J Vet Sci.
2014 Dec;15(4):495-502. 10.4142/jvs.2014.15.4.495.
M gene analysis of canine coronavirus strains detected in Korea
- Affiliations
-
- 1Department of Veterinary Internal Medicine, College of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon 200-701, Korea. kimdoo@kangwon.ac.kr
- 2Department of Veterinary Internal Medicine, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea.
- KMID: 2070235
- DOI: http://doi.org/10.4142/jvs.2014.15.4.495
Abstract
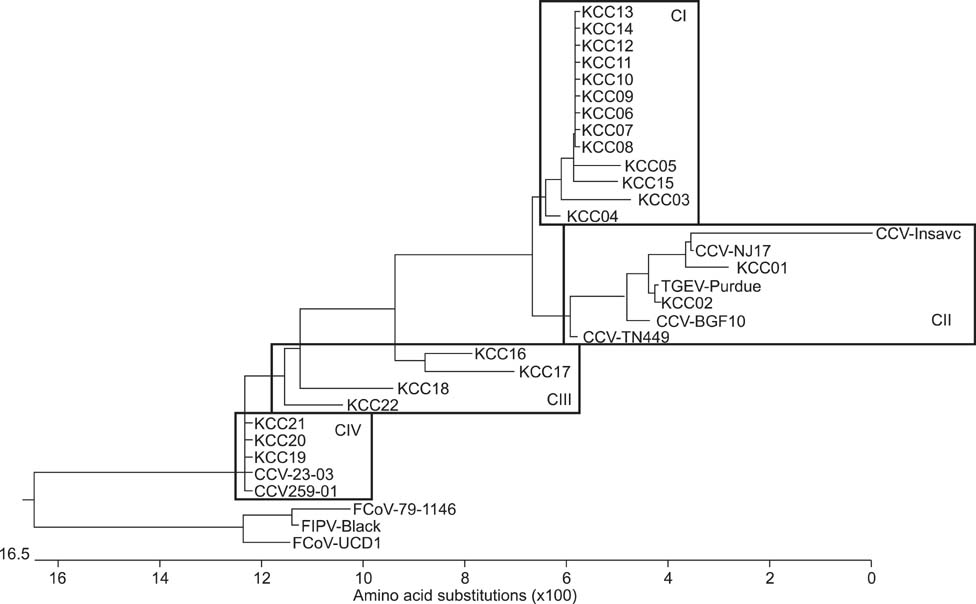
- The purpose of this study was to investigate the genetic features of canine coronavirus (CCV) strains detected in Korea. M gene sequences obtained for isolates from 22 dogs with enteritis over a 5-year period were evaluated. Sequence comparison revealed that the 22 Korean CCV strains had an 87.2 to 100% nucleotide homology. Comparing to the typical reference CCV strains (type II), the nucleotide sequence of Korean strains had homology ranged from 86.3% to 98.3% (89.1% to 99.2% for the amino acid sequence) and 87.7% to 97.8% (92.4% to 100% for the amino acid sequence) when compared to FCoV-like CCV strains (type I). Three amino acid variations in the M gene were characteristic for the Korean CCV strains. Phylogenetic analysis demonstrated that the 22 Korean CCV strains belonged to four typical CCV clusters (i.e., a unique Korean CCV cluster, a type II and transmissible gastroenteritis virus cluster, an intermediate cluster between type I and II, and a type I cluster). This study was the first to identify genetic differences of the M gene from Korean CCV strains and provided a platform for molecular identification of different Korean CCV strains.
Keyword
MeSH Terms
-
Amino Acid Sequence
Animals
Coronavirus Infections/epidemiology/*veterinary/virology
Coronavirus, Canine/*isolation & purification
Dog Diseases/*epidemiology/virology
Dogs
Female
Male
Molecular Sequence Data
Phylogeny
Polymerase Chain Reaction/veterinary
Republic of Korea/epidemiology
Reverse Transcriptase Polymerase Chain Reaction/veterinary
Viral Matrix Proteins/*genetics/metabolism
Viral Matrix Proteins
Figure
Reference
-
1. Addie DD, Schaap IAT, Nicolson L, Jarrett O. Persistence and transmission of natural type I feline coronavirus infection. J Gen Virol. 2003; 84:2735–2744.
Article2. An DJ, Jeong W, Yoon SH, Jeoung HY, Kim HJ, Park BK. Genetic analysis of canine group 2 coronavirus in Korean dogs. Vet Microbiol. 2010; 141:46–52.
