J Gynecol Oncol.
2010 Jun;21(2):119-124. 10.3802/jgo.2010.21.2.119.
Follicular stimulating hormone enhances Notch 1 expression in SK-OV-3 ovarian cancer cells
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Hallym University Sacred Heart Hospital, Anyang, Korea. drparkyh@yahoo.co.kr
- 2Ilsong Institute of Life Science, Hallym University, Anyang, Korea.
- 3Department of Obstetrics and Gynecology, Columbia University Medical Center, New York, NY, USA.
- KMID: 2009582
- DOI: http://doi.org/10.3802/jgo.2010.21.2.119
Abstract
OBJECTIVE
Notch is known as a transmembranous receptor family with four homologous forms - Notch 1, Notch 2, Notch 3, and Notch 4 and related to cell fate regulation and angiogenesis. The purpose is to investigate the effect of follicular stimulating hormone (FSH) on the Notch 1 expression and proliferation in ovarian cancer cells.
METHODS
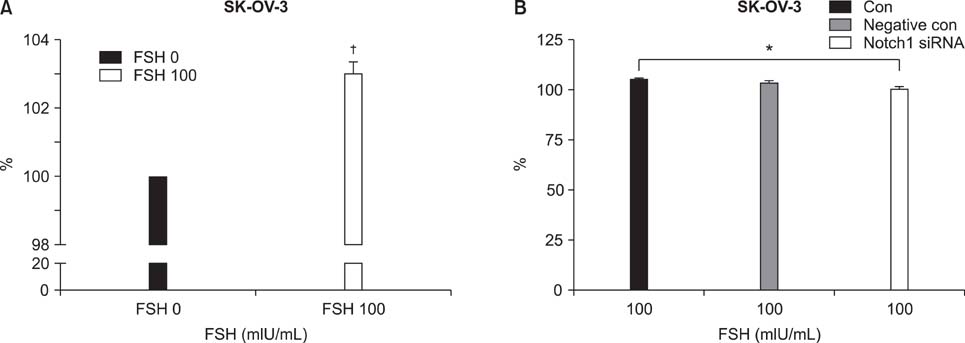
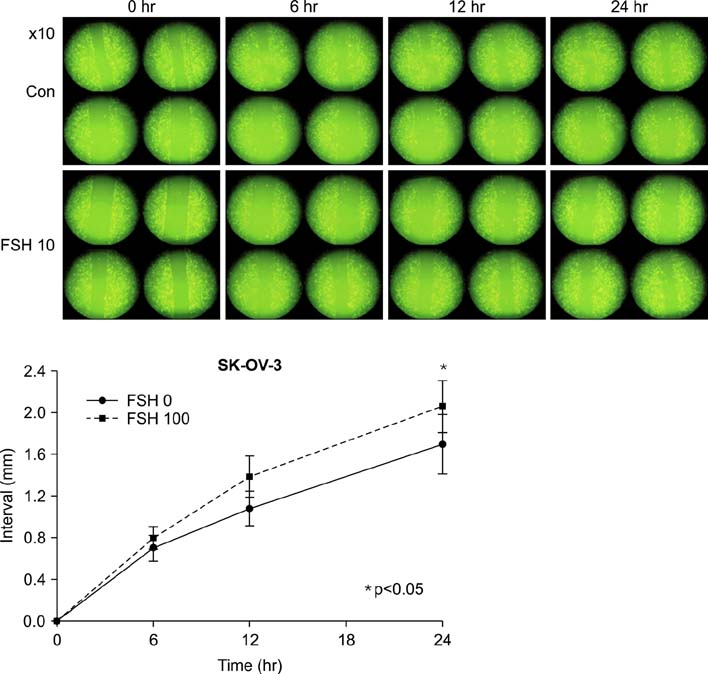
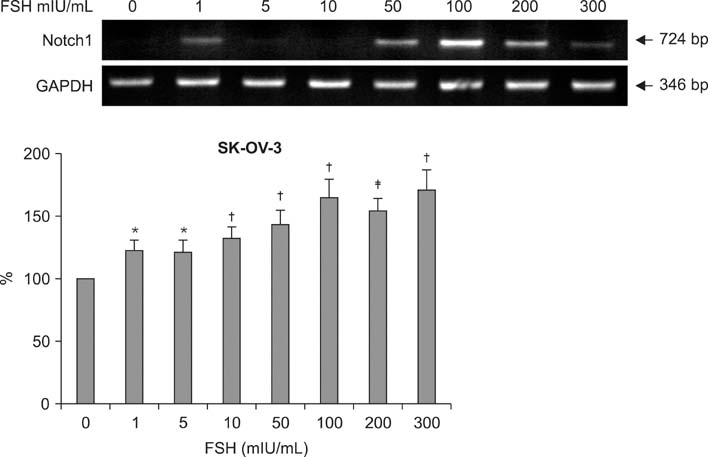
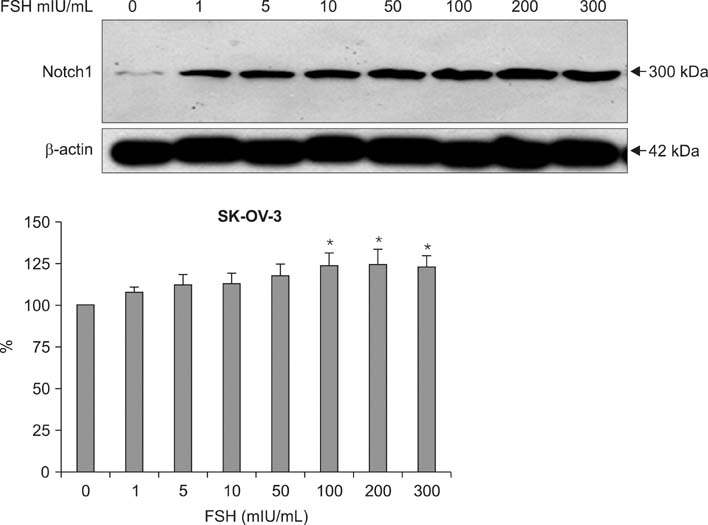
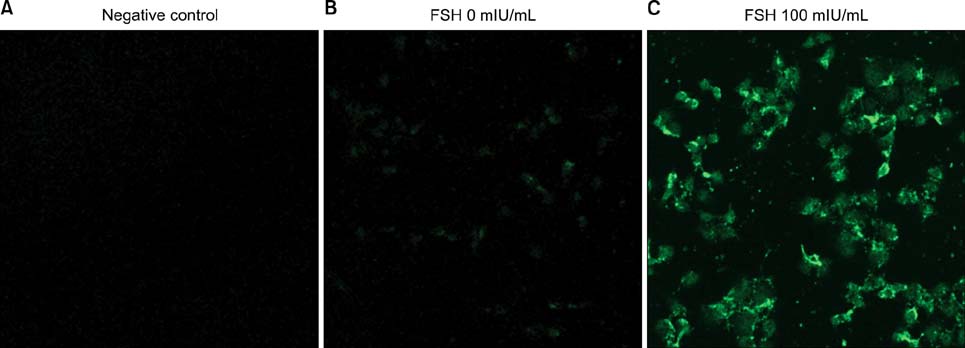
