Yonsei Med J.
2012 May;53(3):587-592. 10.3349/ymj.2012.53.3.587.
Urinary Hypoxia-Inducible Factor-1alpha Levels Are Associated with Histologic Chronicity Changes and Renal Function in Patients with Lupus Nephritis
- Affiliations
-
- 1Department of Neurosurgery, Affiliated Hospital of Hainan Medical College, Hainan, China.
- 2National Hepatobiliary and Enteric Surgery Research Center, Xiangya Hospital, Central South University, Hunan, China.
- 3Department of Nephrology, Hainan Provincial People's Hospital, Hainan, China.
- 4Department of Hepatobiliary Surgery, Hainan Provincial People's Hospital, Hainan, China. LZR59@hotmail.com
- KMID: 1776995
- DOI: http://doi.org/10.3349/ymj.2012.53.3.587
Abstract
- PURPOSE
Tubulointerstitial hypoxia in the kidney is considered a hallmark of injury and a mediator of the progression of tubulointerstitial fibrosis. Hypoxia-inducible factor-1alpha (HIF-1alpha), a master transcription factor in cellular adaptation to hypoxia, regulates a wide variety of genes, some of which are closely associated with tissue fibrosis. The present study set out to characterize urinary HIF-1alpha expressions in patients with lupus nephritis (LN) and to explore whether urinary HIF-1alpha expressions are associated with histologic chronicity changes and renal function.
MATERIALS AND METHODS
Urinary HIF-1alpha levels were measured by enzyme-linked immunosorbent assays in 42 patients with LN and in 30 healthy controls. Activity and chronicity indexes as well as tubular HIF-1alpha expressions were analyzed for each specimen.
RESULTS
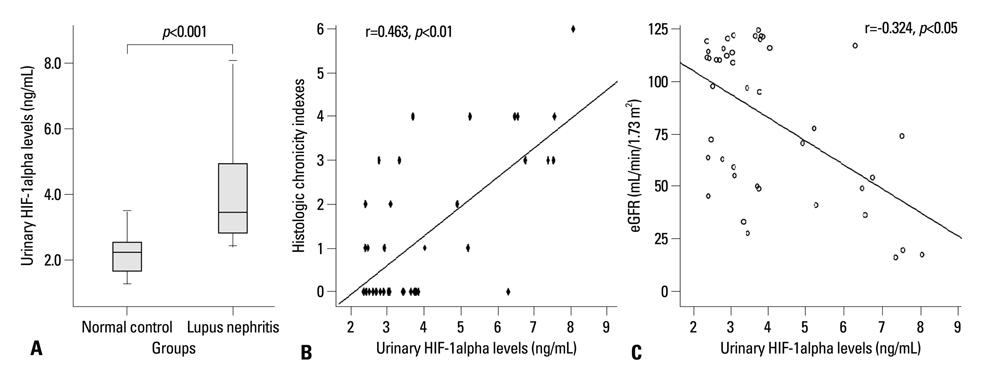
Urinary HIF-1alpha levels were higher in LN patients than in healthy controls (3.977+/-1.696 vs. 2.153+/-0.554 ng/mL, p<0.001) and were associated with histologic chronicity indexes (r=0.463, p<0.01) and eGFR (r=-0.324, p<0.05). However, urinary HIF-1alpha levels showed no correlation with histologic activity indexes, anti-dsDNA, ANA, complement 3 and 4 levels, proteinuria, systemic lupus erythematosis disease activity index, and WHO pathological classification (p>0.05).
CONCLUSION
Urinary HIF-1alpha levels were elevated in LN patients and were associated with histologic chronicity changes and renal function, indicating that HIF-1alpha might contribute to histologic chronicity in LN.
Keyword
MeSH Terms
Figure
Reference
-
1. Bagavant H, Fu SM. Pathogenesis of kidney disease in systemic lupus erythematosus. Curr Opin Rheumatol. 2009. 21:489–494.
Article2. Yu F, Wu LH, Tan Y, Li LH, Wang CL, Wang WK, et al. Tubulointerstitial lesions of patients with lupus nephritis classified by the 2003 International Society of Nephrology and Renal Pathology Society system. Kidney Int. 2010. 77:820–829.
Article3. Rodríguez-Iturbe B, Johnson RJ, Herrera-Acosta J. Tubulointerstitial damage and progression of renal failure. Kidney Int Suppl. 2005. S82–S86.
Article4. Nangaku M. Chronic hypoxia and tubulointerstitial injury: a final common pathway to end-stage renal failure. J Am Soc Nephrol. 2006. 17:17–25.
Article5. Eckardt KU, Bernhardt WM, Weidemann A, Warnecke C, Rosenberger C, Wiesener MS, et al. Role of hypoxia in the pathogenesis of renal disease. Kidney Int Suppl. 2005. S46–S51.
