Korean J Obstet Gynecol.
2011 Oct;54(10):599-604. 10.5468/KJOG.2011.54.10.599.
Oral contraceptive pill pretreatment in ovarian stimulation with GnRH antagonists for in vitro fertilization: A comparative study
- Affiliations
-
- 1Division of Reproductive Endocrinology and Infertility, Department of Obstetrics and Gynecology, Cheil General Hospital and Women's Healthcare Center, Kwandong University College of Medicine, Seoul, Korea. kmlyang@naver.com
- KMID: 1836745
- DOI: http://doi.org/10.5468/KJOG.2011.54.10.599
Abstract
OBJECTIVE
To evaluate whether oral contraceptive pill (OCP) pretreatments in gonadotropin-releasing hormone (GnRH) antagonist ovarian stimulation protocols takes positive effects on in vitro fertilization (IVF) outcomes in respect to retrieved oocyte number, oocyte maturation rate, fertilization rate, good quality embryo rate, cycle cancellation rate, pregnancy rate and clinical abortion rate.
METHODS
A total of 194 cycles using GnRH antagonist protocol was performed at infertility clinic of our institute from September 1st, 2009 to February 28th, 2010. The medical records of GnRH antagonist protocols for IVF with or without OCP pretreatment in our IVF unit were retrospectively analyzed. We compared the IVF outcomes between OCP pretreated (n=41) and no pretreatment group (n=153).
RESULTS
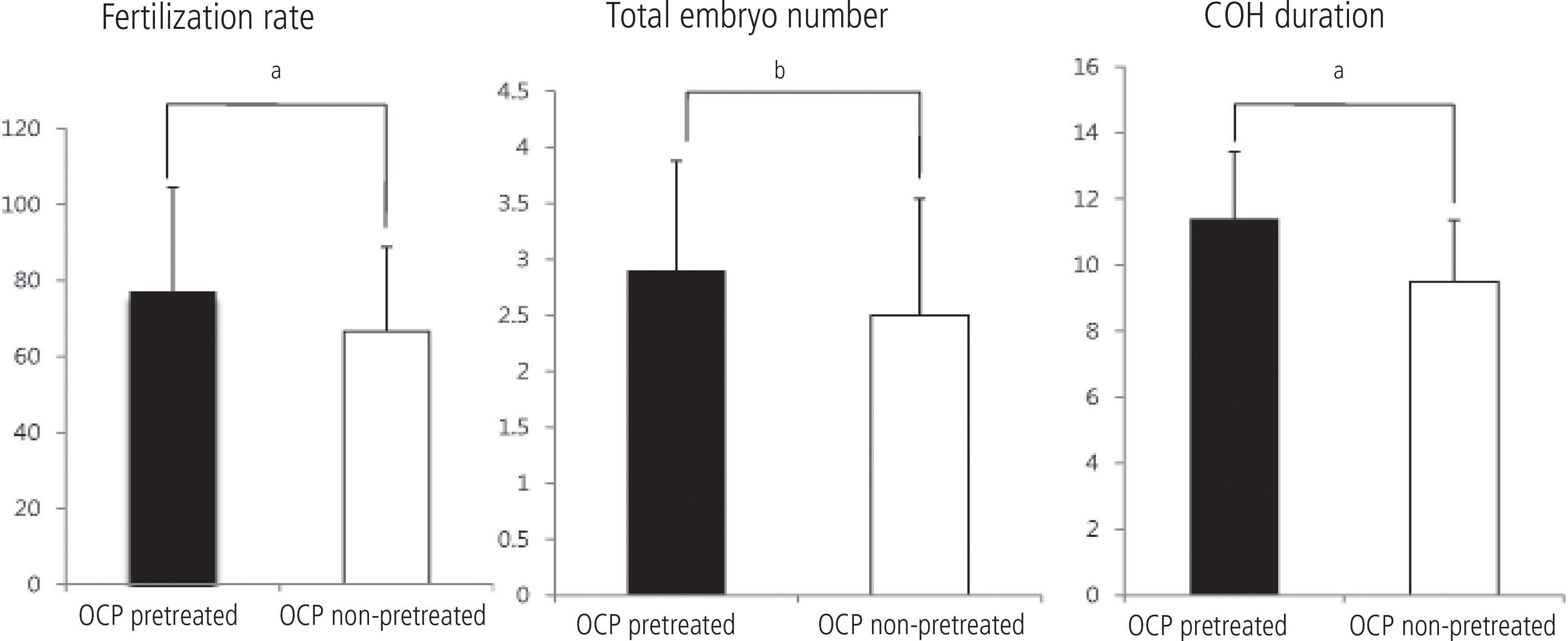
In cycles with OCP pretreated group, the total used dosage of gonadotropin (3019.38+/-1379.00 IU) were higher than that of no pretreatment group (2551.52 +/- 1157.05 IU, P = 0.054). The duration of ovarian stimulation in OCP pretreated group (11.5 +/- 2.0) was significantly longer than that of control group (9.5 +/- 1.9, P = 0.000). The number of gained total embryo (2.8+/-0.9 vs. 2.5+/-1.0, P = 0.055) and fertilization rate (77.2% vs. 65.5%, P = 0.017) were significantly higher in OCP pretreated group. There is no significant difference in pregnancy rate between two groups (39.4% vs. 30.0%, P = 0.304).
