Yonsei Med J.
2015 Mar;56(2):510-518. 10.3349/ymj.2015.56.2.510.
Toxicity of Tomotherapy-Based Simultaneous Integrated Boost in Whole-Pelvis Radiation for Prostate Cancer
- Affiliations
-
- 1Department of Radiation Oncology, Yonsei University Wonju College of Medicine, Wonju, Korea.
- 2Department of Radiation Oncology, Yonsei University College of Medicine, Seoul, Korea. cglee1023@yuhs.ac
- KMID: 2070032
- DOI: http://doi.org/10.3349/ymj.2015.56.2.510
Abstract
- PURPOSE
The validity of tomotherapy-based simultaneous integrated boost (TOMOSIB) was assessed in terms of acute intestinal/urinary toxicity by comparing with 3-dimensional conformal radiotherapy (3DCRT) in cases of whole-pelvis radiation therapy (WPRT) for prostate cancer.
MATERIALS AND METHODS
Thirty-eight consecutive patients who underwent curative WPRT were retrospectively reviewed. Twenty six (68.4%) received 3DCRT and the others (31.6%) were treated with TOMOSIB. A local boost to the prostate circumferential area was added to WPRT sequentially for 3DCRT and concomitantly for TOMOSIB. The total median prostate or prostatic bed dose was 64.8 Gy including median 45.0 Gy of WPRT. Acute toxicities were assessed according to RTOG criteria.
RESULTS
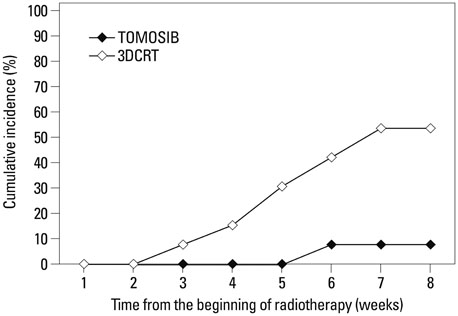
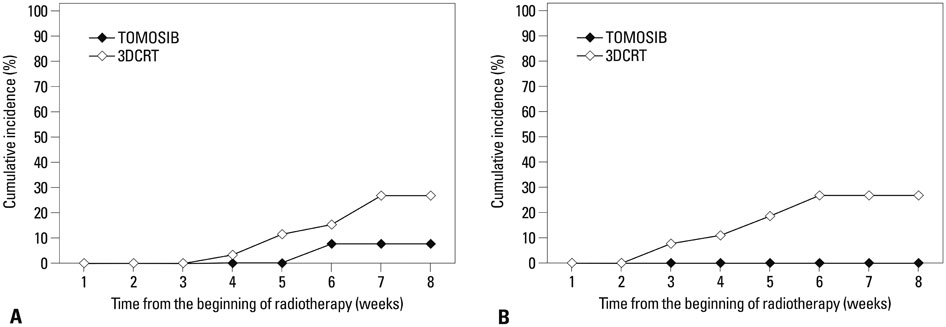
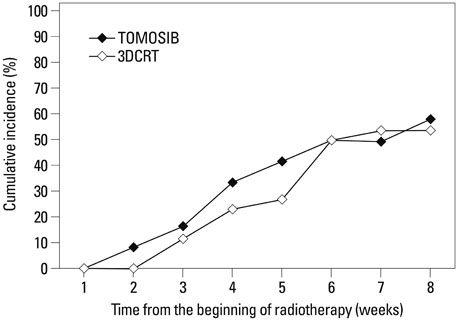
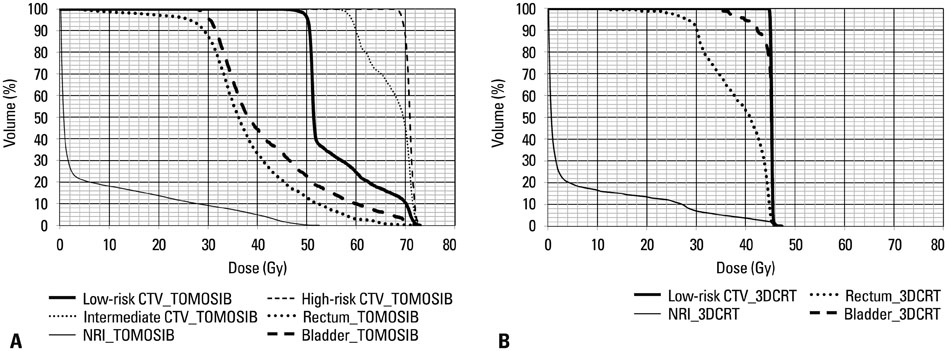
Overall intestinal toxicity was lower in TOMOSIB group than 3DCRT group (p=0.008). When it was divided into rectum and non-rectum intestine (NRI), TOMOSIB showed borderline superiority only in NRI toxicity (p=0.047). For the urinary toxicity, there was no significant difference between two groups (p=0.796). On dosimetric analysis for the rectum and bladder, dose delivered to 80% (p<0.001) and volume receiving 25-40 Gy (p<0.001) were remarkably higher in 3DCRT. For the NRI, only maximum dose showed significant results between two groups (p<0.001).
