Yonsei Med J.
2013 May;54(3):665-671. 10.3349/ymj.2013.54.3.665.
Can Western Based Online Prostate Cancer Risk Calculators Be Used to Predict Prostate Cancer after Prostate Biopsy for the Korean Population?
- Affiliations
-
- 1Department of Urology, Urological Science Institute, Yonsei University College of Medicine, Seoul, Korea. chung646@yuhs.ac
- KMID: 1727880
- DOI: http://doi.org/10.3349/ymj.2013.54.3.665
Abstract
- PURPOSE
To access the predictive value of the European Randomized Screening of Prostate Cancer Risk Calculator (ERSPC-RC) and the Prostate Cancer Prevention Trial Risk Calculator (PCPT-RC) in the Korean population.
MATERIALS AND METHODS
We retrospectively analyzed the data of 517 men who underwent transrectal ultrasound guided prostate biopsy between January 2008 and November 2010. Simple and multiple logistic regression analysis were performed to compare the result of prostate biopsy. Area under the receiver operating characteristics curves (AUC-ROC) and calibration plots were prepared for further analysis to compare the risk calculators and other clinical variables.
RESULTS
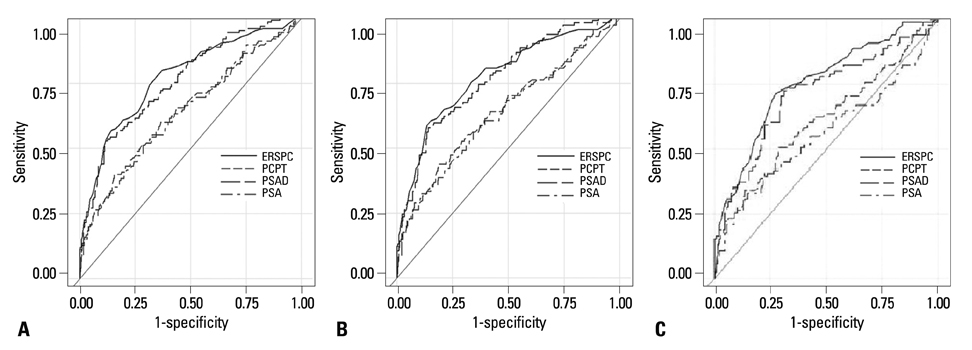
Prostate cancer was diagnosed in 125 (24.1%) men. For prostate cancer prediction, the area under curve (AUC) of the ERSPC-RC was 77.4%. This result was significantly greater than the AUCs of the PCPT-RC and the prostate-specific antigen (PSA) (64.5% and 64.1%, respectively, p<0.01), but not significantly different from the AUC of the PSA density (PSAD) (76.1%, p=0.540). When the results of the calibration plots were compared, the ERSPC-RC plot was more constant than that of PSAD.
CONCLUSION
The ERSPC-RC was better than PCPT-RC and PSA in predicting prostate cancer risk in the present study. However, the difference in performance between the ERSPC-RC and PSAD was not significant. Therefore, the Western based prostate cancer risk calculators are not useful for urologists in predicting prostate cancer in the Korean population.
Keyword
MeSH Terms
Figure
Reference
-
1. Shin HR, Masuyer E, Ferlay J, Curado MP. Asian Contributors to CI5 IX4. Cancer in Asia - Incidence rates based on data in cancer incidence in five continents IX (1998-2002). Asian Pac J Cancer Prev. 2010. 11:Suppl 2. 11–16.2. Lee DH, Jung HB, Chung MS, Lee SH, Chung BH. The change of prostate cancer treatment in Korea: 5 year analysis of a single institution. Yonsei Med J. 2013. 54:87–91.
Article3. Kranse R, Roobol M, Schröder FH. A graphical device to represent the outcomes of a logistic regression analysis. Prostate. 2008. 68:1674–1680.
Article4. van den Bergh RC, Roobol MJ, Wolters T, van Leeuwen PJ, Schröder FH. The Prostate Cancer Prevention Trial and European Randomized Study of Screening for Prostate Cancer risk calculators indicating a positive prostate biopsy: a comparison. BJU Int. 2008. 102:1068–1073.