CONCLUSION
OCP pretreatment before GnRH antagonist protocol for IVF appears not to have reliable benefit in terms of IVF outcomes. Well-controlled and large-scaled studies are needed.
Keyword
MeSH Terms
Figure
Reference
-
1. Martinez-Salazar J, Cerrillo M, Quea G, Pacheco A, Garcia-Velasco JA. GnRH antagonist ganirelix prevents premature luteinization in IUI cycles: rationale for its use. Reprod Biomed Online. 2009; 19:156–61.
Article2. Berin I, Stein DE, Keltz MD. A comparison of gonadotropin-releasing hormone (GnRH) antagonist and GnRH agonist flare protocols for poor responders undergoing in vitro fertilization. Fertil Steril. 2010; 93:360–3.
Article3. Depalo R, Lorusso F, Palmisano M, Bassi E, Totaro I, Vacca M, et al. Follicular growth and oocyte maturation in GnRH agonist and antagonist protocols for in vitro fertilisation and embryo transfer. Gynecol Endocrinol. 2009; 25:328–34.4. Orvieto R, Rabinson J, Meltzer S, Zohav E, Anteby E, Homburg R. Substituting HCG with GnRH agonist to trigger final follicular maturation: a retrospective comparison of three different ovarian stimulation protocols. Reprod Biomed Online. 2006; 13:198–201.5. Nogueira D, Friedler S, Schachter M, Raziel A, Ron-El R, Smitz J. Oocyte maturity and preimplantation development in relation to follicle diameter in gonadotropin-releasing hormone agonist or antagonist treatments. Fertil Steril. 2006; 85:578–83.
Article6. Fanchin R, Schonäuer LM, Cunha-Filho JS, Méndez Lozano DH, Frydman R. Coordination of antral follicle growth: basis for innovative concepts of controlled ovarian hyperstimulation. Semin Reprod Med. 2005; 23:354–62.
Article7. Fanchin R, Méndez Lozano DH, Schonäuer LM, Cunha-Filho JS, Frydman R. Hormonal manipulations in the luteal phase to coordinate subsequent antral follicle growth during ovarian stimulation. Reprod Biomed Online. 2005; 10:721–8.
Article8. Oehninger S. Ovulation induction in IVF. Minerva Ginecol. 2011; 63:137–56.9. Arslan M, Bocca S, Mirkin S, Barroso G, Stadtmauer L, Oehninger S. Controlled ovarian hyperstimulation protocols for in vitro fertilization: two decades of experience after the birth of Elizabeth Carr. Fertil Steril. 2005; 84:555–69.
Article10. Devreker F, Pogonici E, De Maertelaer V, Revelard P, Van den Bergh M, Englert Y. Selection of good embryos for transfer depends on embryo cohort size: implications for the ‘mild ovarian stimulation’ debate. Hum Reprod. 1999; 14:3002–8.
