A single immunization with recombinant rabies virus (ERAG3G) confers complete protection against rabies in mice
- Affiliations
-
- 1Viral Disease Division, Animal and Plant Quarantine Agency, MAFRA, Anyang, Korea. yangdk@korea.kr
- 2The United Graduated School of Veterinary Science, Gifu University, Gifu, Japan.
- KMID: 1730622
- DOI: http://doi.org/10.7774/cevr.2014.3.2.176
Abstract
- PURPOSE
New alternative bait rabies vaccines applicable to pet dogs and wild animals are needed to eradicate rabies in Korea. In this study, recombinant rabies virus, ERAG3G strain was constructed using reverse genetic system and the safety, efficacy and immunogenicity of the ERAG3G strain was evaluated in mice and dogs.
MATERIALS AND METHODS
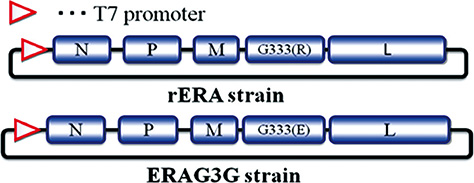
Using the full-length genome mutated amino acid at position 333 of glycoprotein of rabies virus (RABV) and helper plasmids, the ERAG3G strain was rescued in BHK/T7-9 cells successfully. Mice were inoculated with the ERAG3G strain for safety and efficacy. Safety and immunogenicity of the dog inoculated with the ERAG3G strain (1 mL, 10(8.0) FAID50/mL) via intramuscular route was evaluated for 28 days after inoculation.
RESULTS
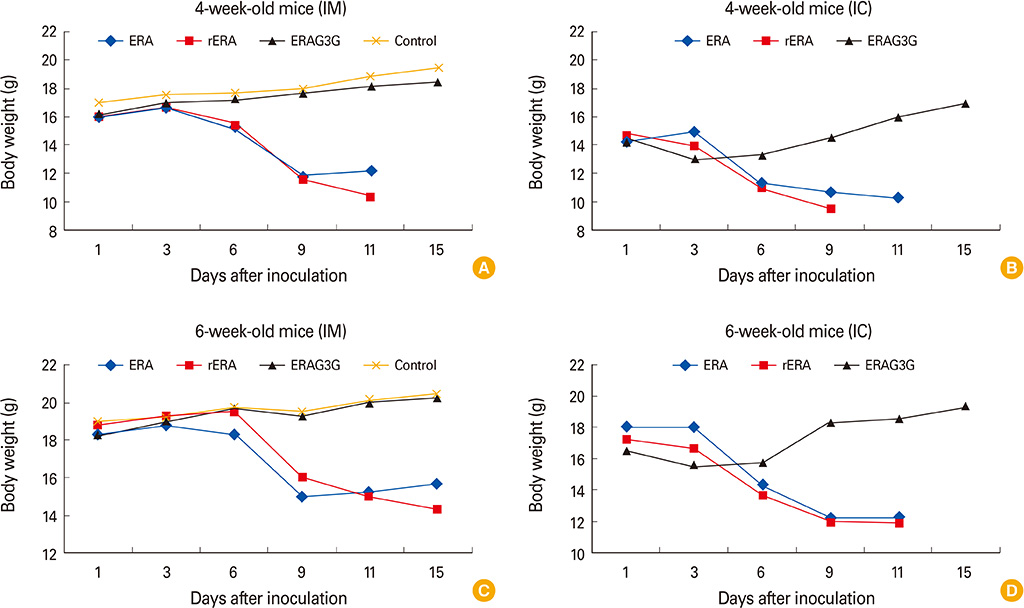
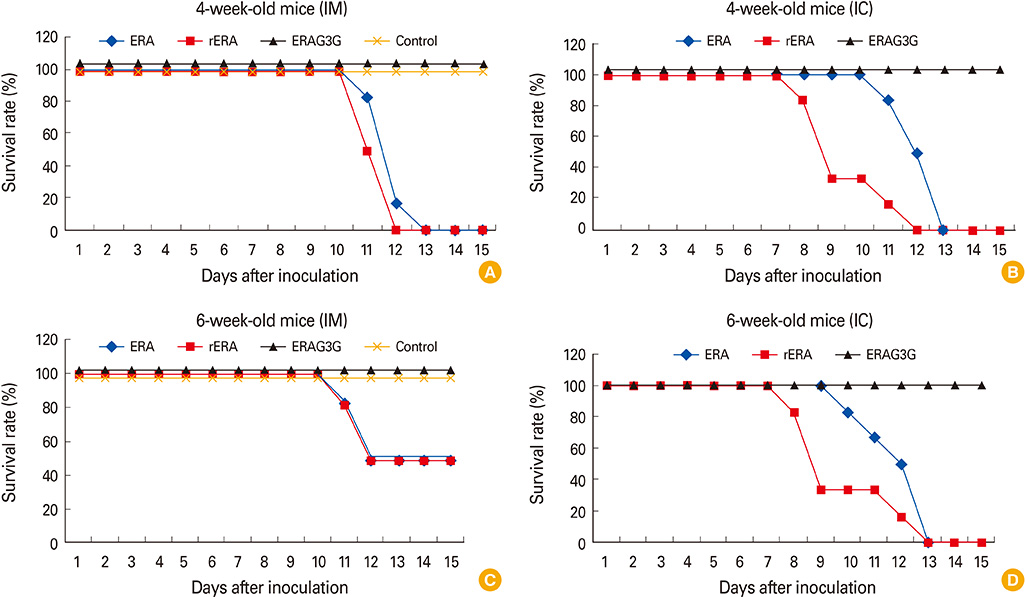
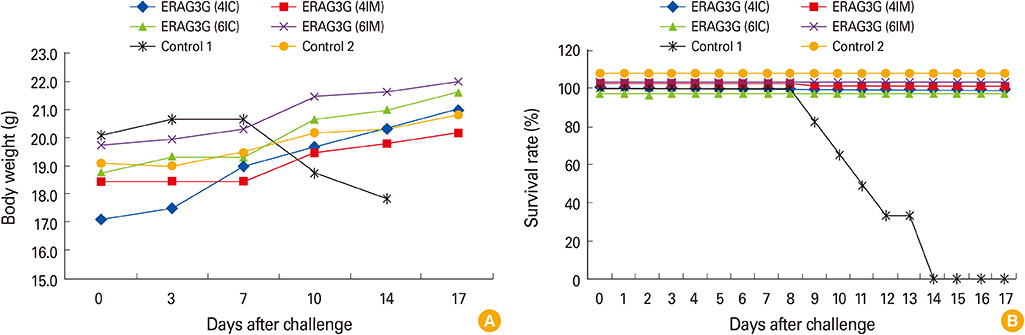
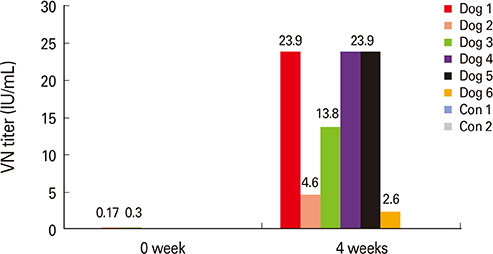
The ERAG3G strain rescued by reverse genetic system was propagated well in the mouse neuroblastoma cells revealing titer of 10(8.5) FAID50/mL and was not pathogenic to 4- or 6-week-old mice that received by intramuscular or intracranical route. Immunization with the ERAG3G strain conferred complete protection from lethal RABV in mice. Dogs inoculated with the vaccine candidate via intramuscular route showed high neutralizing antibody titer ranging from 2.62 to 23.9 IU/mL at 28 days postinoculation.
