J Korean Med Sci.
2009 Jun;24(3):392-397. 10.3346/jkms.2009.24.3.392.
The Effect of Rush Immunotherapy with House Dust Mite in the Production of IL-5 and IFN-gamma from the Peripheral Blood T Cells of Asthmatic Children
- Affiliations
-
- 1Department of Pediatrics, Asthma & Allergy Center, Inje University Sanggye Paik Hospital, Seoul, Korea.
- 2Department of Pediatrics, Childhood Asthma Atopy Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sjhong@amc.seoul.kr
- 3Department of Pediatrics, Hangang Sacred Heart Hospital, Hallym University College of Medicine, Seoul, Korea.
- 4Department of Pediatrics, Ulsan University Hospital, Ulsan, Korea.
- 5Department of Pediatrics, Gangneung Asan Hospital, Gangneung, Korea.
- KMID: 1779153
- DOI: http://doi.org/10.3346/jkms.2009.24.3.392
Abstract
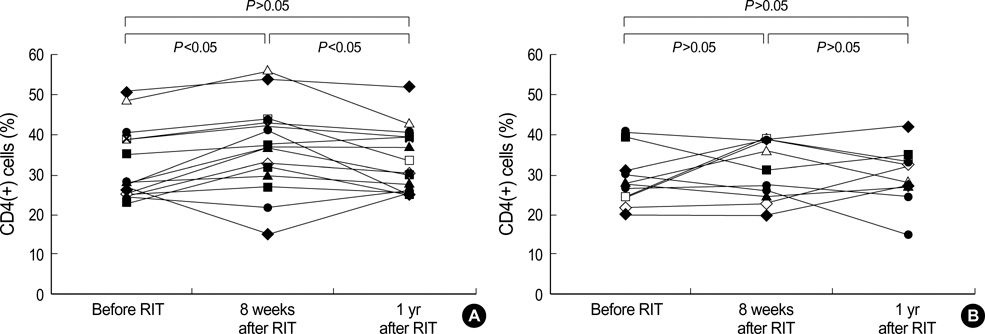
- Although the mechanisms are unclear, rush immunotherapy (RIT) may be effective to treat allergic diseases. We investigated the long-term modifications of cellular immunity as a mechanism of RIT. The RIT group, included 15 house dust mite (HDM)-sensitized asthmatic children, received RIT only with Dermatophagoides farinae (Der f) and Dermatophagoides pteronyssinus (Der p), whereas the control group, consisted of 10 HDM-sensitized asthmatic children, did not receive RIT. The asthma symptom scores and the skin reactivities to Der f were measured. The cellular proliferative responses and intracellular interleukin (IL)-5 and interferon (IFN)-gamma productions from peripheral blood T cells were also measured before, 8 weeks and 1 yr after RIT. The symptom scores, skin reactivity to Der f and cellular proliferative responses to Der f were decreased significantly after 8 weeks and maintained until 1 yr of RIT. The IFN-gamma/IL-5 ratio of the CD3(+) and CD4(+) cells were increased significantly after 8 weeks and maintained until 1 yr of RIT, while there were no changes in the control group. These data indicate that the continuous functional modification from Th2 to Th1 phenotype of the CD4(+) T cells are developed after RIT in the asthmatic children sensitized with HDM.
Keyword
MeSH Terms
Figure
Reference
-
1. Durham SR, Walker SM, Varga EM, Jacobson MR, O'Brien F, Noble W, Till SJ, Hamid QA, Nouri-Aria KT. Long-term clinical efficacy of grass-pollen immunotherapy. N Engl J Med. 1999. 341:468–475.
Article2. Eng PA, Reinhold M, Gnehm HP. Long-term efficacy of preseasonal grass pollen immunotherapy in children. Allergy. 2002. 57:306–312.
Article3. Abramson MJ, Puy RM, Weiner JM. Allergen immunotherapy for asthma. Cochrane Database Syst Rev. 2000. 2:CD001186.
Article4. Lack G, Nelson HS, Amran D, Oshiba A, Jung T, Bradlev KL, Giclas PC, Gelfand EW. Rush immunotherapy results in allergen-specific alterations in lymphocyte function and interferon-γ production in CD4+ T cells. J Allergy Clin Immunol. 1997. 99:530–538.