Article5. Online Prostate Cancer Prevention Trial Risk Calculator. http://prostastecancerinfolink.net/risk-prevention/pcpt-prostate-cancer-risk-calculator/.6. Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003. 349:215–224.
Article7. Online European Randomized Trial of Prostate Cancer. http://www.prostatecancer-riskcalculator.com/.8. Hernandez DJ, Han M, Humphreys EB, Mangold LA, Taneja SS, Childs SJ, et al. Predicting the outcome of prostate biopsy: comparison of a novel logistic regression-based model, the prostate cancer risk calculator, and prostate-specific antigen level alone. BJU Int. 2009. 103:609–614.
Article9. van Vugt HA, Roobol MJ, Busstra M, Kil P, Oomens EH, de Jong IJ, et al. Compliance with biopsy recommendations of a prostate cancer risk calculator. BJU Int. 2012. 109:1480–1488.
Article10. Cavadas V, Osório L, Sabell F, Teves F, Branco F, Silva-Ramos M. Prostate cancer prevention trial and European randomized study of screening for prostate cancer risk calculators: a performance comparison in a contemporary screened cohort. Eur Urol. 2010. 58:551–558.
Article11. Oliveira M, Marques V, Carvalho AP, Santos A. Head-to-head comparison of two online nomograms for prostate biopsy outcome prediction. BJU Int. 2011. 107:1780–1783.
Article12. Shariat SF, Karakiewicz PI, Roehrborn CG, Kattan MW. An updated catalog of prostate cancer predictive tools. Cancer. 2008. 113:3075–3099.
Article13. Eyre SJ, Ankerst DP, Wei JT, Nair PV, Regan MM, Bueti G, et al. Validation in a multiple urology practice cohort of the Prostate Cancer Prevention Trial calculator for predicting prostate cancer detection. J Urol. 2009. 182:2653–2658.
Article14. Karakiewicz PI, Benayoun S, Kattan MW, Perrotte P, Valiquette L, Scardino PT, et al. Development and validation of a nomogram predicting the outcome of prostate biopsy based on patient age, digital rectal examination and serum prostate specific antigen. J Urol. 2005. 173:1930–1934.
Article15. Lee SH, Park KK, Chung MS, Chung BH. Clinical features of familial or hereditary prostate cancer in Korean men: a pilot study. Korean J Urol. 2011. 52:9–12.
Article16. van Vugt HA, Roobol MJ, Kranse R, Määttänen L, Finne P, Hugosson J, et al. Prediction of prostate cancer in unscreened men: external validation of a risk calculator. Eur J Cancer. 2011. 47:903–909.
Article17. Henderson RJ, Eastham JA, Culkin DJ, Kattan MW, Whatley T, Mata J, et al. Prostate-specific antigen (PSA) and PSA density: racial differences in men without prostate cancer. J Natl Cancer Inst. 1997. 89:134–138.
Article18. Swords K, Wallen EM, Pruthi RS. The impact of race on prostate cancer detection and choice of treatment in men undergoing a contemporary extended biopsy approach. Urol Oncol. 2010. 28:280–284.
Article19. Remzi M, Djavan B, Wammack R, Momeni M, Seitz C, Erne B, et al. Can total and transition zone volume of the prostate determine whether to perform a repeat biopsy? Urology. 2003. 61:161–166.
Article20. Ung JO, San Francisco IF, Regan MM, DeWolf WC, Olumi AF. The relationship of prostate gland volume to extended needle biopsy on prostate cancer detection. J Urol. 2003. 169:130–135.
Article21. Rietbergen JB, Kruger AE, Hoedemaeker RF, Bangma CH, Kirkels WJ, Schröder FH. Repeat screening for prostate cancer after 1-year followup in 984 biopsied men: clinical and pathological features of detected cancer. J Urol. 1998. 160(6 Pt 1):2121–2125.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnosis of Prostate Cancer
- Prostate Biopsy: General Consideration and Systematic Biopsy
- A Continuous Increase in Prevalence of state Cancer in Korea and Its Causes
- The Diagnostic Value of Prostate-specific Antigen and the of Routine Laboratory Examination for Early Detection
- How to use new transrectal ultrasound in prostate cancer detection and biopsy for beginners and experts