Clin Orthop Surg.
2014 Dec;6(4):443-454. 10.4055/cios.2014.6.4.443.
Role of Matrix Metalloproteinase (MMP) 2 and MMP-9 in Soft Tissue Sarcoma
- Affiliations
-
- 1Department of Orthopaedic Surgery, Chonnam National University Medical School, Gwangju, Korea. stjung@chonnam.ac.kr
- KMID: 1794734
- DOI: http://doi.org/10.4055/cios.2014.6.4.443
Abstract
- BACKGROUND
We investigated the expression of matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs) in malignant fibrous histiocytoma (MFH), and determined whether these could be useful as prognostic factors.
METHODS
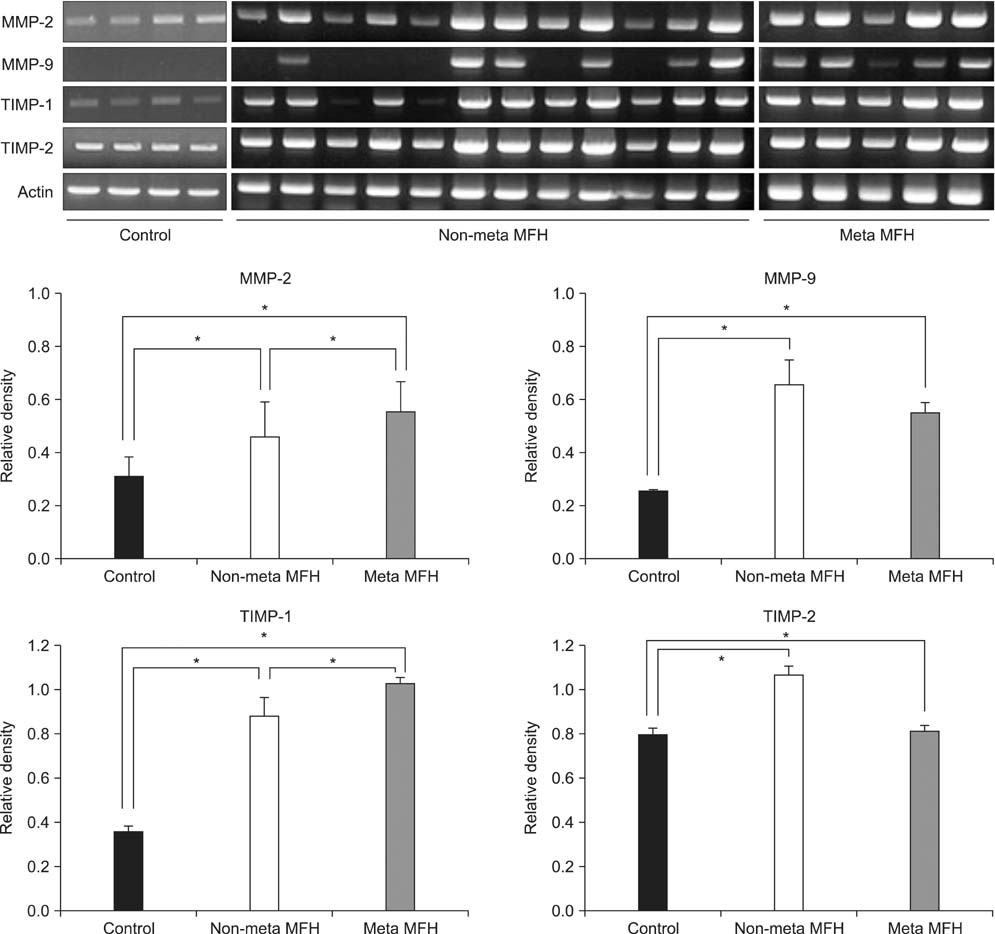