Human ovarian cancer cell line, SK-OV-3 and FSH were used. XTT cell proliferation and cell migration assay were carried out with FSH 100 mIU/mL and Notch 1 siRNA. Western blots and reverse transcriptase-polymerase chain reactions (RT-PCR) were carried out to determine the expression level of the Notch 1 protein and mRNA with FSH treatment in 0, 1, 5, 10, 100, 200, 300 mIU/mL concentrations. Immunofluorescent (IF) stains were performed in SK-OV-3 cell cultures with FSH 100 mIU/mL. Student-t tests were used in statistical analyses.
RESULTS
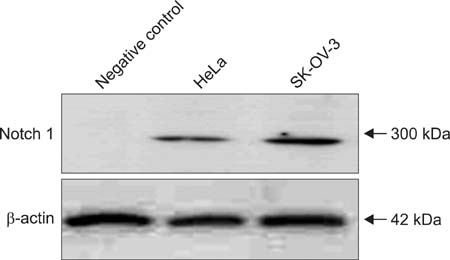
The SK-OV-3 have Notch 1 receptors in their natural status. FSH stimulated SK-OV-3 cells in XTT cell proliferation and cell migration assays and notch 1 siRNA inhibited. The expression level of Notch 1 protein and mRNA were increased in a dose dependent pattern according to FSH concentrations compared to untreated cells. IF stains also showed brighter Notch1 expressions in the FSH treated cells compared to the control cells.
CONCLUSION
FSH enhances proliferation & migration and Notch 1 signaling in SK-OV-3 cells. The Notch signaling probably supports one of the cell proliferating mechanisms of FSH in ovarian cancer cells.
