J Vet Sci.
2014 Mar;15(1):133-140. 10.4142/jvs.2014.15.1.133.
Regulation of matrix metalloproteinase-9 protein expression by 1alpha,25-(OH)2D3 during osteoclast differentiation
- Affiliations
-
- 1College of Veterinary Medicine Yangzhou University, Yangzhou 225009, China. bianjianchun@sina.com
- 2College of Animal Science and Technology, Yangzhou University, Yangzhou 225009, China.
- 3Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou, 225009, China.
- KMID: 1737619
- DOI: http://doi.org/10.4142/jvs.2014.15.1.133
Abstract
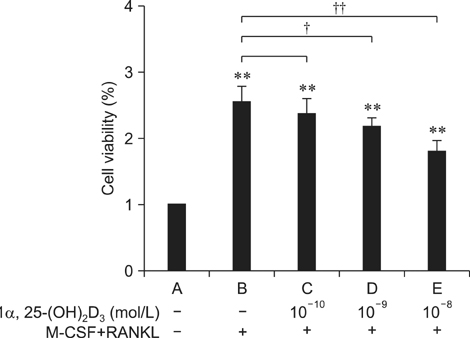
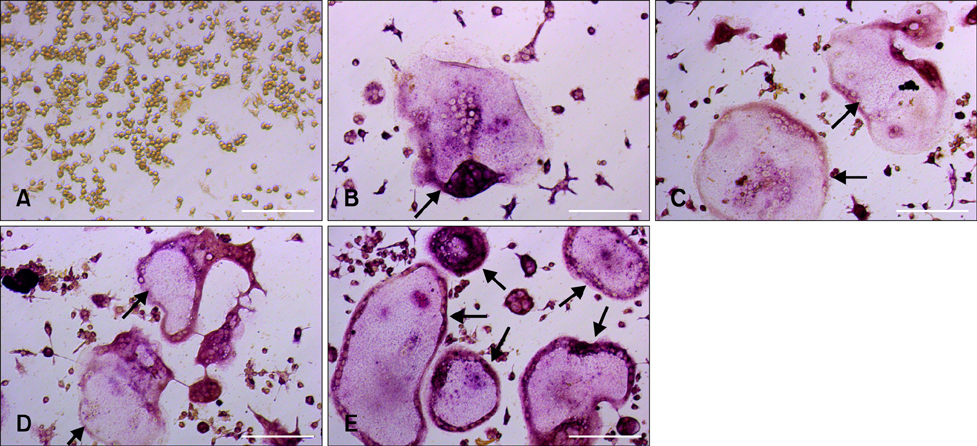
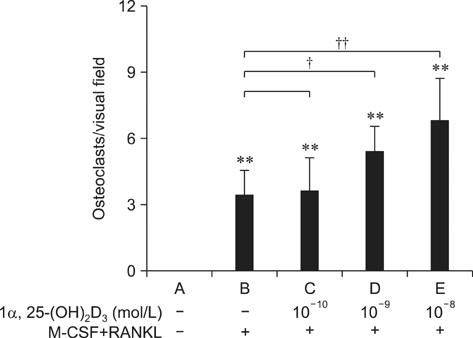
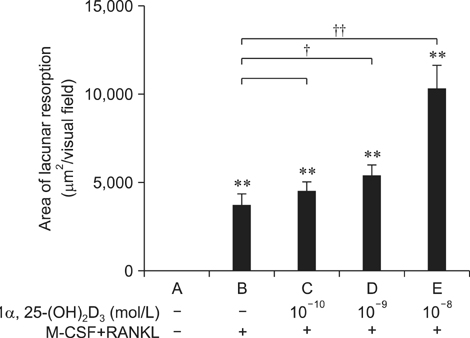
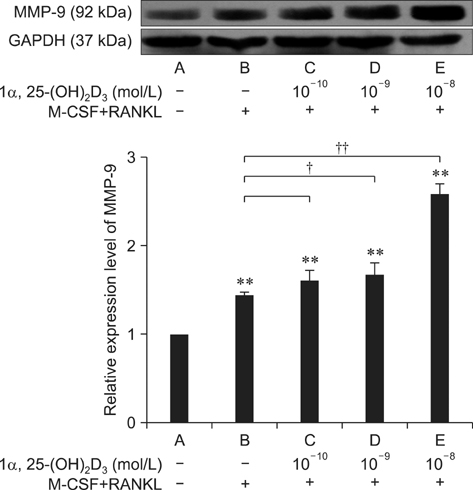
- To investigate 1alpha,25-(OH)2D3 regulation of matrix metalloproteinase-9 (MMP-9) protein expression during osteoclast formation and differentiation, receptor activator of nuclear factor kappaB ligand (RANKL) and macrophage colony-stimulating factor (M-CSF) were administered to induce the differentiation of RAW264.7 cells into osteoclasts. The cells were incubated with different concentrations of 1alpha,25-(OH)2D3 during culturing, and cell proliferation was measured using the methylthiazol tetrazolium method. Osteoclast formation was confirmed using tartrate-resistant acid phosphatase (TRAP) staining and assessing bone lacunar resorption. MMP-9 protein expression levels were measured with Western blotting. We showed that 1alpha,25-(OH)2D3 inhibited RAW264.7 cell proliferation induced by RANKL and M-CSF, increased the numbers of TRAP-positive osteoclasts and their nuclei, enhanced osteoclast bone resorption, and promoted MMP-9 protein expression in a concentration-dependent manner. These findings indicate that 1alpha,25-(OH)2D3 administered at a physiological relevant concentration promoted osteoclast formation and could regulate osteoclast bone metabolism by increasing MMP-9 protein expression during osteoclast differentiation.
Keyword
MeSH Terms
-
Acid Phosphatase/metabolism
Animals
Blotting, Western
Calcitriol/*pharmacology
Calcium Channel Agonists/pharmacology
