Korean J Lab Med.
2006 Dec;26(6):412-417. 10.3343/kjlm.2006.26.6.412.
Mechanism of VanB Phenotype in Vancomycin-Resistant Enterococci carrying vanA gene
- Affiliations
-
- 1Department of Family Medicine, Ajou University School of Medicine, Suwon, Korea.
- 2Department of Laboratory Medicine, Ajou University School of Medicine, Suwon, Korea. weegyo@ajou.ac.kr
- 3Department of Laboratory Medicine, University of Kwandong College of Medicine, Goyang, Korea.
- 4Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea.
- 5Department of Laboratory Medicine, Seoul University College of Medicine, Seoul, Korea.
- 6Department of Laboratory Medicine, Kosin University College of Medicine, Busan, Korea.
- 7Department of Laboratory Medicine, The Catholic University of Korea College of Medicine, Seoul, Korea.
- 8Department of Laboratory Medicine, Hanyang University College of Medicine, Seoul, Korea.
- 9Department of Laboratory Medicine, Yonsei University Wonju University College of Medicine, Wonju, Korea.
- 10Department of Laboratory Medicine, Chonnam National University College of Medicine, Gwangju, Korea.
- 11Department of Laboratory Medicine, Keonyang University College of Medicine, Daejeon, Korea.
- 12Department of Laboratory Medicine, Sooncheonhyang University College of Medicine, Gumi, Korea.
- 13Department of Laboratory Medicine, Cheju Hanmaeum Hospital13, Jeju, Korea.
- 14Center for Food Safety Evaluation, Korea Food and Drug Administration, Seoul, Korea.