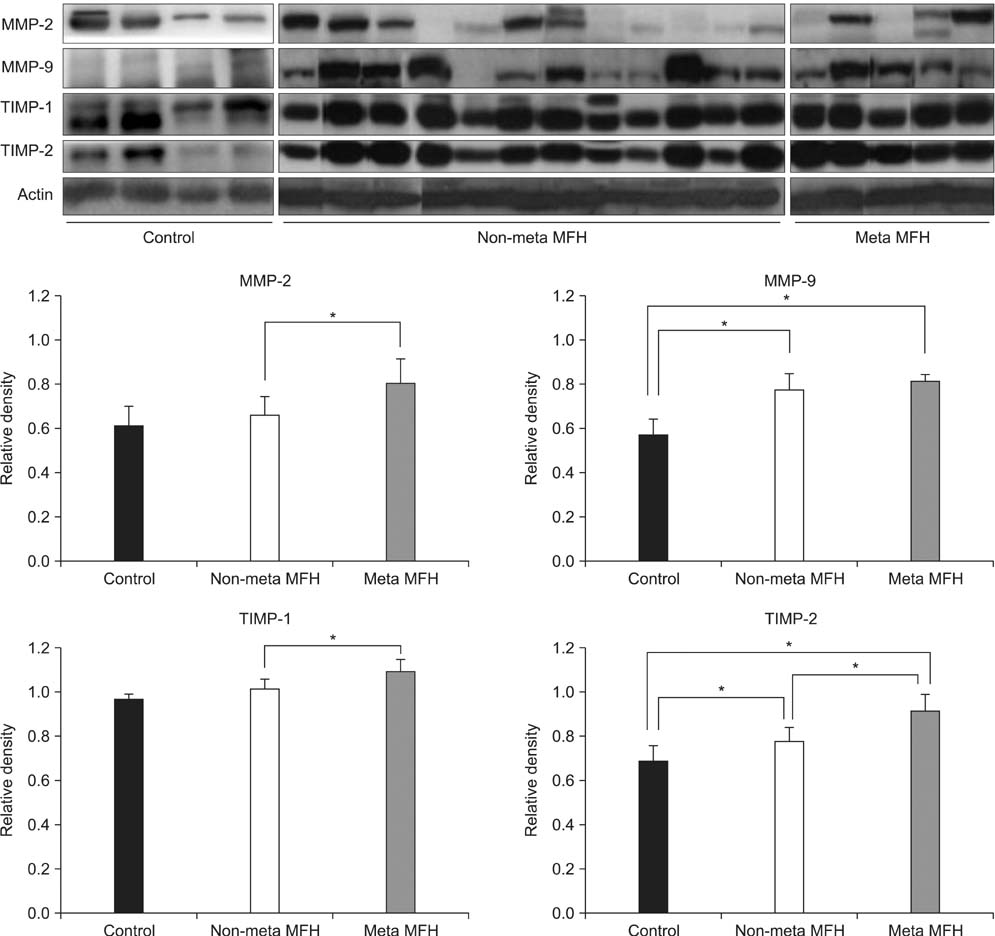
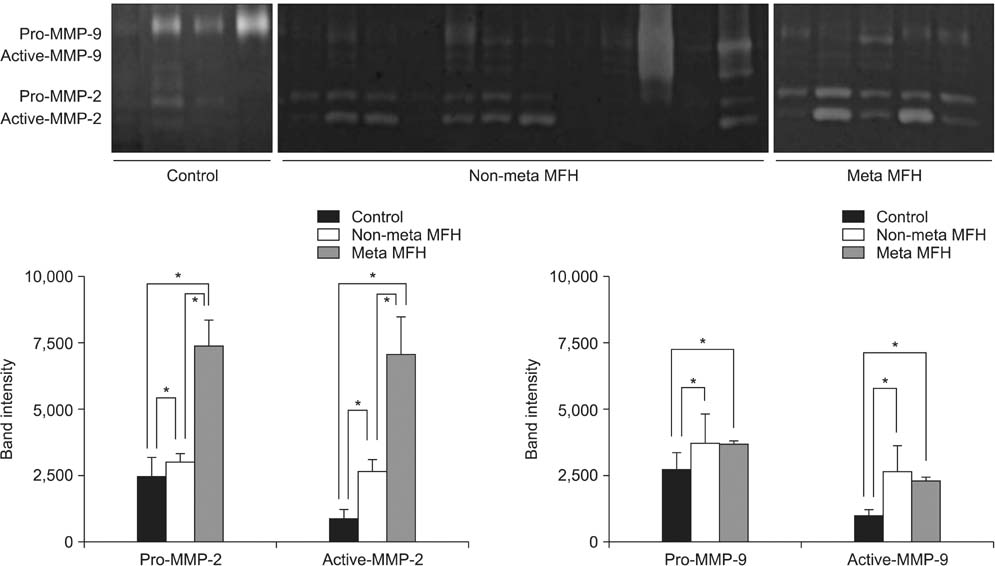
Among patients treated from 1993 to 2007, 30 cases of MFH were evaluated. Immunohistochemical staining was performed for MMP-2, MMP-9, TIMP-1, and TIMP-2 using paraffin wax-embedded blocks of MFH tissues. Reverse transcriptase polymerase chain reaction (RT-PCR) and Western blot and zymography were performed using fresh tissues obtained from 17 of the 30 cases. The levels of MMP and TIMP expression were compared between the MFH and normal control groups, and between non-metastatic and metastatic MFH groups.