*Cell Differentiation
Cell Line
Cell Proliferation
Gene Expression Regulation, Enzymologic/*drug effects
Isoenzymes/metabolism
Matrix Metalloproteinase 9/*genetics/metabolism
Mice
Osteoclasts/*cytology/*enzymology
Tetrazolium Salts
Thiazoles
Acid Phosphatase
Calcium Channel Agonists
Calcitriol
Isoenzymes
Tetrazolium Salts
Thiazoles
Matrix Metalloproteinase 9
Figure
Reference
-
1. Anderson PH, Lam NN, Turner AG, Davey RA, Kogawa M, Atkins GJ, Morris HA. The pleiotropic effects of vitamin D in bone. J Steroid Biochem Mol Biol. 2013; 136:190–194.
Article2. Atkins GJ, Anderson PH, Findlay DM, Welldon KJ, Vincent C, Zannettino ACW, O'Loughlin PD, Morris HA. Metabolism of vitamin D3 in human osteoblasts: Evidence for autocrine and paracrine activities of 1α,25-dihydroxyvitamin D3. Bone. 2007; 40:1517–1528.
Article3. Bergh JJ, Xu Y, Farach-carson MC. Osteoprotegerin expression and secretion are regulated by calcium influx through the L-type voltage-sensitive calcium channel. Endocrinology. 2004; 145:426–436.
Article4. Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003; 423:337–342.
Article5. Brooks PJ, Heckler AF, Wei K, Gong SG. M-CSF accelerates orthodontic tooth movement by targeting preosteoclasts in mice. Angle Orthod. 2011; 81:277–283.
Article6. Brown AJ, Zhong M, Finch J, Ritter C, Slatopolsky E. The roles of calcium and 1,25-dihydroxyvitamin D3 in the regulation of vitamin D receptor expression by rat parathyroid glands. Endocrinology. 1995; 136:1419–1425.
Article7. Christensen MHE, Apalset EM, Nordbø Y, Varhaug JE, Mellgren G, Lien EA. 1,25-dihydroxyvitamin D and the vitamin D receptor gene polymorphism Apa1 influence bone mineral density in primary hyperparathyroidism. PLoS One. 2013; 8:e56019.
Article8. Cuetara BLV, Crotti TN, O'Donoghue AJ, McHugh KP. Cloning and characterization of ostoeclast precursors from the RAW264.7 cell line. In Vitro Cell Dev Biol Anim. 2006; 42:182–188.
Article9. Felix R, Cecchini MG, Fleisch H. Macrophage colony stimulating factor restores in vivo bone resorption in the op/op osteopetrotic mouse. Endocrinology. 1990; 127:2592–2594.
