Korean J Physiol Pharmacol.
2015 May;19(3):241-247. 10.4196/kjpp.2015.19.3.241.
p-Coumaric Acid Attenuates UVB-Induced Release of Stratifin from Keratinocytes and Indirectly Regulates Matrix Metalloproteinase 1 Release from Fibroblasts
- Affiliations
-
- 1Department of Molecular Medicine, Cell and Matrix Research Institute, BK21 Plus KNU Biomedical Convergence Program, Department of Biomedical Science, Kyungpook National University School of Medicine, Daegu 700-842, Korea. ycboo@knu.ac.kr
- KMID: 1791425
- DOI: http://doi.org/10.4196/kjpp.2015.19.3.241
Abstract
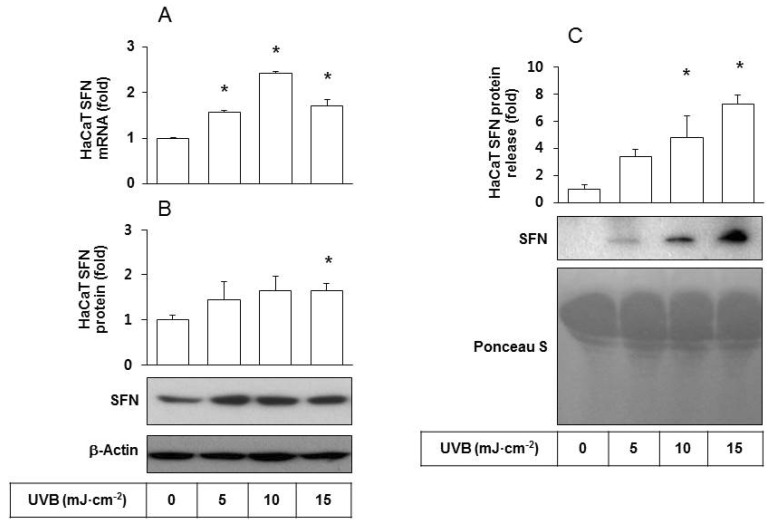
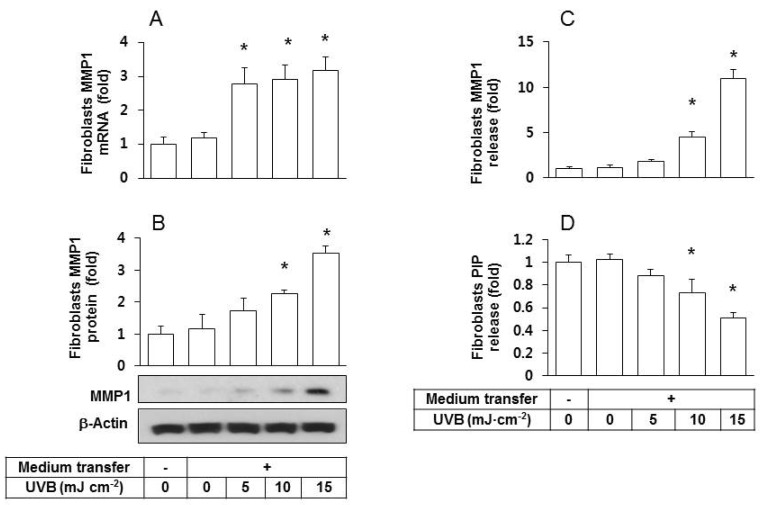
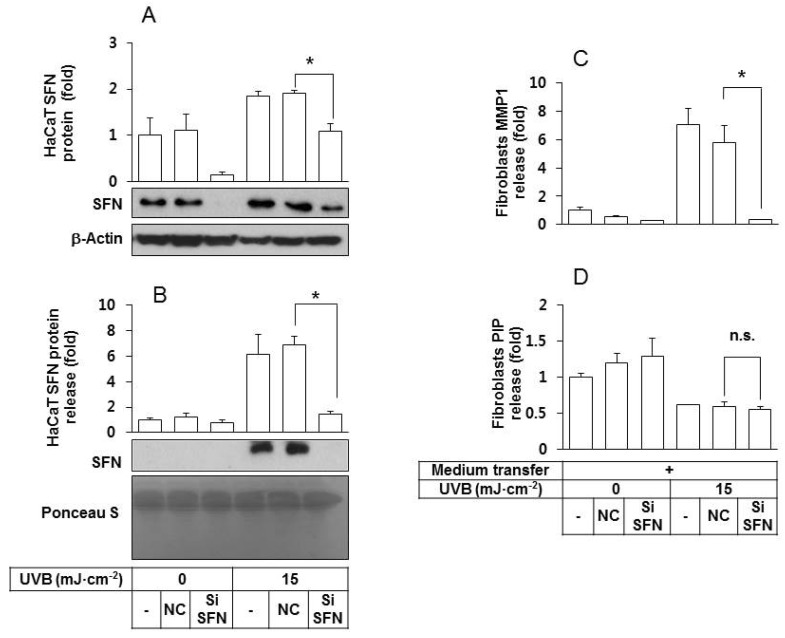
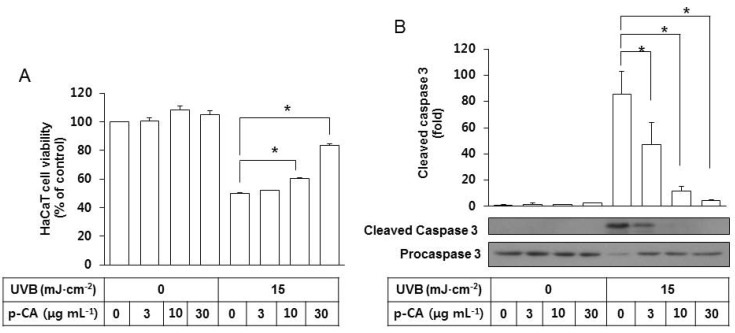
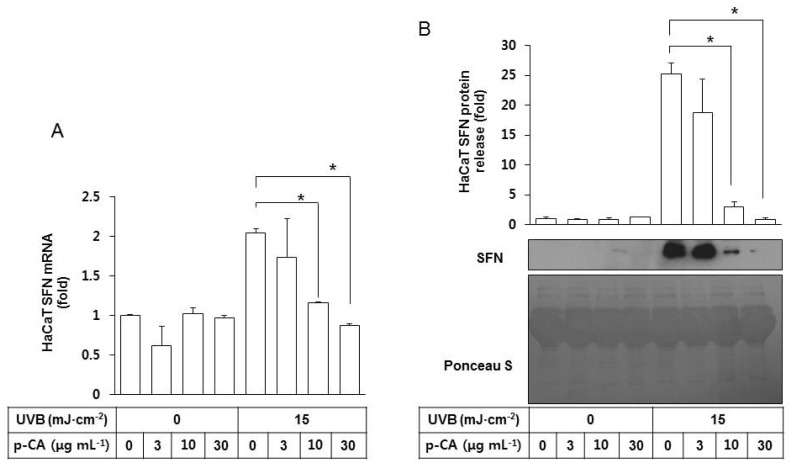
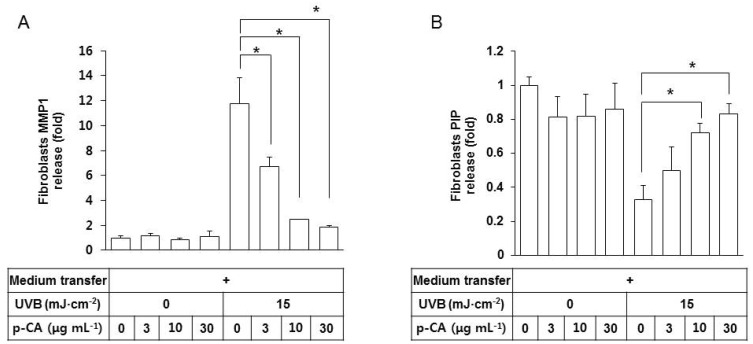
- Ultraviolet (UV) radiation-induced loss of dermal extracellular matrix is associated with skin photoaging. Recent studies demonstrated that keratinocyte-releasable stratifin (SFN) plays a critical role in skin collagen metabolism by inducing matrix metalloproteinase 1 (MMP1) expression in target fibroblasts. In the present study, we examined whether SFN released from UVB-irradiated epidermal keratinocytes increases MMP1 release from dermal fibroblasts, and whether these events are affected by p-coumaric acid (p-CA), a natural phenolic compound with UVB-shielding and antioxidant properties. HaCaT cells were exposed to UVB in the absence and presence of p-CA, and the conditioned medium was used to stimulate fibroblasts in medium transfer experiments. The cells and media were analyzed to determine the expressions/releases of SFN and MMP1. UVB exposure increased SFN release from keratinocytes into the medium. The conditioned medium of UVB-irradiated keratinocytes increased MMP1 release from fibroblasts. The depletion of SFN using a siRNA rendered the conditioned medium of UVB-irradiated keratinocytes ineffective at stimulating fibroblasts to release MMP1. p-CA mitigated UVB-induced SFN expression in keratinocytes, and attenuated the MMP1 release by fibroblasts in medium transfer experiments. In conclusion, the present study demonstrated that the use of UV absorbers such as p-CA would reduce UV-induced SFN-centered signaling events involved in skin photoaging.
Keyword
MeSH Terms
Figure
Reference
-
1. Rabe JH, Mamelak AJ, McElgunn PJ, Morison WL, Sauder DN. Photoaging: mechanisms and repair. J Am Acad Dermatol. 2006; 55:1–19. PMID: 16781287.
Article2. Fisher GJ, Kang S, Varani J, Bata-Csorgo Z, Wan Y, Datta S, Voorhees JJ. Mechanisms of photoaging and chronological skin aging. Arch Dermatol. 2002; 138:1462–1470. PMID: 12437452.