CONCLUSION
Our findings suggest that the ERAG3G strain plays an important role in inducing protective efficacy in mice and causes to arise anti-rabies neutralizing antibody in dogs.
Keyword
MeSH Terms
Figure
Cited by 4 articles
-
Oral immunization of mice with recombinant rabies vaccine strain (ERAG3G) induces complete protection
Dong-Kun Yang, Ha-Hyun Kim, Sung-Suk Choi, Jong-Taek Kim, Woong-Ho Jeong, Jae-Young Song
Clin Exp Vaccine Res. 2015;4(1):107-113. doi: 10.7774/cevr.2015.4.1.107.A recombinant rabies virus (ERAGS) for use in a bait vaccine for swine
Dong-Kun Yang, Ha-Hyun Kim, Sung-Suk Choi, Seong Heon Lee, In-Soo Cho
Clin Exp Vaccine Res. 2016;5(2):169-174. doi: 10.7774/cevr.2016.5.2.169.Safety and immunogenicity of recombinant rabies virus (ERAGS) in mice and raccoon dogs
Dong-Kun Yang, Ha-Hyun Kim, Sung-Suk Choi, Jong-Tack Kim, Kang-Bok Lee, Seong Heon Lee, In-Soo Cho
Clin Exp Vaccine Res. 2016;5(2):159-168. doi: 10.7774/cevr.2016.5.2.159.Safety and Immunogenicity of a Recombinant Rabies Virus Strain (ERAG3G) in Korean Raccoon Dogs
Dong-Kun Yang, Ha-Hyun Kim, Hyun-Ye Jo, Hee-Won Kim, Sung-Suk Choi, In-Soo Cho
J Bacteriol Virol. 2015;45(3):250-255. doi: 10.4167/jbv.2015.45.3.250.
Reference
-
1. Johnson N, Black C, Smith J, et al. Rabies emergence among foxes in Turkey. J Wildl Dis. 2003; 39:262–270.
Article2. Yang DK, Kim SY, Oh YI, et al. Epidemiological characteristics of rabies in South Korea from January 2004 to March 2011. J Bacteriol Virol. 2011; 41:165–171.