MeSH Terms
Figure
Cited by 2 articles
-
Nerve Growth Factor Stimulates Glioblastoma Proliferation through Notch1 Receptor Signaling
Jun Chul Park, In Bok Chang, Jun Hyong Ahn, Ji Hee Kim, Joon Ho Song, Seung Myung Moon, Young-Han Park
J Korean Neurosurg Soc. 2018;61(4):441-449. doi: 10.3340/jkns.2017.0219.Propranolol Inhibits the Proliferation of Human Glioblastoma Cell Lines through Notch1 and Hes1 Signaling System
Hyun Sik Kim, Young Han Park, Heui Seung Lee, Mi Jung Kwon, Joon Ho Song, In Bok Chang
J Korean Neurosurg Soc. 2021;64(5):716-725. doi: 10.3340/jkns.2021.0068.
Reference
-
1. Berg JW, Lampe JG. High-risk factors in gynecologic cancer. Cancer. 1981. 48:429–441.2. Lawson ND, Scheer N, Pham VN, Kim CH, Chitnis AB, Campos-Ortega JA, et al. Notch signaling is required for arterial-venous differentiation during embryonic vascular development. Development. 2001. 128:3675–3683.3. Esni F, Ghosh B, Biankin AV, Lin JW, Albert MA, Yu X, et al. Notch inhibits Ptf1 function and acinar cell differentiation in developing mouse and zebrafish pancreas. Development. 2004. 131:4213–4224.4. Alitalo K, Tammela T, Petrova TV. Lymphangiogenesis in development and human disease. Nature. 2005. 438:946–953.5. Fox KM, Dias JA, Van Roey P. Three-dimensional structure of human follicle-stimulating hormone. Mol Endocrinol. 2001. 15:378–389.6. Monniaux D, Huet C, Besnard N, Clement F, Bosc M, Pisselet C, et al. Follicular growth and ovarian dynamics in mammals. J Reprod Fertil Suppl. 1997. 51:3–23.7. van der Meer M, Hompes PG, Scheele F, Schoute E, Veersema S, Schoemaker J. Follicle stimulating hormone (FSH) dynamics of low dose step-up ovulation induction with FSH in patients with polycystic ovary syndrome. Hum Reprod. 1994. 9:1612–1617.8. Yoo HR, Yoon MK, Park YH, Park HR, Jang PR. The effects of gonadotropins on the development of ovarian cancer. Korean J Obstet Gynecol. 2004. 47:1698–1705.9. Simon WE, Albrecht M, Hansel M, Dietel M, Holzel F. Cell lines derived from human ovarian carcinomas: growth stimulation by gonadotropic and steroid hormones. J Natl Cancer Inst. 1983. 70:839–845.10. Wimalasena J, Dostal R, Meehan D. Gonadotropins, estradiol, and growth factors regulate epithelial ovarian cancer cell growth. Gynecol Oncol. 1992. 46:345–350.11. Ji Q, Liu PI, Chen PK, Aoyama C. Follicle stimulating hormone-induced growth promotion and gene expression profiles on ovarian surface epithelial cells. Int J Cancer. 2004. 112:803–814.12. Hopfer O, Zwahlen D, Fey MF, Aebi S. The Notch pathway in ovarian carcinomas and adenomas. Br J Cancer. 2005. 93:709–718.13. Zagouras P, Stifani S, Blaumueller CM, Carcangiu ML, Artavanis-Tsakonas S. Alterations in Notch signaling in neoplastic lesions of the human cervix. Proc Natl Acad Sci U S A. 1995. 92:6414–6418.14. Reedijk M, Odorcic S, Chang L, Zhang H, Miller N, McCready DR, et al. High-level coexpression of JAG1 and NOTCH1 is observed in human breast cancer and is associated with poor overall survival. Cancer Res. 2005. 65:8530–8537.15. Collins BJ, Kleeberger W, Ball DW. Notch in lung development and lung cancer. Semin Cancer Biol. 2004. 14:357–364.16. Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD, et al. TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell. 1991. 66:649–661.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of steroids, gonadotropins and epidermal growth factor on ovarian surface epithelial cell proliferation in vitro
- Aberrant epigenetic regulation of GABRP associates with aggressive phenotype of ovarian cancer
- A study of low dose purified follicle-stimulating hormone supplemented with gonadotropin releasing hormone agonist in women with polycystic ovarian disease
- DNA Hypomethylation-Mediated Overexpression of Carbonic Anhydrase 9 Induces an Aggressive Phenotype in Ovarian Cancer Cells
- Apoptosis of the mitochondria protein p32 (gc1qbp) in human ovarian cancer cells