Article3. Kähäri VM, Saarialho-Kere U. Matrix metalloproteinases in skin. Exp Dermatol. 1997; 6:199–213. PMID: 9450622.
Article4. Bode W, Fernandez-Catalan C, Tschesche H, Grams F, Nagase H, Maskos K. Structural properties of matrix metalloproteinases. Cell Mol Life Sci. 1999; 55:639–652. PMID: 10357232.
Article5. Brennan M, Bhatti H, Nerusu KC, Bhagavathula N, Kang S, Fisher GJ, Varani J, Voorhees JJ. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem Photobiol. 2003; 78:43–48. PMID: 12929747.
Article6. Scharffetter K, Wlaschek M, Hogg A, Bolsen K, Schothorst A, Goerz G, Krieg T, Plewig G. UVA irradiation induces collagenase in human dermal fibroblasts in vitro and in vivo. Arch Dermatol Res. 1991; 283:506–511. PMID: 1664713.
Article7. Brenneisen P, Oh J, Wlaschek M, Wenk J, Briviba K, Hommel C, Herrmann G, Sies H, Scharffetter-Kochanek K. Ultraviolet B wavelength dependence for the regulation of two major matrix-metalloproteinases and their inhibitor TIMP-1 in human dermal fibroblasts. Photochem Photobiol. 1996; 64:649–657. PMID: 8863471.
Article8. Ghahary A, Marcoux Y, Karimi-Busheri F, Li Y, Tredget EE, Kilani RT, Lam E, Weinfeld M. Differentiated keratinocyte-releasable stratifin (14-3-3 sigma) stimulates MMP-1 expression in dermal fibroblasts. J Invest Dermatol. 2005; 124:170–177. PMID: 15654971.