Article3. Orciari LA, Niezgoda M, Hanlon CA, et al. Rapid clearance of SAG-2 rabies virus from dogs after oral vaccination. Vaccine. 2001; 19:4511–4518.
Article4. Faber M, Dietzschold B, Li J. Immunogenicity and safety of recombinant rabies viruses used for oral vaccination of stray dogs and wildlife. Zoonoses Public Health. 2009; 56:262–269.
Article5. Baer GM, Abelseth MK, Debbie JG. Oral vaccination of foxes against rabies. Am J Epidemiol. 1971; 93:487–490.
Article6. Muller TF, Schuster P, Vos AC, Selhorst T, Wenzel UD, Neubert AM. Effect of maternal immunity on the immune response to oral vaccination against rabies in young foxes. Am J Vet Res. 2001; 62:1154–1158.
Article7. Cliquet F, Gurbuxani JP, Pradhan HK, et al. The safety and efficacy of the oral rabies vaccine SAG2 in Indian stray dogs. Vaccine. 2007; 25:3409–3418.
Article8. Fehlner-Gardiner C, Rudd R, Donovan D, Slate D, Kempf L, Badcock J. Comparing ONRAB(R) AND RABORAL V-RG(R) oral rabies vaccine field performance in raccoons and striped skunks, New Brunswick, Canada, and Maine, USA. J Wildl Dis. 2012; 48:157–167.
Article9. Taylor J, Meignier B, Tartaglia J, et al. Biological and immunogenic properties of a canarypox-rabies recombinant, ALVAC-RG (vCP65) in non-avian species. Vaccine. 1995; 13:539–549.
Article10. Rupprecht CE, Blass L, Smith K, et al. Human infection due to recombinant vaccinia-rabies glycoprotein virus. N Engl J Med. 2001; 345:582–586.
Article11. Franka R, Wu X, Jackson FR, et al. Rabies virus pathogenesis in relationship to intervention with inactivated and attenuated rabies vaccines. Vaccine. 2009; 27:7149–7155.
Article12. Inoue K, Shoji Y, Kurane I, Iijima T, Sakai T, Morimoto K. An improved method for recovering rabies virus from cloned cDNA. J Virol Methods. 2003; 107:229–236.
Article13. Ito N, Takayama-Ito M, Yamada K, Hosokawa J, Sugiyama M, Minamoto N. Improved recovery of rabies virus from cloned cDNA using a vaccinia virus-free reverse genetics system. Microbiol Immunol. 2003; 47:613–617.
Article14. Yang DK, Oh YI, Cho SD, et al. Molecular identification of the vaccine strain from the inactivated rabies vaccine. J Bacteriol Virol. 2011; 41:47–54.
Article15. Cliquet F, Aubert M, Sagne L. Development of a fluorescent antibody virus neutralisation test (FAVN test) for the quantitation of rabies-neutralising antibody. J Immunol Methods. 1998; 212:79–87.
Article16. Fu ZF. The rabies situation in Far East Asia. Dev Biol (Basel). 2008; 131:55–61.17. Streicker DG, Altizer SM, Velasco-Villa A, Rupprecht CE. Variable evolutionary routes to host establishment across repeated rabies virus host shifts among bats. Proc Natl Acad Sci U S A. 2012; 109:19715–19720.
Article18. Cliquet F, Aubert M. Elimination of terrestrial rabies in Western European countries. Dev Biol (Basel). 2004; 119:185–204.19. Schnell MJ, Mebatsion T, Conzelmann KK. Infectious rabies viruses from cloned cDNA. EMBO J. 1994; 13:4195–4203.
Article20. Takayama-Ito M, Inoue K, Shoji Y, et al. A highly attenuated rabies virus HEP-Flury strain reverts to virulent by single amino acid substitution to arginine at position 333 in glycoprotein. Virus Res. 2006; 119:208–215.
Article21. Tuffereau C, Leblois H, Benejean J, Coulon P, Lafay F, Flamand A. Arginine or lysine in position 333 of ERA and CVS glycoprotein is necessary for rabies virulence in adult mice. Virology. 1989; 172:206–212.
Article22. Bankovskiy D, Safonov G, Kurilchuk Y. Immunogenicity of the ERA G 333 rabies virus strain in foxes and raccoon dogs. Dev Biol (Basel). 2008; 131:461–466.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Oral immunization of mice with recombinant rabies vaccine strain (ERAG3G) induces complete protection
- Safety and Immunogenicity of a Recombinant Rabies Virus Strain (ERAG3G) in Korean Raccoon Dogs
- Outbreaks and Control of Animal Rabies in Korea
- Immunogenicity and efficacy of a plasmid DNA rabies vaccine incorporating Myd88 as a genetic adjuvant
- General Features and Post-Exposure Prophylaxis of Rabies