Article5. Majori M, Caminati A, Corradi M, Brianti E, Scarpa S, Pesci A. T-cell cytokine pattern at three time points during specific immunotherapy for mite-sensitive asthma. Clin Exp Allergy. 2000. 30:341–347.
Article6. Des Roches A, Paradis L, Menardo JL, Bouges S, Daures JP, Bousquet J. Immunotherapy with a standardized Dermatophagoides pteronyssinus extract. VI: specific immunotherapy prevents the onset of new sensitizations in children. J Allergy Clin Immunol. 1997. 99:450–453.7. Pajno GB, Barberio G, De Luca F, Morabito L, Parmiani S. Prevention of new sensitizations in asthmatic children monosensitized to house dust mite by specific immunotherapy: a six-year follow-up study. Clin Exp Allergy. 2001. 31:1392–1397.
Article8. Moller C, Dreborg S, Ferdousi HA, Halken S, Host A, Jacobsen L, Koivikko A, Koller DY, Niggemann B, Norberg LA, Urbanek R, Valvirta E, Wahn U. Pollen immunotherapy reduces the development of asthma in children with seasonal rhinoconjunctivitis (the PAT-study). J Allergy Clin Immunol. 2002. 109:251–256.9. Aalbers R, Van der Gaag R, Van Leeuwen J. Serologic aspects of IgG4 antibodies. I. Prolonged immunization results in an IgG4 antibody response. J Immunol. 1983. 130:722–726.10. Moss RB, Hsu YP, Kwasnicki JM, Sullivan MM, Reid MJ. Isotypic and antigenic restriction of the blocking antibody response to rye grass pollen: correlation of rye group I antigen-specific IgG1 with clinical response. J Allergy Clin Immunol. 1987. 79:387–398.11. Kim BS, Lee SK, Park HS, Hong SJ. The early changes of humoral immune response after rush immunotherapy with Dermatophagoides farinae and Dermatophagoides pteronyssinus in house dust mite sensitive asthmatic children. J Asthma Allergy Clin Immunol. 2001. 21:543–551.12. Creticos PS, Adkinson NF Jr, Kagery-Sobotak A, Proud D, Meier HL, Naclerio RM, Lichtenstein LM, Norman PS. Nasal challenge with ragweed pollen in hay fever patients. Effect of immunotherapy. J Clin Invest. 1985. 76:2247–2253.
Article13. Rak S, Lowhagen O, Venge P. The effect of immunotherapy on bronchial hyperresponsiveness and eosinophil cationic protein in pollen-allergic patients. J Allergy Clin Immunol. 1988. 82:470–480.
Article14. Shim JY, Kim BS, Cho SH, Min KU, Hong SJ. Allergen-specific conventional immunotherapy decreases IgE-receptor-mediated basophil histamine release. Clin Exp Allergy. 2003. 33:52–57.15. Jutel M, Pichler WJ, Skrbic D, Urwyler A, Dahinden C, Muller UR. Bee venom immunotherapy results in decrease of IL-4 and IL-5 and increase of IFN-γ secretion in specific allergen-stimulated T cell cultures. J Immunol. 1995. 154:4187–4194.16. Secrist H, Chelen CJ, Wen Y, Marshall JD, Umetsu DT. Allergen immunotherapy decreases interleukin 4 production in CD4+ T cells from allergic individuals. J Exp Med. 1993. 178:2123–2130.
Article17. Van Bever HP, Vereecke IF, Bridts CH, De Clerck LS, Stevens WJ. Comparison between the in vitro cytokine production of mononuclear cells of young asthmatics with and without immunotherapy (IT). Clin Exp Allergy. 1998. 28:943–949.
Article18. Pène J, Desroches A, Paradis L, Lebel B, Farce M, Nicodemus CF, Yssel H, Bousquet J. Immunotherapy with Fel d 1 peptides decreases IL-4 release by peripheral blood T cells of patients allergic to cats. J Allergy Clin Immunol. 1998. 102:571–578.19. Mamessier E, Birnbaum J, Dupuy P, Vervloet D, Magnan A. Ultra-rush venom immunotherapy induces differential T cell activation and regulatory patterns according to the severity of allergy. Clin Exp Allergy. 2006. 36:704–713.