Article6. Heyman SN, Khamaisi M, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia response and the progression of chronic kidney disease. Am J Nephrol. 2008. 28:998–1006.
Article7. Fine LG, Norman JT. Chronic hypoxia as a mechanism of progression of chronic kidney diseases: from hypothesis to novel therapeutics. Kidney Int. 2008. 74:867–872.
Article8. Palm F, Friederich M, Carlsson PO, Hansell P, Teerlink T, Liss P. Reduced nitric oxide in diabetic kidneys due to increased hepatic arginine metabolism: implications for renomedullary oxygen availability. Am J Physiol Renal Physiol. 2008. 294:F30–F37.
Article9. Fine LG, Bandyopadhay D, Norman JT. Is there a common mechanism for the progression of different types of renal diseases other than proteinuria? Towards the unifying theme of chronic hypoxia. Kidney Int Suppl. 2000. 75:S22–S26.
Article10. Haase VH. Hypoxia-inducible factors in the kidney. Am J Physiol Renal Physiol. 2006. 291:F271–F281.
Article11. Nangaku M, Inagi R, Miyata T, Fujita T. Hypoxia and hypoxia-inducible factor in renal disease. Nephron Exp Nephrol. 2008. 110:e1–e7.
Article12. Henze AT, Acker T. Feedback regulators of hypoxia-inducible factors and their role in cancer biology. Cell Cycle. 2010. 9:2749–2763.
Article13. Semenza GL. Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol. 1999. 15:551–578.14. Pugh CW, Chang GW, Cockman M, Epstein AC, Gleadle JM, Maxwell PH, et al. Regulation of gene expression by oxygen levels in mammalian cells. Adv Nephrol Necker Hosp. 1999. 29:191–206.15. Sowter HM, Ratcliffe PJ, Watson P, Greenberg AH, Harris AL. HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res. 2001. 61:6669–6673.16. Maxwell PH, Pugh CW, Ratcliffe PJ. Inducible operation of the erythropoietin 3' enhancer in multiple cell lines: evidence for a widespread oxygen-sensing mechanism. Proc Natl Acad Sci U S A. 1993. 90:2423–2427.
Article17. Wiesener MS, Turley H, Allen WE, Willam C, Eckardt KU, Talks KL, et al. Induction of endothelial PAS domain protein-1 by hypoxia: characterization and comparison with hypoxia-inducible factor-1alpha. Blood. 1998. 92:2260–2268.
Article18. Higgins DF, Biju MP, Akai Y, Wutz A, Johnson RS, Haase VH. Hypoxic induction of Ctgf is directly mediated by Hif-1. Am J Physiol Renal Physiol. 2004. 287:F1223–F1232.19. Higgins DF, Kimura K, Bernhardt WM, Shrimanker N, Akai Y, Hohenstein B, et al. Hypoxia promotes fibrogenesis in vivo via HIF-1 stimulation of epithelial-to-mesenchymal transition. J Clin Invest. 2007. 117:3810–3820.
Article20. Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997. 40:1725.
Article21. Weening JJ, D'Agati VD, Schwartz MM, Seshan SV, Alpers CE, Appel GB, et al. The classification of glomerulonephritis in systemic lupus erythematosus revisited. Kidney Int. 2004. 65:521–530.
Article22. Austin HA 3rd, Muenz LR, Joyce KM, Antonovych TA, Kullick ME, Klippel JH, et al. Prognostic factors in lupus nephritis. Contribution of renal histologic data. Am J Med. 1983. 75:382–391.23. Manotham K, Tanaka T, Matsumoto M, Ohse T, Inagi R, Miyata T, et al. Transdifferentiation of cultured tubular cells induced by hypoxia. Kidney Int. 2004. 65:871–880.
Article24. Norman JT, Clark IM, Garcia PL. Hypoxia promotes fibrogenesis in human renal fibroblasts. Kidney Int. 2000. 58:2351–2366.
Article25. Norman JT, Orphanides C, Garcia P, Fine LG. Hypoxia-induced changes in extracellular matrix metabolism in renal cells. Exp Nephrol. 1999. 7:463–469.
Article26. Kudo Y, Kakinuma Y, Iguchi M, Sato T, Sugiura T, Furihata M, et al. Modification in the von Hippel-Lindau protein is involved in the progression of experimentally induced rat glomerulonephritis. Nephron Exp Nephrol. 2007. 106:e97–e106.
Article27. Rosenberger C, Heyman SN, Rosen S, Shina A, Goldfarb M, Griethe W, et al. Up-regulation of HIF in experimental acute renal failure: evidence for a protective transcriptional response to hypoxia. Kidney Int. 2005. 67:531–542.
Article28. Nangaku M, Fujita T. Activation of the renin-angiotensin system and chronic hypoxia of the kidney. Hypertens Res. 2008. 31:175–184.