Article3. Barlough JE, Stoddart CA, Sorresso GP, Jacobson RH, Scott FW. Experimental inoculation of cats with canine coronavirus and subsequent challenge with feline infectious peritonitis virus. Lab Anim Sci. 1984; 34:592–597.4. Benetka V, Kolodziejek J, Walk K, Rennhofer M, Möstl K. M gene analysis of atypical strains of feline and canine coronavirus circulating in an Austrian animal shelter. Vet Rec. 2006; 159:170–175.
Article5. Benetka V, Kübber-Heiss A, Kolodziejek J, Nowotny N, Hofmann-Parisot M, Möstl K. Prevalence of feline coronavirus types I and II in cats with histopathologically verified feline infectious peritonitis. Vet Microbiol. 2004; 99:31–42.
Article6. Dolja VV, Carrington JC. Evolution of positive-strand RNA viruses. Semin Virol. 1992; 3:315–326.7. East ML, Moestl K, Benetka V, Pitra C, Höner OP, Wachter B, Hofer H. Coronavirus infection of spotted hyenas in the Serengeti ecosystem. Vet Microbiol. 2004; 102:1–9.
Article8. Enjuanes L, Spaan W, Snijder E, Cavanach D. Nidovirales. In : van Regenmortel MHV, Fauquet CM, Bishop DHL, Carstens EB, Estes MK, Lemon SM, Maniloff J, Mayo MA, McGeoch DJ, Pringle CR, Wickner RB, editors. Virus Taxonomy. Seventh Report of the International Committee on Taxonomy of Viruses. San Diego: Academic Press;2000. p. 835–849.9. Escutenaire S, Isaksson M, Renström LHM, Klingeborn B, Buonavoglia C, Berg M, Belák S, Thorén P. Characterization of divergent and atypical canine coronaviruses from Sweden. Arch Virol. 2007; 152:1507–1514.
Article10. Herrewegh AAPM, Smeenk I, Horzinek MC, Rottier PJM, de Groot RJ. Feline coronavirus type II strains 79-1683 and 79-1146 originate from a double recombination between feline coronavirus type I and canine coronavirus. J Virol. 1998; 72:4508–4514.
Article11. Hohdatsu T, Okada S, Ishizuka Y, Yamada H, Koyama H. The prevalence of types I and II feline coronavirus infections in cats. J Vet Med Sci. 1992; 54:557–562.
Article12. Horsburgh BC, Brierley I, Brown TDK. Analysis of a 9·6kb sequence from the 3' end of canine coronavirus genomic RNA. J Gen Virol. 1992; 73:2849–2862.
Article13. Horzinek MC, Lutz H, Pedersen NC. Antigenic relationships among homologous structural polypeptides of porcine, feline, and canine coronaviruses. Infect Immun. 1982; 37:1148–1155.
Article14. Jarvis TC, Kirkegaard K. The polymerase in its labyrinth: mechanisms and implications of RNA recombination. Trends Genet. 1991; 7:186–191.
Article15. Ma G, Wang Y, Lu C. Molecular characterization of the 9.36 kb C-terminal region of canine coronavirus 1-71 strain. Virus Genes. 2008; 36:491–497.
Article16. McArdle F, Bennett M, Gaskell RM, Tennant B, Kelly DF, Gaskell CJ. Induction and enhancement of feline infectious peritonitis by canine coronavirus. Am J Vet Res. 1992; 53:1500–1506.17. Motokawa K, Hohdatsu T, Hashimoto H, Koyama H. Comparison of the amino acid sequence and phylogenetic analysis of the peplomer, integral membrane and nucleocapsid proteins of feline, canine and porcine coronaviruses. Microbiol Immunol. 1996; 40:425–433.
Article18. Naylor MJ, Harrison GA, Monckton RP, McOrist S, Lehrbach PR, Deane EM. Identification of canine coronavirus strains from feces by S gene nested PCR and molecular characterization of a new Australian isolate. J Clin Microbiol. 2001; 39:1036–1041.
Article19. Naylor MJ, Walia CS, McOrist S, Lehrbach PR, Deane EM, Harrison GA. Molecular characterization confirms the presence of a divergent strain of canine coronavirus (UWSMN-1) in Australia. J Clin Microbiol. 2002; 40:3518–3522.