RESULTS
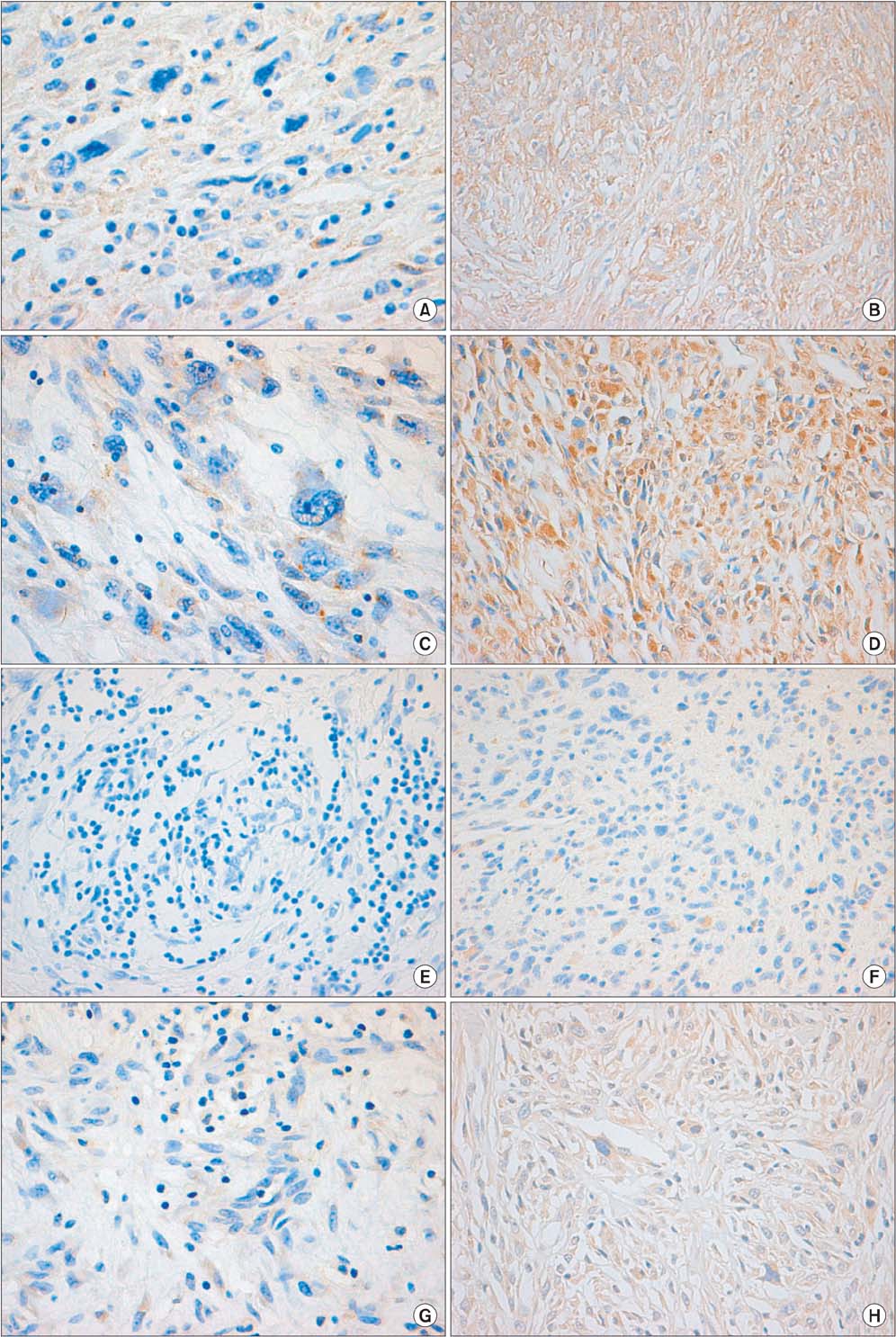
Expression levels of MMP-2, MMP-9, TIMP-1, and TIMP-2 were higher in the MFH group than the control group by RT-PCR, Western blotting, and zymography. Immunohistochemical staining revealed that MMP-2 and MMP-9 protein expression was higher in the metastatic than in the non-metastatic group. The expression levels of MMP-2 and TIMP-1 were significantly higher in the metastatic than in the non-metastatic group (p < 0.05) by RT-PCR. By Western blot analysis, the expression levels of MMP-2, TIMP-1, and TIMP-2 were higher in the metastatic group (p < 0.05), but MMP-9 showed only a slight increase in the metastatic group compared with the non-metastatic group (p > 0.05). Finally, gelatin zymography analysis showed that the expression levels of the pro- and active forms of MMP-2 were significantly higher in the metastatic group (p < 0.05), but the expression of the pro- and active forms of MMP-9 showed a slight decrease in the metastatic group (p > 0.05).