CONCLUSION
Intestinal toxicity should be verified with more detailed anatomic categorization such as rectum and NRI. TOMOSIB could not reduce urinary toxicity because of inevitably high dose exposure to the prostatic urethra. Current dosimetry system did not properly reflect intestinal/urinary toxicity, and suitable dosimetric guidelines are needed in TOMOSIB.
MeSH Terms
-
Adenocarcinoma/pathology/*radiotherapy
Aged
Humans
Intestine, Small/*radiation effects
Male
Middle Aged
Pelvis/*radiation effects
Prostatic Neoplasms/pathology/*radiotherapy
Radiation Injuries
Radiotherapy Dosage
Radiotherapy, Intensity-Modulated/*adverse effects/methods
Rectum/radiation effects
Retrospective Studies
Urinary Bladder/*radiation effects
Figure
Reference
-
1. Di Muzio N, Fiorino C, Cozzarini C, Alongi F, Broggi S, Mangili P, et al. Phase I-II study of hypofractionated simultaneous integrated boost with tomotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2009; 74:392–398.
Article2. Arcangeli S, Saracino B, Petrongari MG, Gomellini S, Marzi S, Landoni V, et al. Analysis of toxicity in patients with high risk prostate cancer treated with intensity-modulated pelvic radiation therapy and simultaneous integrated dose escalation to prostate area. Radiother Oncol. 2007; 84:148–155.
Article3. Muren LP, Wasbø E, Helle SI, Hysing LB, Karlsdottir A, Odland OH, et al. Intensity-modulated radiotherapy of pelvic lymph nodes in locally advanced prostate cancer: planning procedures and early experiences. Int J Radiat Oncol Biol Phys. 2008; 71:1034–1041.
Article4. Cozzarini C, Fiorino C, Di Muzio N, Alongi F, Broggi S, Cattaneo M, et al. Significant reduction of acute toxicity following pelvic irradiation with helical tomotherapy in patients with localized prostate cancer. Radiother Oncol. 2007; 84:164–170.
Article5. Chang JS, Yoon HI, Cha HJ, Chung Y, Cho Y, Keum KC, et al. Bladder filling variations during concurrent chemotherapy and pelvic radiotherapy in rectal cancer patients: early experience of bladder volume assessment using ultrasound scanner. Radiat Oncol J. 2013; 31:41–47.
Article6. Ruchala KJ, Olivera GH, Schloesser EA, Mackie TR. Megavoltage CT on a tomotherapy system. Phys Med Biol. 1999; 44:2597–2621.
Article7. Shepard DM, Olivera GH, Reckwerdt PJ, Mackie TR. Iterative approaches to dose optimization in tomotherapy. Phys Med Biol. 2000; 45:69–90.
Article8. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995; 31:1341–1346.
Article9. Roach M 3rd, DeSilvio M, Lawton C, Uhl V, Machtay M, Seider MJ, et al. Phase III trial comparing whole-pelvic versus prostate-only radiotherapy and neoadjuvant versus adjuvant combined androgen suppression: Radiation Therapy Oncology Group 9413. J Clin Oncol. 2003; 21:1904–1911.
Article10. Seaward SA, Weinberg V, Lewis P, Leigh B, Phillips TL, Roach M 3rd. Improved freedom from PSA failure with whole pelvic irradiation for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 1998; 42:1055–1062.
Article11. Seaward SA, Weinberg V, Lewis P, Leigh B, Phillips TL, Roach M 3rd. Identification of a high-risk clinically localized prostate cancer subgroup receiving maximum benefit from whole-pelvic irradiation. Cancer J Sci Am. 1998; 4:370–377.12. Roach M 3rd. Re: The use of prostate specific antigen, clinical stage and Gleason score to predict pathological stage in men with localized prostate cancer. J Urol. 1993; 150:1923–1924.
Article13. Roach M 3rd, Marquez C, Yuo HS, Narayan P, Coleman L, Nseyo UO, et al. Predicting the risk of lymph node involvement using the pre-treatment prostate specific antigen and Gleason score in men with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 1994; 28:33–37.