- KMID: 1889819
- DOI: http://doi.org/10.3343/kjlm.2006.26.6.412
Abstract
-
BACKGROUND: Recently, vancomycin-resistant enterococci (VRE) with the vanA genotype that are susceptible to teicoplanin have been described in Japan, Taiwan, and Korea. The investigators suggested three point mutations in the putative sensor domain of vanS or impairment of accessory proteins VanY and VanZ as an explanation for the VanB phenotype-vanA genotype VRE. In this study, we analyzed Tn1546-like elements to determine the molecular mechanisms responsible for the impaired glycopeptide resistance of clinical VRE isolates with VanB phenotype-vanA genotype from Korea.
METHODS
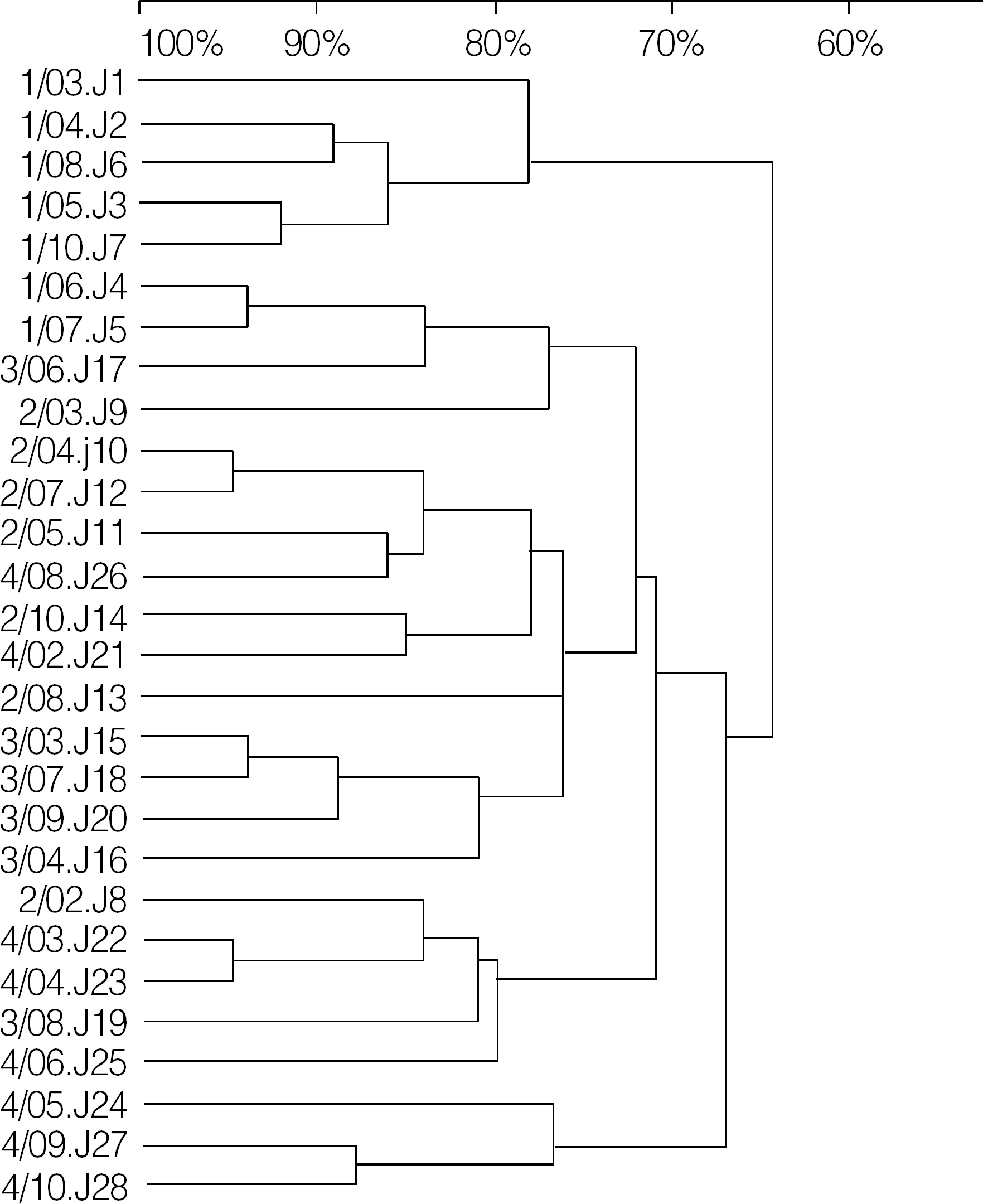
From 2001 to 2004, 28 clinical isolates of Enterococcus faecium with VanB phenotypevanA genotype were collected from 8 different university hospitals in diverse geographic areas in Korea. For structural analysis of Tn1546-like elements, PCR amplifications for internal regions of Tn1546 were performed. The purified PCR products were directly sequenced with an ABI Prism 3100 DNA sequencer.
RESULTS
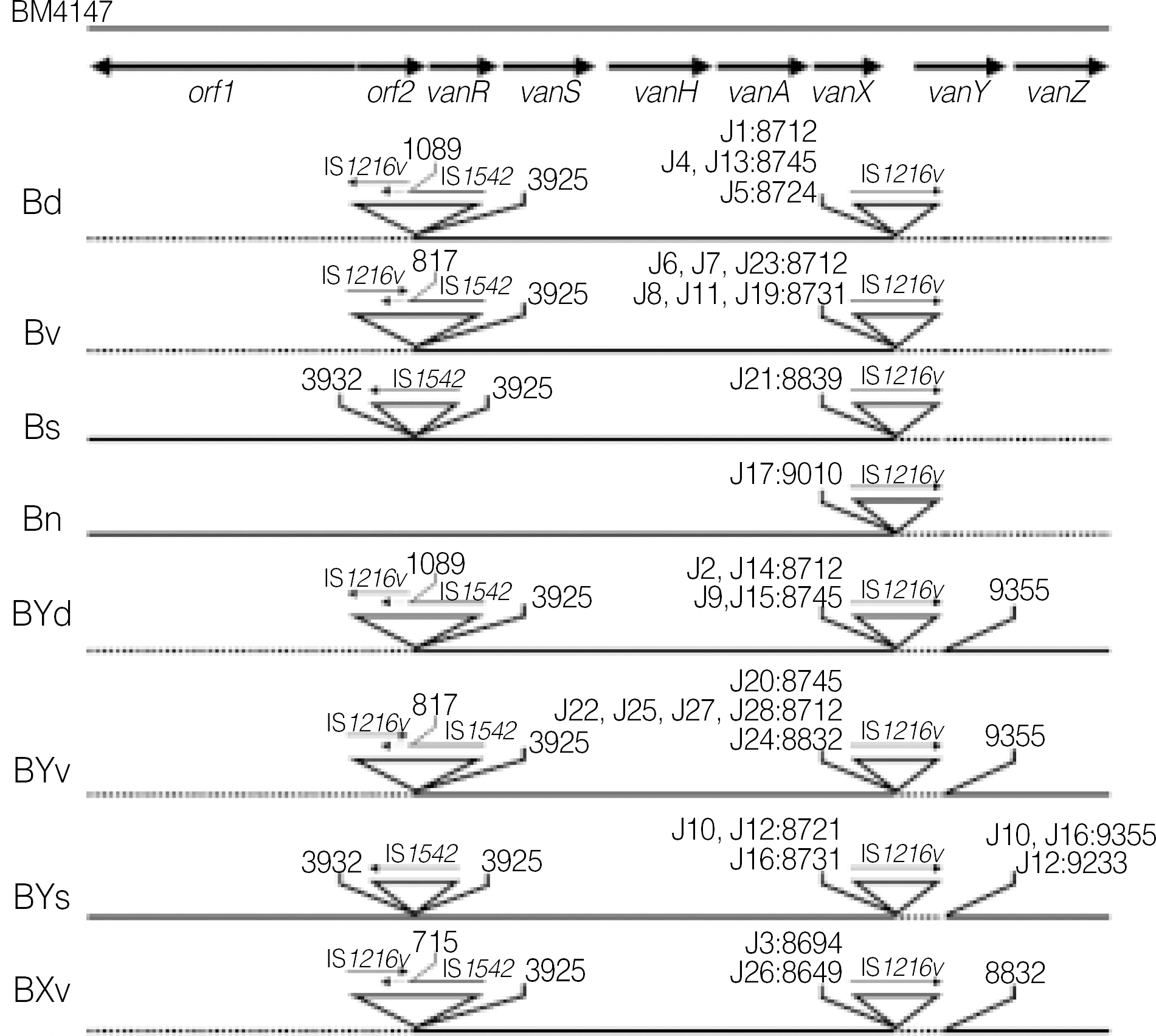
The sequence data of the vanS regulatory gene revealed that none of the isolates had any point mutations in this gene. All 28 isolates had a complete or incomplete deletion of vanY gene. Of these, 13 strains represented a complete deletion of vanZ, and 2 strains showed the deletion of nucleotides near the end point of vanX.