CONCLUSIONS
These results suggest that MMP-2, MMP-9, TIMP-1, and TIMP-2 may have important roles in the development and progression of MFH, and that the degree of expression of these metalloproteinases and their inhibitors, especially MMP-2, could be useful as prognostic factors related to metastasis in MFH.
Keyword
MeSH Terms
-
Adult
Aged
Aged, 80 and over
Female
Histiocytoma, Malignant Fibrous/*metabolism
Humans
Immunohistochemistry
Male
Matrix Metalloproteinase 2/*biosynthesis
Matrix Metalloproteinase 9/*biosynthesis
Middle Aged
Neoplasm Metastasis
Prognosis
Tissue Inhibitor of Metalloproteinase-1/*biosynthesis
Tissue Inhibitor of Metalloproteinase-2/*biosynthesis
Matrix Metalloproteinase 2
Matrix Metalloproteinase 9
Tissue Inhibitor of Metalloproteinase-1
Tissue Inhibitor of Metalloproteinase-2
Figure
Reference
-
1. Bertoni F, Capanna R, Biagini R, et al. Malignant fibrous histiocytoma of soft tissue: an analysis of 78 cases located and deeply seated in the extremities. Cancer. 1985; 56(2):356–367.2. Enzinger FM. Malignant fibrous histiocytoma 20 years after Stout. Am J Surg Pathol. 1986; 10:Suppl 1. 43–53.3. Le Doussal V, Coindre JM, Leroux A, et al. Prognostic factors for patients with localized primary malignant fibrous histiocytoma: a multicenter study of 216 patients with multivariate analysis. Cancer. 1996; 77(9):1823–1830.4. Weiss SW, Enzinger FM. Malignant fibrous histiocytoma: an analysis of 200 cases. Cancer. 1978; 41(6):2250–2266.5. Belal A, Kandil A, Allam A, et al. Malignant fibrous histiocytoma: a retrospective study of 109 cases. Am J Clin Oncol. 2002; 25(1):16–22.6. Gibbs JF, Huang PP, Lee RJ, et al. Malignant fibrous histiocytoma: an institutional review. Cancer Invest. 2001; 19(1):23–27.7. Peiper M, Zurakowski D, Knoefel WT, Izbicki JR. Malignant fibrous histiocytoma of the extremities and trunk: an institutional review. Surgery. 2004; 135(1):59–66.8. Hsu HC, Huang EY, Wang CJ. Treatment results and prognostic factors in patients with malignant fibrous histiocytoma. Acta Oncol. 2004; 43(6):530–535.9. Salo JC, Lewis JJ, Woodruff JM, Leung DH, Brennan MF. Malignant fibrous histiocytoma of the extremity. Cancer. 1999; 85(8):1765–1772.10. Stetler-Stevenson WG, Aznavoorian S, Liotta LA. Tumor cell interactions with the extracellular matrix during invasion and metastasis. Annu Rev Cell Biol. 1993; 9:541–573.11. Sato H, Takino T, Miyamori H. Roles of membrane-type matrix metalloproteinase-1 in tumor invasion and metastasis. Cancer Sci. 2005; 96(4):212–217.12. Jodele S, Blavier L, Yoon JM, DeClerck YA. Modifying the soil to affect the seed: role of stromal-derived matrix metalloproteinases in cancer progression. Cancer Metastasis Rev. 2006; 25(1):35–43.13. Johansson N, Ahonen M, Kahari VM. Matrix metalloproteinases in tumor invasion. Cell Mol Life Sci. 2000; 57(1):5–15.14. Koga K, Nabeshima K, Aoki M, et al. Emmprin in epithelioid sarcoma: expression in tumor cell membrane and stimulation of MMP-2 production in tumor-associated fibroblasts. Int J Cancer. 2007; 120(4):761–768.15. Liu J, Zhan M, Hannay JA, et al. Wild-type p53 inhibits nuclear factor-kappaB-induced matrix metalloproteinase-9 promoter activation: implications for soft tissue sarcoma growth and metastasis. Mol Cancer Res. 2006; 4(11):803–810.16. Scapolan M, Perin T, Wassermann B, et al. Expression profiles in malignant fibrous histiocytomas: clues for differentiating 'spindle cell' and 'pleomorphic' subtypes. Eur J Cancer. 