Article14. Deville C, Both S, Hwang WT, Tochner Z, Vapiwala N. Clinical toxicities and dosimetric parameters after whole-pelvis versus prostate-only intensity-modulated radiation therapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2010; 78:763–772.
Article15. Storey MR, Pollack A, Zagars G, Smith L, Antolak J, Rosen I. Complications from radiotherapy dose escalation in prostate cancer: preliminary results of a randomized trial. Int J Radiat Oncol Biol Phys. 2000; 48:635–642.
Article16. Peeters ST, Heemsbergen WD, van Putten WL, Slot A, Tabak H, Mens JW, et al. Acute and late complications after radiotherapy for prostate cancer: results of a multicenter randomized trial comparing 68 Gy to 78 Gy. Int J Radiat Oncol Biol Phys. 2005; 61:1019–1034.
Article17. Michalski JM, Winter K, Purdy JA, Parliament M, Wong H, Perez CA, et al. Toxicity after three-dimensional radiotherapy for prostate cancer on RTOG 9406 dose Level V. Int J Radiat Oncol Biol Phys. 2005; 62:706–713.
Article18. Cho JH, Lee CG, Kang DR, Kim J, Lee S, Suh CO, et al. Positional reproducibility and effects of a rectal balloon in prostate cancer radiotherapy. J Korean Med Sci. 2009; 24:894–903.
Article19. Bastasch MD, Teh BS, Mai WY, McGary JE, Grant WH 3rd, Butler EB. Tolerance of endorectal balloon in 396 patients treated with intensity-modulated radiation therapy (IMRT) for prostate cancer. Am J Clin Oncol. 2006; 29:8–11.
Article20. Ronson BB, Yonemoto LT, Rossi CJ, Slater JM, Slater JD. Patient tolerance of rectal balloons in conformal radiation treatment of prostate cancer. Int J Radiat Oncol Biol Phys. 2006; 64:1367–1370.
Article21. Keiler L, Dobbins D, Kulasekere R, Einstein D. Tomotherapy for prostate adenocarcinoma: a report on acute toxicity. Radiother Oncol. 2007; 84:171–176.
Article22. Zelefsky MJ, Chan H, Hunt M, Yamada Y, Shippy AM, Amols H. Long-term outcome of high dose intensity modulated radiation therapy for patients with clinically localized prostate cancer. J Urol. 2006; 176(4 Pt 1):1415–1419.
Article23. Teh BS, Mai WY, Augspurger ME, Uhl BM, McGary J, Dong L, et al. Intensity modulated radiation therapy (IMRT) following prostatectomy: more favorable acute genitourinary toxicity profile compared to primary IMRT for prostate cancer. Int J Radiat Oncol Biol Phys. 2001; 49:465–472.
Article24. Yoshimura R, Iwata M, Shibuya H, Sakai Y, Kihara K. Acute and late genitourinary toxicity of conformal radiotherapy for prostate cancer. Radiat Med. 2006; 24:553–559.
Article25. Ishikawa H, Tsuji H, Kamada T, Hirasawa N, Yanagi T, Mizoe JE, et al. Adverse effects of androgen deprivation therapy on persistent genitourinary complications after carbon ion radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2008; 72:78–84.
Article26. Peeters ST, Hoogeman MS, Heemsbergen WD, Slot A, Tabak H, Koper PC, et al. Volume and hormonal effects for acute side effects of rectum and bladder during conformal radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2005; 63:1142–1152.
Article27. Schultheiss TE, Lee WR, Hunt MA, Hanlon AL, Peter RS, Hanks GE. Late GI and GU complications in the treatment of prostate cancer. Int J Radiat Oncol Biol Phys. 1997; 37:3–11.
Article28. Zelefsky MJ, Levin EJ, Hunt M, Yamada Y, Shippy AM, Jackson A, et al. Incidence of late rectal and urinary toxicities after three-dimensional conformal radiotherapy and intensity-modulated radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2008; 70:1124–1129.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A comparison between high dose rate brachytherapy and stereotactic body radiotherapy boost after elective pelvic irradiation for high and very high-risk prostate cancer
- Helical Tomotherapy in Elderly Prostate Cancer Patients
- Dose escalated simultaneous integrated boost of gross nodal disease in gynecologic cancers: a multi-institutional retrospective analysis and review of the literature
- Clinical Outcomes of CyberKnife Radiotherapy in Prostate Cancer Patients: Short-term, Single-Center Experience
- Magnetic resonance image-based tomotherapy planning for prostate cancer