J Korean Med Sci.
2012 Jan;27(1):78-83. 10.3346/jkms.2012.27.1.78.
Assessment of Serologic Immunity to Diphtheria-Tetanus-Pertussis After Treatment of Korean Pediatric Hematology and Oncology Patients
- Affiliations
-
- 1Department of Pediatrics, College of Medicine, The Catholic University of Korea, Seoul, Korea. kjhan@catholic.ac.kr
- KMID: 1792983
- DOI: http://doi.org/10.3346/jkms.2012.27.1.78
Abstract
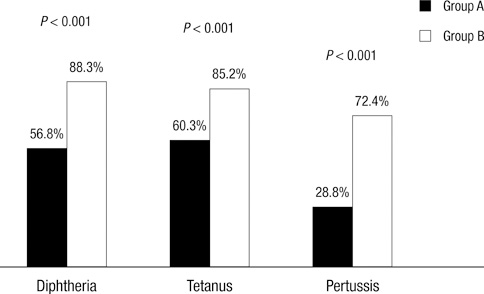
- The aim of this study was to investigate the diphtheria-tetanus-pertussis antibody titers after antineoplastic treatment and to suggest an appropriate vaccination approach for pediatric hemato-oncologic patients. A total of 146 children with either malignancy in remission after cessation of therapy or bone marrow failure were recruited. All children had received routine immunization including diphtheria-tetanus-acellular pertussis vaccination before diagnosis of cancer. The serologic immunity to diphtheria, tetanus and pertussis was classified as: completely protective, partially protective, or non-protective. Non-protective serum antibody titer for diphtheria, tetanus and pertussis was detected in 6.2%, 11.6%, and 62.3% of patients, respectively, and partial protective serum antibody titer for diphtheria, tetanus and pertussis was seen in 37%, 28.1%, and 8.9% of patients. There was no significant correlation between the severity of immune defect and age, gender or underlying disease. Revaccination after antineoplastic therapy showed significantly higher levels of antibody for each vaccine antigen. Our data indicates that a large proportion of children lacked protective serum concentrations of antibodies against diphtheria, tetanus, and pertussis. This suggests that reimmunization of these patients is necessary after completion of antineoplastic treatment. Also, prospective studies should be undertaken with the aim of devising a common strategy of revaccination.
MeSH Terms
-
Adolescent
Age Factors
Antibodies, Bacterial/blood/immunology
Antineoplastic Agents/therapeutic use
Child
Child, Preschool
Diphtheria/immunology/prevention & control
Diphtheria-Tetanus-acellular Pertussis Vaccines/*immunology
Female
Hematologic Neoplasms/*diagnosis/drug therapy
Humans
Immunization, Secondary
Lymphoma/diagnosis/drug therapy
Male
Neuroblastoma/diagnosis/drug therapy
Sex Factors
Tetanus/immunology/prevention & control
Whooping Cough/immunology/prevention & control
Figure
Cited by 2 articles
-
Knowledge and Acceptability about Adult Pertussis Immunization in Korean Women of Childbearing Age
Hyun Sun Ko, Yun Seong Jo, Yeun Hee Kim, Yong-Gyu Park, Jeong Ha Wie, Juyoung Cheon, Hee Bong Moon, Young Lee, Jong Chul Shin
Yonsei Med J. 2015;56(4):1071-1078. doi: 10.3349/ymj.2015.56.4.1071.Immunity to tetanus in major beta thalassemia patients
Abdolreza Sotoodeh Jahromi, Karamatollah Rahmanian
Clin Exp Vaccine Res. 2015;4(2):184-188. doi: 10.7774/cevr.2015.4.2.184.
Reference
-
1. Pui CH, Gajjar AJ, Kane JR, Qaddoumi IA, Pappo AS. Medscape. Challenging issues in pediatric oncology. Nat Rev Clin Oncol. 2011. 8:540–549.2. Alanko S, Pelliniemi TT, Salmi TT. Recovery of blood B-lymphocytes and serum immunoglobulins after chemotherapy for childhood acute lymphoblastic leukemia. Cancer. 1992. 69:1481–1486.3. Smith S, Schiffman G, Karayalcin G, Bonagura V. Immunodeficiency in long-term survivors of acute lymphoblastic leukemia treated with Berlin-Frankfurt-Münster therapy. J Pediatr. 1995. 127:68–75.4. Lehrnbecher T, Foster C, Vázquez N, Mackall CL, Chanock SJ. Therapy-induced alterations in host defense in children receiving therapy for cancer. J Pediatr Hematol Oncol. 1997. 19:399–417.5. Kang JH. The assessment of DTaP vaccine; age related seroepidemiology of diphtheria, tetanus and pertussis in Korea. The annual report of KFDA. 2008. Korea Food & Drug Administration;783–784.6. Chessells JM, Richards SM, Bailey CC, Lilleyman JS, Eden OB. Gender and treatment outcome in childhood lymphoblastic leukaemia: report from the MRC UKALL trials. Br J Haematol. 1995. 89:364–372.7. Pui CH, Evans WE. Treatment of acute lymphoblastic leukemia. N Engl J Med. 2006. 354:166–178.8. Kosmidis S, Baka M, Bouhoutsou D, Doganis D, Kallergi C, Douladiris N, Pourtsidis A, Varvoutsi M, Saxoni-Papageorgiou F, Vasilatou-Kosmidis H. Longitudinal assessment of immunological status and rate of immune recovery following treatment in children with ALL. Pediatr Blood Cancer. 2008. 50:528–532.9. Kristinsson VH, Kristinsson JR, Jonmundsson GK, Jonsson OG, Thorsson AV, Haraldsson A. Immunoglobulin class and subclass concentrations after treatment of childhood leukemia. Pediatr Hematol Oncol. 