CONCLUSIONS
The mechanism of VanB phenotype-vanA genotype in VRE isolates from Korea is not point mutations of vanS but the rearrangements of vanX, vanY and vanZ.
MeSH Terms
Figure
Cited by 1 articles
-
Evaluation of the iNtRON VRE
vanA/vanB Real-Time PCR Assay for Detection of Vancomycin-Resistant Enterococci
Hee Jae Huh, Mi-Ae Jang, Ja Young Seo, Ji-Youn Kim, Chang-Seok Ki, Jong-Won Kim, Nam Yong Lee
Ann Lab Med. 2015;35(1):76-81. doi: 10.3343/alm.2015.35.1.76.
Reference
-
References
1. Leclercq R, Derlot E, Duval J, Courvalin P. Plasmid-mediated resistance to vancomycin & teicoplanin in Enterococcus faecium. N Engl J Med. 1988; 319:157–61.2. Uttley AH, Collins CH, Naidoo J, George RC. Vancomycin-resistant enterococci. Lancet. 1988; 1:57–8.
Article3. Centers for Disease Control and Prevention. Nosocomial enterococci resistance to vancomycin-United States, 1989–1993. MMWR Morb Mortal Wkly Rep. 1993; 42:597–9.4. National Nosocomial Infections Surveillance System. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control. 2004; 32:470–85.5. Park JW, Kim YR, Shin WS, Kang MW, Han KJ, Shim SI. Susceptibility tests of vancomycin-resistant enterococci. Korean J Infect Dis. 1992; 24:133–8.6. Shin JW, Yong D, Kim MS, Chang KH, Lee K, Kim JM, et al. Sudden increase of vancomycin-resistant enterococcal infections in a Korean tertiary care hospital: possible consequences of increased use of oral vancomycin. J Infect Chemother. 2003; 9:62–7.
