascherio A, Rimm EB, Giovannucci EL, Colditz GA, Rosner B, Willett WC, et al. A prospective study of nutritional factors and hypertension among US men. Circulation. 1992. 86:1475–1484.
Article7. Sacks FM, Kass EH. Low blood pressure in vegetarians: effects of specific foods and nutrients. Am J Clin Nutr. 1988. 48:3 Suppl. 795–800.
Article8. Cao G, Sofic E, Prior RL. Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radic Biol Med. 1997. 22:749–760.
Article9. Havsteen B. Flavonoids, a class of natural products of high pharmacological potency. Biochem Pharmacol. 1983. 32:1141–1148.
Article10. Middleton E Jr, Kandaswami C. Effects of flavonoids on immune and inflammatory cell functions. Biochem Pharmacol. 1992. 43:1167–1179.
Article11. Das DK. Naturally occurring flavonoids: structure, chemistry, and highperformance liquid chromatography methods for separation and characterization. Methods Enzymol. 1994. 234:410–420.12. Haramaki N, Aggarwal S, Kawabata T, Droy-Lefaix MT, Packer L. Effects of natural antioxidant ginkgo biloba extract (EGB 761) on myocardial ischemia-reperfusion injury. Free Radic Biol Med. 1994. 16:789–794.
Article13. Rump AF, Schüssler M, Acar D, Cordes A, Theisohn M, Rösen R, et al. Functional and antiischemic effects of luteolin-7-glucoside in isolated rabbit hearts. Gen Pharmacol. 1994. 25:1137–1142.
Article14. Wu TW, Fung KP, Zeng LH, Wu J, Hempel A, Grey AA, et al. Molecular properties and myocardial salvage effects of morin hydrate. Biochem Pharmacol. 1995. 49:537–543.
Article15. Oyama Y, Chikahisa L, Ueha T, Kanemaru K, Noda K. Ginkgo biloba extract protects brain neurons against oxidative stress induced by hydrogen peroxide. Brain Res. 1996. 712:349–352.
Article16. Choi EM, Hwang JK. Effect of some medicinal plants on plasma antioxidant system and lipid levels in rats. Phytother Res. 2005. 19:382–386.
Article17. Choi HS, Kim MK, Park HS, Kim YS, Shin DH. Alcoholic fermentation of Bokbunja (Rubus coreanus Miq.) wine. Korean J Food Sci Technol. 2006. 38:543–547.18. Wang SY, Lin HS. Antioxidant activity in fruits and leaves of blackberry, raspberry, and strawberry varies with cultivar and developmental stage. J Agric Food Chem. 2000. 48:140–146.
Article19. Wada L, Ou B. Antioxidant activity and phenolic content of Oregon caneberries. J Agric Food Chem. 2002. 50:3495–3500.
Article20. Choi J, Lee KT, Ha J, Yun SY, Ko CD, Jung HJ, et al. Antinociceptive and antiinflammatory effects of Niga-ichigoside F1 and 23-hydroxytormentic acid obtained from Rubus coreanus. Biol Pharm Bull. 2003. 26:1436–1441.
Article21. Park JH, Oh SM, Lim SS, Lee YS, Shin HK, Oh YS, et al. Induction of heme oxygenase-1 mediates the anti-inflammatory effects of the ethanol extract of Rubus coreanus in murine macrophages. Biochem Biophys Res Commun. 2006. 351:146–152.
Article22. Shin TY, Kim SH, Lee ES, Eom DO, Kim HM. Action of Rubus coreanus extract on systemic and local anaphylaxis. Phytother Res. 2002. 16:508–513.23. Lee KH, Choi EM. Rubus coreanus Miq. extract promotes osteoblast differentiation and inhibits bone-resorbing mediators in MC3T3-E1 cells. Am J Chin Med. 2006. 34:643–654.
Article24. Nam JH, Jung HJ, Choi J, Lee KT, Park HJ. The anti-gastropathic and anti-rheumatic effect of niga-ichigoside F1 and 23-hydroxytormentic acid isolated from the unripe fruits of Rubus coreanus in a rat model. Biol Pharm Bull. 2006. 29:967–970.
Article25. Singleton VL, Orthofer R, Lamuela-Raventos RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999. 299:152–178.26. Motoyama T, Miki M, Mino M, Takahashi M, Niki E. Synergistic inhibition of oxidation in dispersed phosphatidylcholine liposomes by a combination of vitamin E and cysteine. Arch Biochem Biophys. 1989. 270:655–661.
Article27. Ames BN. Assay of inorganic phosphate, total phosphate and phosphatases. Methods Enzymol. 1966. 8:115–118.28. Rangkadilok N, Sitthimonchai S, Worasuttayangkurn L, Mahidol C, Ruchirawat M, Satayavivad J. Evaluation of free radical scavenging and antityrosinase activities of standardized longan fruit extract. Food Chem Toxicol. 2007. 45:328–336.
Article29. Lee MW, Ahn BW, Lee KY, Park BJ, Yang SY, Sull JE. Production of superoxide radical by reaction of phenazine methosulfate with erythrocyte membranes. Chonnam Med J. 1984. 21:389–393.30. Asplund KU, Jansson PJ, Lindqvist C, Nordström T. Measurement of ascorbic acid (vitamin C) induced hydroxyl radical generation in household drinking water. Free Radic Res. 2002. 36:1271–1276.
Article31. Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978. 52:302–310.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment for Two Cases of Acne Vulgaris with Rubus Coreanus Miquel Extract
- Growth inhibition effect of Rubus coreanus Miquel on Candida albicans
- Antimicrobial effects of green tea extract-containing dentifrice
- Comparison of whitening effect of Rubus coreanus fruit according to maturity
- Effects of Green Tea on Weight Gain, Plasma and Liver Lipids and Lipid Peroxidation in Pair Fed Rats