Article10. Fujikawa Y, Sabokbar A, Neale SD, Itonaqa I, Torisu T, Athanasou NA. The effect of macrophage-colony stimulating factor and other humoral factors (interleukin-1, -3, -6, and -11, tumor necrosis factor-α, and granulocyte macrophage-colony stimulating factor) on human osteoclast formation from circulating cells. Bone. 2001; 28:261–267.
Article11. Fujisaki K, Tanabe N, Suzuki N, Kawato T, Takeichi O, Tsuzukibashi O, Makimura M, Ito K, Maeno M. Receptor activator of NF-κB ligand induces the expression of carbonic anhydrase II, cathepsin K, and matrix metalloproteinase-9 in osteoclast precursor RAW264.7 cells. Life Sci. 2007; 80:1311–1318.
Article12. Fu YX, Gu JH, Zhang YR, Tong XS, Zhao HY, Yuan Y, Liu XZ, Bian JC, Liu ZP. Osteoprotegerin influences the bone resorption activity of osteoclasts. Int J Mol Med. 2013; 31:1411–1417.
Article13. Hodge JM, Kirkland MA, Nicholson GC. Multiple roles of M-CSF in human osteoclastgenesis. J Cell Biochem. 2007; 102:759–768.14. Inui T, Ishibashi O, Origane Y, Fujimori K, Kokubo T, Nakajima M. Matrix metalloproteinases and lysosomal cysteine proteases in osteoclasts contribute to bone resorption through distinct modes of action. Biochem Biophys Res Commun. 1999; 258:173–178.
Article15. Kaneshita Y, Goda S, Kawamoto T. The effect of matrix metalloproteinase-9 on the differentiation into osteoclast cells on RAW264 cells. Orthod Waves. 2007; 66:122–128.
Article16. Kawatani M, Osada H. Osteoclast-targeting small molecules for the treatment of neoplastic bone metastases. Cancer Sci. 2009; 100:1999–2005.
Article17. Kido S, Inoue D, Hiura K, Javier W, Ito Y, Matsumoto T. Expression of RANK is dependent upon differentiation into the macrophage/osteoclast lineage: induction by 1α,25-dihydroxyvitamin D3 and TPA in a human myelomonocytic cell line, HL60. Bone. 2003; 32:621–629.
Article18. Kudo O, Sabokbar A, Pocock A, Itonaga I, Athanasou NA. Isolation of human osteoclasts formed in vitro: hormonal effects on the bone-resorbing activity of human osteoclasts. Calcif Tissue Int. 2002; 71:539–546.
Article19. Langub MC, Reinhardt TA, Horst RL, Malluche HH, Koszewski NJ. Characterization of vitamin D receptor immunoreactivity in human bone cells. Bone. 2000; 27:383–387.
Article20. Lieben L, Carmeliet G. Vitamin D signaling in osteocytes: effects on bone and mineral homeostasis. Bone. 2013; 54:237–243.
Article21. Matsuzaki K, Udagawa N, Takahashi N, Yamaguchi K, Yasuda H, Shima N, Morinaga T, Toyama Y, Yabe Y, Higashio K, Suda T. Osteoclast differentiation factor (ODF) induces osteoclast-like cell formation in human peripheral blood mononuclear cell cultures. Biochem Biophys Res Commun. 1998; 246:199–204.
Article22. Naruse M, Otsuka E, Naruse M, Ishihara Y, Miyagawa-Tomita S, Hagiwara H. Inhibition of osteoclast formation by 3-methylcholanthrene, a ligand for arylhydrocarbon receptor: suppression of osteoclast differentiation factor in osteogenic cells. Biochem Pharmacol. 2004; 67:119–127.
Article23. Piek E, Sleumer LS, van Someren EP, Heuver L, de Haan JR, de Grijs I, Gilissen C, Hendriks JM, van Ravestein-van Os RI, Bauerschmidt S, Dechering KJ, van Zoelen EJ. Osteo-transcriptomics of human mesenchymal stem cells: ccelerated gene expression and osteoblast differentiation induced by vitamin D reveals c-MYC as an enhancer of BMP2-induced osteogenesis. Bone. 2010; 46:613–627.
Article24. Reynolds JJ. Collagenases and tissue inhibitors of metalloproteinases: a functional balance in tissue degradation. Oral Dis. 1996; 2:70–76.
Article25. Reponen P, Sahlberg C, Munaut C, Thesleff I, Tryggvason K. High expression of 92-kD type IV collagenase (Gelatinase B) in the osteoclast lineage during mouse development. J Cell Biol. 1994; 124:1091–1102.