2001. 18:167–172.10. Crespo I, Cardeñosa N, Godoy P, Carmona G, Sala MR, Barrabeig I, Alvarez J, Minguel S, Camps N, Caylà J, Batalla J, Codina G, Domínguez A. Epidemiology of pertussis in a country with high vaccination coverage. Vaccine. 2011. 29:4244–4248.11. Bell GC, Foster SL. 2010 pertussis outbreak and updated Tdap recommendations. J Am Pharm Assoc (2003). 2011. 51:118–120.12. Saikia L, Nath R, Saikia NJ, Choudhury G, Sarkar M. A diphtheria outbreak in Assam, India. Southeast Asian J Trop Med Public Health. 2010. 41:647–652.13. Morrison C, Maurtua-Neumann P, Myint MT, Drury SS, Bégué RE. Pandemic (H1N1) 2009 outbreak at camp for children with hematologic and oncologic conditions. Emerg Infect Dis. 2011. 17:87–89.14. Buchbinder N, Dumesnil C, Pinquier D, Merle V, Filhon B, Schneider P, Vannier JP. Pandemic A/H1N1/2009 influenza in a paediatric haematology and oncology unit: successful management of a sudden outbreak. J Hosp Infect. 2011. 79:155–160.15. So JS, Go UY, Lee DH, Park KS, Lee JK. Epidemiological investigation of a measles outbreak in a preschool in Incheon, Korea, 2006. J Prev Med Public Health. 2008. 41:153–158.16. von der Hardt K, Jüngert J, Beck JD, Heininger U. Humoral immunity against diphtheria, tetanus and poliomyelitis after antineoplastic therapy in children and adolescents: a retrospective analysis. Vaccine. 2000. 18:2999–3004.17. Ek T, Mellander L, Hahn-Zoric M, Abrahamsson J. Intensive treatment for childhood acute lymphoblastic leukemia reduces immune responses to diphtheria, tetanus, and Haemophilus influenzae type b. J Pediatr Hematol Oncol. 2004. 26:727–734.18. Small TN, Zelenetz AD, Noy A, Rice RD, Trippett TM, Abrey L, Portlock CS, McCullagh EJ, Vanak JM, Mulligan AM, Moskowitz CH. Pertussis immunity and response to tetanus-reduced diphtheria-reduced pertussis vaccine (Tdap) after autologous peripheral blood stem cell transplantation. Biol Blood Marrow Transplant. 2009. 15:1538–1542.19. Zignol M, Peracchi M, Tridello G, Pillon M, Fregonese F, D'Elia R, Zanesco L, Cesaro S. Assessment of humoral immunity to poliomyelitis, tetanus, hepatitis B, measles, rubella, and mumps in children after chemotherapy. Cancer. 2004. 101:635–641.20. Nilsson A, De Milito A, Engström P, Nordin M, Narita M, Grillner L, Chiodi F, Björk O. Current chemotherapy protocols for childhood acute lymphoblastic leukemia induce loss of humoral immunity to viral vaccination antigens. Pediatrics. 2002. 109:e91.21. Urschel S, Cremer S, Birnbaum J, Dallapozza R, Fuchs A, Jager G, Schmitz C, Belohradsky BH, Netz H. Lack of serologic immunity against vaccine-preventable diseases in children after thoracic transplantation. Transpl Int. 2010. 23:619–627.22. Ljungman P, Engelhard D, de la Cámara R, Einsele H, Locasciulli A, Martino R, Ribaud P, Ward K, Cordonnier C. Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Vaccination of stem cell transplant recipients: recommendations of the Infectious Diseases Working Party of the EBMT. Bone Marrow Transplant. 2005. 35:737–746.23. Center for Disease Control and Prevention. Infectious Diseases Society of America. American Society of Blood and Marrow Transplantation. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. Biol Blood Marrow Transplant. 2000. 6:659–713. 715717–727.24. Yu J, Chou AJ, Lennox A, Kleiman P, Wexler LH, Meyers PA, Gorlick R. Loss of antibody titers and effectiveness of revaccination in post-chemotherapy pediatric sarcoma patients. Pediatr Blood Cancer. 2007. 49:656–660.25. Patel SR, Ortin M, Cohen BJ, Borrow R, Irving D, Sheldon J, Heath PT. Revaccination of children after completion of standard chemotherapy for acute leukemia. Clin Infect Dis. 2007. 44:635–642.26. Ercan TE, Soycan LY, Apak H, Celkan T, Ozkan A, Akdenizli E, Kasapçopur O, Yildiz I. Antibody titers and immune response to diphtheria-tetanus-pertussis and measles-mumps-rubella vaccination in children treated for acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2005. 27:273–277.27. Fioredda F, Plebani A, Hanau G, Haupt R, Giacchino M, Barisone E, Balbo L, Castagnola E. Re-immunisation schedule in leukaemic children after intensive chemotherapy: a possible strategy. Eur J Haematol. 2005. 74:20–23.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Rates of Adverse Reactions Associated with Modified DPT Vaccine in Korean Infants and Children
- New DTaP Vaccine
- Efficacy and adverse effects of the new DTaP vaccine
- Tetanus–diphtheria–acellular pertussis vaccination for adults: an update
- The Need of Td Vaccination According to the Changes of Tetanus and Diphtheria Immunity