2008; 44(2):298–309.17. Roebuck MM, Helliwell TR, Chaudhry IH, et al. Matrix metalloproteinase expression is related to angiogenesis and histologic grade in spindle cell soft tissue neoplasms of the extremities. Am J Clin Pathol. 2005; 123(3):405–414.18. Nelson AR, Fingleton B, Rothenberg ML, Matrisian LM. Matrix metalloproteinases: biologic activity and clinical implications. J Clin Oncol. 2000; 18(5):1135–1149.19. Schrohl AS, Holten-Andersen MN, Peters HA, et al. Tumor tissue levels of tissue inhibitor of metalloproteinase-1 as a prognostic marker in primary breast cancer. Clin Cancer Res. 2004; 10(7):2289–2298.20. Sternlicht MD, Werb Z. How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol. 2001; 17:463–516.21. Murphy G, Willenbrock F. Tissue inhibitors of matrix metalloendopeptidases. Methods Enzymol. 1995; 248:496–510.22. Soini Y, Salo T, Oikarinen A, Autio-Harmainen H. Expression of 72 kilodalton and 92 kilodalton type IV collagenase in malignant fibrous histiocytomas and dermatofibromas. Lab Invest. 1993; 69(3):305–311.23. Hurskainen T, Soini Y, Tuuttila A, Hoyhtya M, Oikarinen A, Autio-Harmainen H. Expression of the tissue metal-loproteinase inhibitors TIMP-1 and TIMP-2 in malignant fibrous histiocytomas and dermatofibromas as studied by in situ hybridization and immunohistochemistry. Hum Pathol. 1996; 27(1):42–49.24. Himelstein BP, Asada N, Carlton MR, Collins MH. Matrix metalloproteinase-9 (MMP-9) expression in childhood osseous osteosarcoma. Med Pediatr Oncol. 1998; 31(6):471–474.25. Albini A, Melchiori A, Santi L, Liotta LA, Brown PD, Stetler-Stevenson WG. Tumor cell invasion inhibited by TIMP-2. J Natl Cancer Inst. 1991; 83(11):775–779.26. Reich R, Thompson EW, Iwamoto Y, et al. Effects of inhibitors of plasminogen activator, serine proteinases, and collagenase IV on the invasion of basement membranes by metastatic cells. Cancer Res. 1988; 48(12):3307–3312.27. Holten-Andersen M, Christensen IJ, Nilbert M, et al. Association between preoperative plasma levels of tissue inhibitor of metalloproteinases 1 and rectal cancer patient survival. a validation study. Eur J Cancer. 2004; 40(1):64–72.28. Ferrari C, Benassi S, Ponticelli F, et al. Role of MMP-9 and its tissue inhibitor TIMP-1 in human osteosarcoma: findings in 42 patients followed for 1-16 years. Acta Orthop Scand. 2004; 75(4):487–491.29. Yoshiji H, Harris SR, Raso E, et al. Mammary carcinoma cells over-expressing tissue inhibitor of metalloproteinases-1 show enhanced vascular endothelial growth factor expression. Int J Cancer. 1998; 75(1):81–87.30. Maguire PD, Qi W, Lallemand R, Scully SP. Gelatinase and inhibitor expression in soft tissue sarcomas: lack of correlation with distant metastasis. Oncology. 2000; 59(2):139–144.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Matrix Metalloproteinase-2 (MMP-2) and Tissue Inhibitor of Metalloproteinase-2 (TIMP-2) in Pancreatic Ductal Adenocarcinoma
- MMP-2 and MMP-9 Expressions in Breast Carcinomas and Relationship with Major Prognostic Factors
- Expression of MMP-2, MT1-MMP, and TIMP-2 mRNA in Breast Carcinomas
- Immunodetections of Metalloproteinase-2 and Tissue Inhibitor of Metalloproteinase-2 in the Uterine Endometrium
- The Expressions of Matrix Metalloproteinase-9 and Tissue Inhibitors of Metalloproteinase-1 in Scar Endometriosis and Full Term Endometrium