Article20. Pedersen NC, Ward J, Mengeling WL. Antigenic relationship of the feline infections peritonitis virus to coronaviruses of other species. Arch Virol. 1978; 58:45–53.
Article21. Pratelli A. Genetic evolution of canine coronavirus and recent advances in prophylaxis. Vet Res. 2006; 37:191–200.
Article22. Pratelli A, Buonavoglia D, Martella V, Tempesta M, Lavazza A, Buonavoglia C. Diagnosis of canine coronavirus infection using nested-PCR. J Virol Methods. 2000; 84:91–94.
Article23. Pratelli A, Elia G, Decaro N, Tola S, Tinelli A, Martella V, Rocca S, Tempesta M, Buonavoglia C. Cloning and expression of two fragments of the S gene of canine coronavirus type I. J Virol Methods. 2004; 117:61–65.
Article24. Pratelli A, Elia G, Martella V, Palmieri A, Cirone F, Tinelli A, Corrente M, Buonavoglia C. Prevalence of canine coronavirus antibodies by an enzyme-linked immunosorbent assay in dogs in the south of Italy. J Virol Methods. 2002; 102:67–71.
Article25. Pratelli A, Elia G, Martella V, Tinelli A, Decaro N, Marsilio F, Buonavoglia D, Tempesta M, Buonavoglia C. M gene evolution of canine coronavirus in naturally infected dogs. Vet Rec. 2002; 151:758–761.26. Pratelli A, Martella V, Decaro N, Tinelli A, Camero M, Cirone F, Elia G, Cavalli A, Corrente M, Greco G, Buonavoglia D, Gentile M, Tempesta M, Buonavoglia C. Genetic diversity of a canine coronavirus detected in pups with diarrhoea in Italy. J Virol Methods. 2003; 110:9–17.
Article27. Pratelli A, Martella V, Pistello M, Elia G, Decaro N, Buonavoglia D, Camero M, Tempesta M, Buonavoglia C. Identification of coronaviruses in dogs that segregate separately from the canine coronavirus genotype. J Virol Methods. 2003; 107:213–222.
Article28. Pratelli A, Tempesta M, Greco G, Martella V, Buonavoglia C. Development of a nested PCR assay for the detection of canine coronavirus. J Virol Methods. 1999; 80:11–15.
Article29. Sanchez-Morgado JM, Poynter S, Morris TH. Molecular characterization of a virulent canine coronavirus BGF strain. Virus Res. 2004; 104:27–31.
Article30. Siddell SG. The coronaviridae: an introduction. In : Siddell SG, editor. The coronaviridae. New York: Plenum Press;1995. p. 1–10.31. Stoddart CA, Barlough JE, Baldwin CA, Scott FW. Attempted immunisation of cats against feline infectious peritonitis using canine coronavirus. Res Vet Sci. 1988; 45:383–388.
Article32. Tresnan DB, Levis R, Holmes KV. Feline aminopeptidase N serves as a receptor for feline, canine, porcine, and human coronaviruses in serogroup I. J Virol. 1996; 70:8669–8674.
Article33. Vennema H, de Groot RJ, Harbour DA, Horzinek MC, Spaan WJ. Primary structure of the membrane and nucleocapsid protein genes of feline infectious peritonitis virus and immunogenicity of recombinant vaccinia viruses in kittens. Virology. 1991; 181:327–335.
Article34. Wang Y, Ma G, Lu C, Wen H. Detection of canine coronaviruses genotype I and II in raised Canidae animals in China. Berl Munch Tierarztl Wochenschr. 2006; 119:35–39.35. Zhang X, Hasoksuz M, Spiro D, Halpin R, Wang S, Stollar S, Janies D, Hadya N, Tang Y, Ghedin E, Saif L. Complete genomic sequences, a key residue in the spike protein and deletions in nonstructural protein 3b of US strains of the virulent and attenuated coronaviruses, transmissible gastroenteritis virus and porcine respiratory coronavirus. Virology. 2007; 358:424–435.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of Commercial Immunochromatographic Test Kits for the Detection of Canine Distemper Virus
- Detection of canine respiratory coronavirus from dogs with respiratory disease
- Clinical evaluation of a rapid diagnostic test kit for detection of canine coronavirus
- Brain invasion of bovine coronavirus: Virology molecular analysis of bovine coronavirus infection in calves with severe pneumonia and neurological signs
- Establishment of multiplex RT-PCR for differentiation between rabies virus with and that without mutation at position 333 of glycoprotein