Article11. Opsahl MS, Blauer KL, Black SH, Lincoln SR, Thorsell L, Sherins RJ. The number of embryos available for transfer predicts successful pregnancy outcome in women over 39 years with normal ovarian hormonal reserve testing. J Assist Reprod Genet. 2001; 18:551–6.12. Fanchin R, Cunha-Filho JS, Schonäuer LM, Kadoch IJ, Cohen-Bacri P, Frydman R. Coordination of early antral follicles by luteal estradiol administration provides a basis for alternative controlled ovarian hyperstimulation regimens. Fertil Steril. 2003; 79:316–21.
Article13. Fanchin R, Cunha-Filho JS, Schonäuer LM, Righini C, de Ziegler D, Frydman R. Luteal estradiol administration strengthens the relationship between day 3 follicle-stimulating hormone and inhibin B levels and ovarian follicular status. Fertil Steril. 2003; 79:585–9.
Article14. Klein NA, Battaglia DE, Fujimoto VY, Davis GS, Bremner WJ, Soules MR. Reproductive aging: accelerated ovarian follicular development associated with a monotropic follicle-stimulating hormone rise in normal older women. J Clin Endocrinol Metab. 1996; 81:1038–45.
Article15. McNatty KP, Hillier SG, van den Boogaard AM, Trimbos-Kemper TC, Reichert LE Jr, van Hall EV. Follicular development during the luteal phase of the human menstrual cycle. J Clin Endocrinol Metab. 1983; 56:1022–31.
Article16. Fanchin R, Salomon L, Castelo-Branco A, Olivennes F, Frydman N, Frydman R. Luteal estradiol pre-treatment coordinates follicular growth during controlled ovarian hyperstimulation with GnRH antagonists. Hum Reprod. 2003; 18:2698–703.
Article17. Kim CH, Jeon GH, Cheon YP, Jeon I, Kim SH, Chae HD, et al. Comparison of GnRH antagonist protocol with or without oral contraceptive pill pretreatment and GnRH agonist low-dose long protocol in low responders undergoing IVF/intracytoplasmic sperm injection. Fertil Steril. 2009; 92:1758–60.
Article18. Bodri D, Sunkara SK, Coomarasamy A. Gonadotropin-releasing hormone agonists versus antagonists for controlled ovarian hyperstimulation in oocyte donors: a systematic review and meta-analysis. Fertil Steril. 2011; 95:164–9.
Article19. Kolibianakis EM, Papanikolaou EG, Camus M, Tournaye H, Van Steirteghem AC, Devroey P. Effect of oral contraceptive pill pretreatment on ongoing pregnancy rates in patients stimulated with GnRH antagonists and recombinant FSH for IVF. A randomized controlled trial. Hum Reprod. 2006; 21:352–7.
Article20. Griesinger G, Venetis CA, Marx T, Diedrich K, Tarlatzis BC, Kolibianakis EM. Oral contraceptive pill pretreatment in ovarian stimulation with GnRH antagonists for IVF: a systematic review and meta-analysis. Fertil Steril. 2008; 90:1055–63.
Article21. Franco JG Jr, Baruffi RL, Mauri AL, Petersen CG, Felipe V, Cor-nicelli J, et al. GnRH agonist versus GnRH antagonist in poor ovarian responders: a meta-analysis. Reprod Biomed Online. 2006; 13:618–27.
Article22. Griesinger G, Kolibianakis EM, Venetis C, Diedrich K, Tarlatzis B. Oral contraceptive pretreatment signifi cantly reduces ongoing pregnancy likelihood in gonadotropin-releasing hormone antagonist cycles: an updated meta-analysis. Fertil Steril.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- GnRH antagonist multiple dose protocol with oral contraceptive pill pretreatment in poor responders undergoing IVF/ICSI
- The effect of oral contraceptive pre-treatment in controlled ovarian hyperstimulation using GnRH antagonist on IVF-ET outcome
- The efficacy of oral contraceptive pre-treatment in controlled ovarian hyperstimulation using a GnRH antagonist in low responders
- The use of gonadotropin-releasing hormone antagonist post-ovulation trigger in ovarian hyperstimulation syndrome
- Pretreatment of normal responders in fresh in vitro fertilization cycles: A comparison of transdermal estradiol and oral contraceptive pills