Article26. Skjødt H, Gallagher JA, Beresford JN, Couch M, Poser JW, Russell RGG. Vitamin D metabolites regulate osteocalcin synthesis and proliferation of human bone cell in vitro. J Endocrinol. 1985; 105:391–396.
Article27. Tang X, Meng H. Osteogenic induction and 1,25-dihydroxyvitamin D3 oppositely regulate the proliferation and expression of RANKL and the vitamin D receptor of human periodontal ligament cells. Arch Oral Biol. 2009; 54:625–633.
Article28. Thomas GP, Baker SUK, Eisman JA, Gardiner EM. Changing RANKL/OPG mRNA expression in differentiating murine primary osteoblasts. J Endocrinol. 2001; 170:451–460.
Article29. Ueda Y, Kanayama M, Yamauchi N, Iio C, Taira Z. Effects of Hachimi-jio-gan extract on intestinal absorption of calcium in ovariectomized mice and stimulation of RANKL-induced osteoclast differentiation of Raw264.7 cells by lipopolysaccharide. J Hard Tissue Biol. 2012; 21:469–476.
Article30. Van Wesenbeeck L, Odgren PR, MacKay CA, D'Angelo M, Safadi FF, Popoff SN, Van Hul W, Marks SC Jr. The osteopetrotic mutation toothless (tl) is a loss-of-function frameshift mutation in the rat Csf1 gene: evidence of a crucial role for CSF-1 in osteoclastogenesis and endochondral ossification. Proc Natl Acad Sci U S A. 2002; 99:14303–14308.
Article31. Vincent C, Kogawa M, Findlay DM, Atkins GJ. The generation of osteoclasts from RAW 264.7 precursors in defined, serum-free conditions. J Bone Miner Metab. 2009; 27:114–119.
Article32. de Vrieze E, Sharif F, Metz JR, Flik G, Richardson MK. Matrix metalloproteinases in osteoclasts of ontogenetic and regenerating zebrafish scales. Bone. 2011; 48:704–712.
Article33. Willems HME, van den Heuvel EGHM, Carmeliet G, Schaafsma A, Klein-Nulend J, Bakker AD. VDR dependent and independent effects of 1,25-dihydroxyvitamin D3 on nitric oxide production by osteoblasts. Steroids. 2012; 77:126–131.
Article34. Woods C, Domenget C, Solari F, Gandrillon O, Lazarides E, Jurdic P. Antagonistic role of vitamin D3 and retinoic acid on the differentiation of chicken hematopoietic macrophages into osteoclast precursor cells. Endocrinology. 1995; 136:85–95.
Article35. Yao Z, Xing L, Qin C, Schwarz EM, Boyce BF. Osteoclast precursor interaction with bone matrix induces osteoclast formation directly by an interleukin-1-mediated autocrine mechanism. J Biol Chem. 2008; 283:9917–9924.
Article36. Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, Tsuda E, Morinaga T, Higashio K, Udagawa N, Takahashi N, Suda T. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci U S A. 1998; 95:3597–3602.
Article37. Yavropoulou MP, Yovos JG. Osteoclastogenesis - current knowledge and future perspectives. J Musculoskelet Neuronal Interact. 2008; 8:204–216.38. Zhu J, Chen C, Xing X, Ye S, Wang X, Wang M, Fu S. Effect of pioglitazone on expression of MMP-9 mRNA in human osteoclast-like cells induced by RANKL and M-CSF. Bone. 2010; 47:S385–S458.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of 17beta-Estradiol and 1,25-Dihydroxyvitamin D3 on Interleukin-6 Production of Periodontal Ligament Cells
- Hyperosmotic Stimulus Down-regulates 1alpha, 25-dihydroxyvitamin D3-induced Osteoclastogenesis by Suppressing the RANKL Expression in a Co-culture System
- In Vitro Effects of 1,25-Dihydroxyvitamin D3 on the Production of Interleukin-1alpha by Ultraviolet B Irradiation in Cultured Human Keratinocyte Cell Line HaCaT Cells
- Deflazacort Increases Osteoclast Formation in Mouse Bone Marrow Culture and the Ratio of RANKL/OPG mRNA Expression in Marrow Stromal Cells
- Regulation of NFATc1 in Osteoclast Differentiation