Article7. Cetinkaya Y, Falk P, Mayhall CG. Vancomycin-resistant enterococci. Clin Microbiol Rev. 2000; 13:686–707.
Article8. Arthur M, Courvalin P. Genetics and mechanisms of glycopeptide resistance in enterococci. Antimicrob Agents Chemother. 1993; 37:1563–71.
Article9. Leclercq R, Dutka-Malen S, Duval J, Courvalin P. Vancomycin resistance gene vanC is specific to Enterococcus gallinarum. Antimicrob Agents Chemother. 1992; 36:2005–8.10. Perichon B, Reynolds P, Courvalin P. VanD-type glycopeptide-resistant Enterococcus faecium BM4339. Antimicrob Agents Chemother. 1997; 41:2016–8.11. Fines M, Perichon B, Reynolds P, Sahm DF, Courvalin P. VanE, a new type of acquired glycopeptide resistance in Enterococcus faecalis BM4405. Antimicrob Agents Chemother. 1999; 43:2161–4.12. McKessar SJ, Berry AM, Bell JM, Turnidge JD, Paton JC. Genetic characterization of vanG, a novel vancomycin resistance locus of Enterococcus faecalis. Antimicrob Agents Chemother. 2000; 44:3224–8.13. Hashimoto Y, Tanimoto K, Ozawa Y, Murata T, Ike Y. Amino acid substitutions in the vanS sensor of the vanA-type vancomycin-resistant enterococcus strains result in high-level vancomycin resistance and low-level teicoplanin resistance. FEMS Microbiol Lett. 2000; 185:247–54.14. Lauderdale TL, McDonald LC, Shiau YR, Chen PC, Wang HY, Lai JF, et al. Vancomycin-resistant enterococci from humans and retail chickens in Taiwan with unique VanB phenotype-vanA genotype incongruence. Antimicrob Agents Chemother. 2002; 46:525–7.15. Eom JS, Hwang IS, Hwang BY, Lee JG, Lee YJ, Cheong HJ, et al. Emergence of vanA genotype vancomycin-resistant enterococci with low or moderate levels of teicoplanin resistance in Korea. J Clin Microbiol. 2004; 42:1785–6.16. Lee WG, Huh JY, Cho SR, Lim YA. Reduction in glycopeptide resistance in vancomycin-resistant enterococci as a result of vanA cluster rearrangements. Antimicrob Agents Chemother. 2004; 48:1379–81.17. Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 7th ed.Approved standard M7-A7. Wayne, Pa: Clinical and Laboratory Standards Institute;2006.18. Murray BE, Singh KV, Heath JD, Sharma BR, Weinstock GM. Comparison of genomic DNAs of different enterococcal isolates using restriction endonucleases with infrequent recognition sites. J Clin Microbiol. 1990; 28:2059–63.
Article19. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Sixteenth informational supplement, M2-A9 and M7-A7. Wayne, PA: Clinical and Laboratory Standards Institute;2006.20. Arthur M, Molinas C, Depardieu F, Courvallin P. Characterization of Tn1546, a Tn3-related transposon conferring glycopeptide resistance by synthesis of depsipeptide peptidoglycan precursors in Enterococcus faecium BM4147. J Bacteriol. 1993; 175:117–27.21. Arthur M, Molinas C, Courvalin P. Sequence of the vanY gene required for production of a vancomycin-inducible D,D-carboxypeptidase in Enterococcus faecium BM4147. Gene. 1992; 120:111–4.22. Arthur M, Molinas C, Courvallin P. The VanS-VanR two-component regulatory system controls synthesis of depsipeptide peptidoglycan precursors in Enterococcus faecium BM4147. J Bacteriol. 1992; 174:2582–91.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- An Enterococcus gallinarum Strain Carrying Both vanA and vanC1 Genes
- Characterization of Vancomycin-Resistant Enterococci Isolated from Stools and Their Acquisition of Vancomycin Resistance
- A Study on Molecular Epidemiology of Vancomycin-Resistant Enterococci Isolated from Hospitals in Korea
- Occurrence of a PCR-Positive but Culture-Negative Case for vanB Vancomycin-Resistant Enterococci in Stool Surveillance
- Use of Methyl-alpha-D-glucopyranoside Test for Species Identification of Vancomycin Resistant Enterococci