Immunogenicity and Safety of Trivalent Inactivated Influenza Vaccine: A Randomized, Double-Blind, Multi-Center, Phase 3 Clinical Trial in a Vaccine-Limited Country
- Affiliations
-
- 1Division of Infectious Diseases, Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea. wjkim@medimail.co.kr
- 2Division of Infectious Diseases, Department of Internal Medicine, Hallym University College of Medicine, Chuncheon, Korea.
- 3Division of Infectious Diseases, Department of Internal Medicine, The Catholic University of Korea, School of Medicne, St. Vincent's Hospital, Suwon, Korea.
- 4Division of Infectious Diseases, Department of Internal Medicine, Inha University College of Medicine, Incheon, Korea.
- 5Division of Infectious Diseases, Department of Internal Medicine, The Catholic University of Korea, School of Medicne, Uijeongbu St. Mary's Hospital, Uijeongbu, Korea.
- 6Division of Infectious Diseases, Department of Internal Medicine, Chonnam National University Medical School, Gwangju, Korea.
- 7Division of Infectious Diseases, Department of Internal Medicine, Soon Chun Hyang University Hospital, Seoul, Korea.
- 8Division of Respiratory and Allergy Medicine, Department of Internal Medicine, Soon Chun Hyang University Hospital, Seoul, Korea.
- KMID: 1782105
- DOI: http://doi.org/10.3346/jkms.2011.26.2.191
Abstract
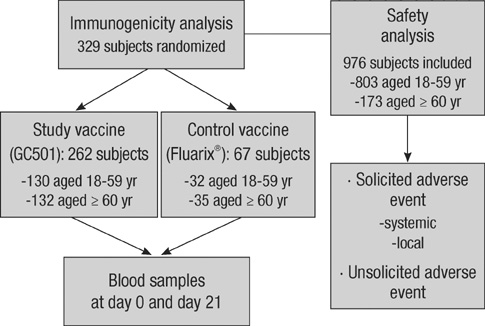
- Influenza vaccines are the primary method for controlling influenza and its complications. This study was conducted as a phase 3, randomized, double-blind, controlled, multi-center trial at seven university hospitals to evaluate the immunogenicity and safety of an inactivated, split, trivalent influenza vaccine (GC501, Green Cross Corporation, Yongin, Korea), which was newly manufactured in Korea in 2008. Between September 21 and 26, a total of 329 healthy subjects were recruited for the immunogenicity analysis, while 976 subjects were enrolled for the safety analysis. The GC501 vaccine met both FDA and EMEA criteria with > or = 80% of subjects achieving post-vaccination titers > or = 40 for all three subtypes, even in the elderly. The vaccine was well tolerated with only mild systemic and local adverse events. In summary, GC501 showed excellent immunogenicity and a good safety profile in both young adults and the elderly. The licensure of GC501 might be an important basis in preparation for the future influenza pandemic.
MeSH Terms
Figure
Cited by 4 articles
-
Immunogenicity and Safety of Trivalent Split Influenza Vaccine in Healthy Korean Adults with Low Pre-Existing Antibody Levels: An Open Phase I Trial
Kyuri Kang, Seunghoon Han, Taegon Hong, Sangil Jeon, Jeongki Paek, Jin Han Kang, Dong-Seok Yim
Yonsei Med J. 2016;57(6):1354-1360. doi: 10.3349/ymj.2016.57.6.1354.Protective Field Efficacy Study of Influenza Vaccines for Korean Children and Adolescent in 2010-2011 Season
Seung Youn Kim, Nam Hee Kim, Byung Wook Eun, So Hee Kim, Ki Won Park, Hyun Oh Jang, Eun Kyeong Kang, Dong Ho Kim
Korean J Pediatr Infect Dis. 2012;19(3):149-156. doi: 10.14776/kjpid.2012.19.3.149.Comparison of immunogenicity and safety of an influenza vaccine administered concomitantly with a 13-valent pneumococcal conjugate vaccine or 23-valent polysaccharide pneumococcal vaccine in the elderly
Yu Bin Seo, Won Suk Choi, Jacob Lee, Joon Young Song, Hee Jin Cheong, Woo Joo Kim
Clin Exp Vaccine Res. 2017;6(1):38-44. doi: 10.7774/cevr.2017.6.1.38.The Impacts of Influenza Infection and Vaccination on Exacerbation of Myasthenia Gravis
Hung Youl Seok, Ha Young Shin, Jong Kuk Kim, Byoung Joon Kim, Jeeyoung Oh, Bum Chun Suh, Sun-Young Kim, Sa-Yoon Kang, Suk-Won Ahn, Jong Seok Bae, Byung-Jo Kim
J Clin Neurol. 2017;13(4):325-330. doi: 10.3988/jcn.2017.13.4.325.