Article20. Akdis CA, Akdis M, Blesken T, Wymann D, Alkan SS, Muller U, Blaser K. Epitope-specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J Clin Invest. 1996. 98:1676–1683.
Article21. Jutel M, Akdis M, Budak F, Aebischer-Casaulta C, Wrzyszcz M, Blaser K, Akdis CA. IL-10 and TGF-beta cooperate in the regulatory T cell response to mucosal allergens in normal immunity and specific immunotherapy. Eur J Immunol. 2003. 33:1205–1214.22. Akdis CA, Blesken T, Akdis M, Wuthrich B, Blaser K. Role of interleukin 10 in specific immunotherapy. J Clin Invest. 1998. 102:98–106.
Article23. Pereira-Santos MC, Baptista AP, Melo A, Alves RR, Soares RS, Pedro E, Pereira-Barbosa M, Victorino RM, Sousa AE. Expansion of circulatory Foxp3+CD25brightCD4+ T cells during specific venom immunotherapy. Clin Exp Allergy. 2007. 38:291–297.24. American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. Am Rev Respir Dis. 1987. 136:225–244.25. Wasserfallen JB, God K, Schulman KA, Baraniuk JN. Development and validation of rhinoconjunctivitis and asthma symptom score for use as an outcome measure in clinical trials. J Allergy Clin Immunol. 1997. 100:16–22.26. Jung T, Schauer U, HeusSeries C, Neumann C, Rieger C. Detection of intracellular cytokines by flow cytometry. J Immunol Methods. 1993. 159:197–207.
Article27. O'Brien RM, Byron KA, Varigos GA, Thomas WR. House dust mite immunotherapy results in a decrease in Der p 2-specific IFN-γ and IL-4 expression by circulating T lymphocytes. Clin Exp Allergy. 1997. 27:46–51.28. Steinbrink K, Wolfl M, Jonuleit H, Knop J, Enk AH. Induction of tolerance by IL-10-treated dendritic cells. J Immunol. 1997. 159:4772–4780.29. Kim JH, Kim BS, Lee SY, Seo JH, Shim JY, Hong TJ, Hong SJ. Different IL-5 and IFN-γ production from peripheral blood T cell subsets in atopic and nonatopic asthmatic children. J Asthma. 2004. 41:869–871.30. Tamir R, Castracane JM, Rocklin RE. Generation of suppressor cells in atopic patients during immunotherapy that modulate IgE synthesis. J Allergy Clin Immunol. 1987. 79:591–598.
Article31. Lamb JR, Skidmore BJ, Green N, Chiller JM, Feldmann M. Induction of tolerance in influenza virus-immune T lymphocyte clones with synthetic peptides of influenza hemagglutinin. J Exp Med. 1983. 157:1434–1447.
Article32. Gajewski TF, Fitch FW. Anti-proliferative effect of IFN-γ in immune regulation. I. IFN-γ inhibits the proliferation of Th2 but not Th1 murine helper T lymphocyte clones. J Immunol. 1988. 140:4245–4252.33. DeKruff RH, Fan Y, Umetsu DT. IL-4 synthesis by in vivo primed keyhole limpet hemocyanin-specific CD4+ T cells. J Immunol. 1992. 149:3468–3476.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The effect of house dust mite conventional immunotherapy on the production of IL-4 and interferon-gamma from the peripheral blood T cells in asthmatic children
- Demonstration of in vivo and in vitro Changes of T Helper Lymphocytes by Using Cytokine Flow Cytometry
- Production of interleukin 4 and interferon gamma in CD8+ T cells from patients with intrinsic and extrinsic asthma
- The early changes of humoral immune response after rush immunotherapy with Dermatophagoides farinae (D.f) and Dermatophagoides pteronyssinus (D.p) in house dust mite sensitive asthmatic children
- The Effect of Specific Immunotherapy with House Dust Mite AIleI-gen in ChiIdhood Asthma