Article9. Lam E, Kilani RT, Li Y, Tredget EE, Ghahary A. Stratifin-induced matrix metalloproteinase-1 in fibroblast is mediated by c-fos and p38 mitogen-activated protein kinase activation. J Invest Dermatol. 2005; 125:230–238. PMID: 16098031.
Article10. Adachi H, Murakami Y, Tanaka H, Nakata S. Increase of stratifin triggered by ultraviolet irradiation is possibly related to premature aging of human skin. Exp Dermatol. 2014; 23(Suppl 1):32–36. PMID: 25234834.
Article11. An SM, Lee SI, Choi SW, Moon SW, Boo YC. p-Coumaric acid, a constituent of Sasa quelpaertensis Nakai, inhibits cellular melanogenesis stimulated by alpha-melanocyte stimulating hormone. Br J Dermatol. 2008; 159:292–299. PMID: 18544081.12. Kim M, Park J, Song K, Kim HG, Koh JS, Boo YC. Screening of plant extracts for human tyrosinase inhibiting effects. Int J Cosmet Sci. 2012; 34:202–208. PMID: 22220689.
Article13. Song K, Boo YC. UVB shielding effects of para-Coumaric acid. J Soc Cosmet Scientists Korea. 2012; 38:263–273.14. Lee SJ, Mun GI, An SM, Boo YC. Evidence for the association of peroxidases with the antioxidant effect of p-coumaric acid in endothelial cells exposed to high glucose plus arachidonic acid. BMB Rep. 2009; 42:561–567. PMID: 19788856.
Article15. Song K, An SM, Kim M, Koh JS, Boo YC. Comparison of the antimelanogenic effects of p-coumaric acid and its methyl ester and their skin permeabilities. J Dermatol Sci. 2011; 63:17–22. PMID: 21530181.
Article16. Zang LY, Cosma G, Gardner H, Shi X, Castranova V, Vallyathan V. Effect of antioxidant protection by p-coumaric acid on lowdensity lipoprotein cholesterol oxidation. Am J Physiol Cell Physiol. 2000; 279:C954–C960. PMID: 11003575.17. Lee SI, An SM, Mun GI, Lee SJ, Park KM, Park SH, Boo YC. Protective effect of Sasa quelpaertensis and p-coumaric acid on ethanol-induced hepatotoxicity in mice. J Appl Biol Chem. 2008; 51:148–154.
Article18. Park J, Seok JK, Suh HJ, Boo YC. Gardenia jasminoides extract attenuates the UVB-induced expressions of cytokines in keratinocytes and indirectly inhibits matrix metalloproteinase-1 expression in human dermal fibroblasts. Evid Based Complement Alternat Med. 2014; 2014:429246. PMID: 24711853.19. Canty-Laird EG, Lu Y, Kadler KE. Stepwise proteolytic activation of type I procollagen to collagen within the secretory pathway of tendon fibroblasts in situ. Biochem J. 2012; 441:707–717. PMID: 21967573.20. Sitailo LA, Tibudan SS, Denning MF. Activation of caspase-9 is required for UV-induced apoptosis of human keratinocytes. J Biol Chem. 2002; 277:19346–19352. PMID: 11919192.
Article21. Lee CH, Wu SB, Hong CH, Yu HS, Wei YH. Molecular mechanisms of UV-induced apoptosis and its effects on skin residential cells: the implication in UV-based phototherapy. Int J Mol Sci. 2013; 14:6414–6435. PMID: 23519108.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Low-dose UVB irradiation stimulates matrix metalloproteinase-1 expression via a BLT2-linked pathway in HaCaT cells
- The Effect of Supernatant from UVB - Irradiated Cultured Keratinocytes on the Growth , Melanin Content , and Tyrosinase Activity of Human Melanocyte
- Simvastatin as a Modulator of Tissue Remodeling through Inhibition of Matrix Metalloproteinase (MMP) Release from Human Lung Fibroblasts
- Oleanolic Acid Protects the Skin from Particulate Matter-Induced Aging
- Effects of Dexamethasone on Endothelin-1(ET-1) Production by Keratinocytes