Reference
-
1. Wilschut JC, McElhaney J, Palache B. Influenza. 2006. 2nd ed. Philadelphia, PA: Elsevier Ltd;117–132.2. Park SC, Choeng HJ, Sohn JW, Choi SJ, Eom JS, Woo HJ, Chun BC, Kim WJ. Efficacy of influenza vaccination among chronic ill patients: retrospective case control study. Infect Chemother. 2004. 36:207–212.3. Lee JS, Shin KC, Na BK, Lee JY, Kang C, Kim JH, Park O, Jeong EK, Lee JK, Kwon JW, Park SC, Kim WJ. Influenza surveillance in Korea: establishment and first results of an epidemiological and virological surveillance scheme. Epidemiol Infect. 2007. 135:1117–1123.4. Kee SY, Lee JS, Cheong HJ, Chun BC, Song JY, Choi WS, Jo YM, Seo YB, Kim WJ. Influenza vaccine coverage rates and perceptions on vaccination in South Korea. J Infect. 2007. 55:273–281.5. Song JY, Cheong HJ, Hwang IS, Choi WS, Jo YM, Park DW, Cho GJ, Hwang TG, Kim WJ. Long-term immunogenicity of influenza vaccine among the elderly: Risk factors for poor immune response and persistence. Vaccine. 2010. 28:3929–3935.6. Note for guidance on harmonisation of requirements for influenza vaccines (CPMP/BWP/214/96). European Committee for Proprietary Medicinal Products. c1997. accessed on 17 Aug 2010. London, UK: The Europen Agency for the Evaluation of Medicinal Products;Available at http://www.ema.europa.eu/pdfs/human/bwp/021496en.pdf.7. Guidance for industry: clinical data needed to support the licensure of trivalent inactivated influenza vaccines. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research. c2006. accessed on 17 Aug 2010. Available at http://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/Vaccines/ucm091985.pdf.8. Guidance for Industry: Toxicity Grading Scale for Healthy Adult and Adolescent Volunteers Enrolled in Preventive Vaccine Clinical Trials. U.S. Department of Health and Human Services FDA, Center for Biologics Evaluation and Research. 2007. accessed on 17 Aug 2010. Available at http://www.fda.gov/BiologicsBloodVaccines/GuidanceComplianceRe.9. El Sahly HM, Keitel WA. Clinical data on Fluarix: an inactivated split seasonal influenza vaccine. Expert Rev Vaccines. 2008. 7:713–719.10. Fedson DS. Pandemic influenza and the global vaccine supply. Clin Infect Dis. 2003. 36:1552–1561.11. Cheong HJ. Safety of influenza vaccine. J Korean Med Assoc. 2007. 50:267–273.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Influenza Vaccine
- Safety and Immunogenicity of a New Trivalent Inactivated Split-virus Influenza Vaccine in Healthy Korean Children: A Randomized, Double-blinded, Active-controlled, Phase III Study
- Immunogenicity and Safety of Trivalent Split Influenza Vaccine in Healthy Korean Adults with Low Pre-Existing Antibody Levels: An Open Phase I Trial
- Seasonal influenza and vaccine herd effect
- Comparison of immunogenicity and safety of an influenza vaccine administered concomitantly with a 13-valent pneumococcal conjugate vaccine or 23-valent polysaccharide pneumococcal vaccine in the elderly