Article29. Higgins DF, Kimura K, Iwano M, Haase VH. Hypoxia-inducible factor signaling in the development of tissue fibrosis. Cell Cycle. 2008. 7:1128–1132.
Article30. Haase VH. Pathophysiological Consequences of HIF Activation: HIF as a modulator of fibrosis. Ann N Y Acad Sci. 2009. 1177:57–65.31. Rosenberger C, Rosen S, Shina A, Frei U, Eckardt KU, Flippin LA, et al. Activation of hypoxia-inducible factors ameliorates hypoxic distal tubular injury in the isolated perfused rat kidney. Nephrol Dial Transplant. 2008. 23:3472–3478.
Article32. Nangaku M, Eckardt KU. Hypoxia and the HIF system in kidney disease. J Mol Med (Berl). 2007. 85:1325–1330.
Article33. Hill P, Shukla D, Tran MG, Aragones J, Cook HT, Carmeliet P, et al. Inhibition of hypoxia inducible factor hydroxylases protects against renal ischemia-reperfusion injury. J Am Soc Nephrol. 2008. 19:39–46.
Article34. Tanaka T, Matsumoto M, Inagi R, Miyata T, Kojima I, Ohse T, et al. Induction of protective genes by cobalt ameliorates tubulointerstitial injury in the progressive Thy1 nephritis. Kidney Int. 2005. 68:2714–2725.
Article35. Tanaka T, Kojima I, Ohse T, Ingelfinger JR, Adler S, Fujita T, et al. Cobalt promotes angiogenesis via hypoxia-inducible factor and protects tubulointerstitium in the remnant kidney model. Lab Invest. 2005. 85:1292–1307.
Article36. Song YR, You SJ, Lee YM, Chin HJ, Chae DW, Oh YK, et al. Activation of hypoxia-inducible factor attenuates renal injury in rat remnant kidney. Nephrol Dial Transplant. 2010. 25:77–85.
Article37. Yu X, Fang Y, Ding X, Liu H, Zhu J, Zou J, et al. Transient hypoxia-inducible factor activation in rat renal ablation and reduced fibrosis with L-mimosine. Nephrology (Carlton). 2012. 17:58–67.
Article38. Ohtomo S, Nangaku M, Izuhara Y, Takizawa S, Strihou CY, Miyata T. Cobalt ameliorates renal injury in an obese, hypertensive type 2 diabetes rat model. Nephrol Dial Transplant. 2008. 23:1166–1172.
Article39. Iwano M, Neilson EG. Mechanisms of tubulointerstitial fibrosis. Curr Opin Nephrol Hypertens. 2004. 13:279–284.
Article40. Klahr S, Morrissey J. Progression of chronic renal disease. Am J Kidney Dis. 2003. 41:3 Suppl 1. S3–S7.
Article41. Kimura K, Iwano M, Higgins DF, Yamaguchi Y, Nakatani K, Harada K, et al. Stable expression of HIF-1alpha in tubular epithelial cells promotes interstitial fibrosis. Am J Physiol Renal Physiol. 2008. 295:F1023–F1029.42. Basu RK, Hubchak S, Hayashida T, Runyan CE, Schumacker PT, Schnaper HW. Interdependence of HIF-1α and TGF-β/Smad3 signaling in normoxic and hypoxic renal epithelial cell collagen expression. Am J Physiol Renal Physiol. 2011. 300:F898–F905.
Article43. Wang Z, Tang L, Zhu Q, Yi F, Zhang F, Li PL, et al. Hypoxia-inducible factor-1α contributes to the profibrotic action of angiotensin II in renal medullary interstitial cells. Kidney Int. 2011. 79:300–310.
Article44. Fine LG, Norman JT. Chronic hypoxia as a mechanism of progression of chronic kidney diseases: from hypothesis to novel therapeutics. Kidney Int. 2008. 74:867–872.
Article45. Zhu Q, Wang Z, Xia M, Li PL, Van Tassell BW, Abbate A, et al. Silencing of hypoxia-inducible factor-1α gene attenuated angiotensin II-induced renal injury in Sprague-Dawley rats. Hypertension. 2011. 58:657–664.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Histologic evaluation of activity and chronicity of lupus nephritis and its clinical significance
- A case of Lupus Nephritis
- The Histological Changes and Expression of Hypoxia Inducible Factor-1alpha and Vascular Endothelial Growth Factor according to the Differential Renal Function during Total Ureteral Obstruction in the Rabbit Model
- The Relationship between Expression of Hypoxia Inducible Factor-1alpha or Vascular Endothelial Growth Factor and Histopathological Characteristics in Human Renal Cell Carcinoma
- Hypoxia Inducible Factor-1alpha Directly Induces the Expression of Receptor Activator of Nuclear Factor-kappaB Ligand in MLO-Y4 Osteocytes