J Korean Med Sci.
2008 Apr;23(2):185-192. 10.3346/jkms.2008.23.2.185.
The Spectrum of Epstein-Barr Virus-Associated Lymphoproliferative Disease in Korea: Incidence of Disease Entities by Age Groups
- Affiliations
-
- 1Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. yhk0310@skku.edu
- 2Division of Hematologyoncology, Department of Internal Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 1713447
- DOI: http://doi.org/10.3346/jkms.2008.23.2.185
Abstract
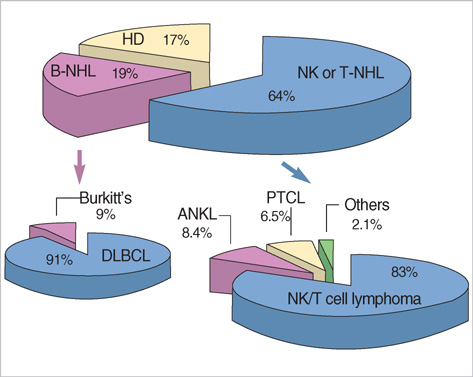
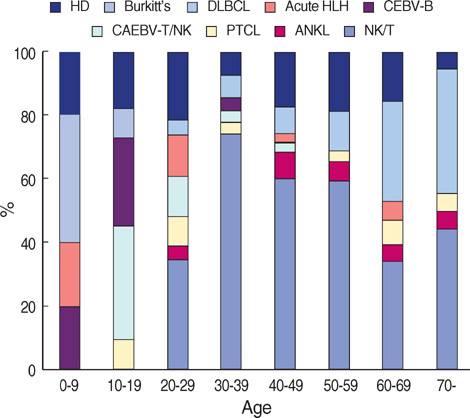
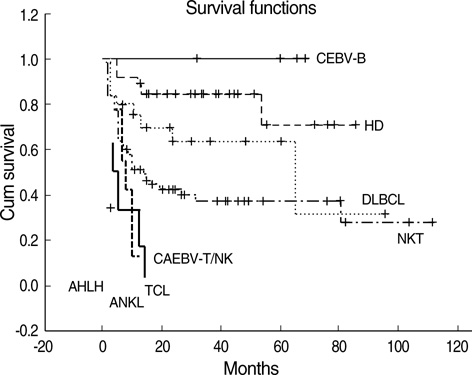
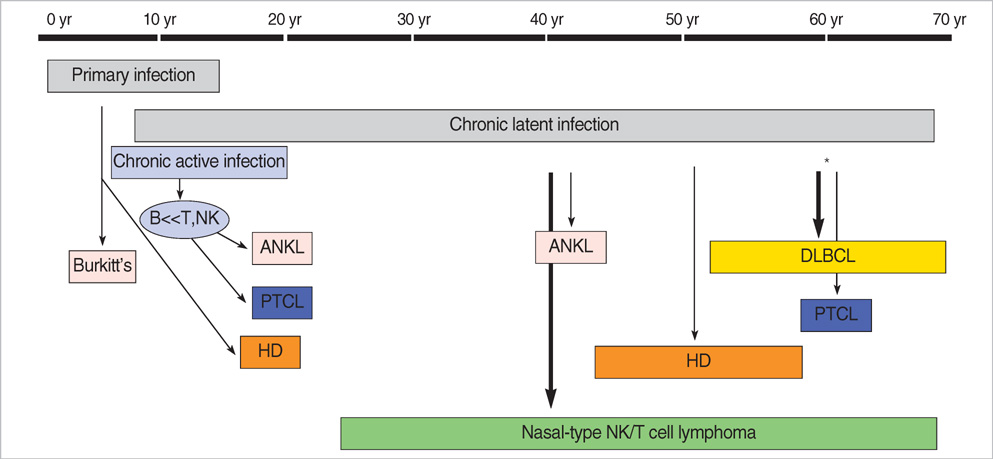
- This study is to identify the spectrum of Epstein-Barr virus (EBV)-positive lymphoproliferative diseases (LPD) and relationships between these diseases in Korea. The EBV status and clinicopathology of 764 patients, including acute EBV-associated hemophagocytic lymphohistiocytosis (EBV-HLH), chronic active EBV (CAEBV) infections, B-LPD arising in chronic latent EBV infection, T & natural killer (NK) cell non-Hodgkin's lymphomas (NHL), B-NHLs, and Hodgkin's lymphomas (HD), were analyzed. T or NK cell NHLs were the most common forms of EBV-positive NHLs (107/167, 64%); among these, nasal-type NK/T cell lymphomas were the most common (89/107, 83%). According to the age, Burkitt's lymphoma was the most common in early childhood; in teenagers, chronic (active) EBV infection-associated LPD was the most common type. The incidence of NK/T cell lymphoma began to increase from the twenties and formed the major type of EBV-associated tumor throughout life. Diffuse large B cell lymphoma formed the major type in the sixties and seventies. In conclusion, primary infections in early childhood are complicated by the development of CAEBV infections that are main predisposing factors for EBV-associated T or NK cell malignancies in young adults. In old patients, decreased immunity associated with old age and environmental cofactors may provoke the development of peripheral T cell lymphoma, unspecified, and diffuse large B cell lymphoma.
MeSH Terms
Figure
Cited by 2 articles
-
CD30-Positive T-Cell Lymphoproliferative Disease of the Oral Mucosa in Children: A Manifestation of Epstein-Barr Virus-Associated T-Lymphoproliferative Disorder
Mineui Hong, Young Hyeh Ko
J Pathol Transl Med. 2015;49(6):525-530. doi: 10.4132/jptm.2015.07.13.Epstein-Barr Virus–Associated Lymphoproliferative Disorders: Review and Update on 2016 WHO Classification
Hyun-Jung Kim, Young Hyeh Ko, Ji Eun Kim, Seung-Sook Lee, Hyekyung Lee, Gyeongsin Park, Jin Ho Paik, Hee Jeong Cha, Yoo-Duk Choi, Jae Ho Han, Jooryung Huh
J Pathol Transl Med. 2017;51(4):352-358. doi: 10.4132/jptm.2017.03.15.
Reference
-
1. Epstein MA, Achong BG, Barr YM. Virus particles in cultured lymphoblasts from Burkitt's lymphoma. Lancet. 1964. 15:702–703.
Article2. Weiss LM, Movahed LA, Warnke RA, Sklar J. Detection of Epstein-Barr viral genomes in Reed-Sternberg cells of Hodgkin's disease. N Engl J Med. 1989. 320:502–506.
Article3. Berg LC, Copenhaver CM, Morrison VA, Gruber SA, Dunn DL, Gajl-Peczalska K, Strickler JG. B-cell lymphoproliferative disorders in solid-organ transplant patients: detection of Epstein-Barr virus by in situ hybridization. Hum Pathol. 1992. 23:159–163.
Article4. MacMahon EM, Glass JD, Hayward SD, Hayward SD, Mann RB, Becker PS, Charache P, McArthur JC, Ambinbder RF. Epstein-Barr virus in AIDS-related primary central nervous system lymphoma. Lancet. 1991. 338:969–973.
Article5. Quintanilla-Martinez L, Franklin JL, Guerrero I, Krenacs L, Naresh KN, Rama-Rao C, Bhatia K, Raffeld M, Magrath IT. Histological and immunophenotypic profile of nasal NK/T cell lymphomas from Peru: high prevalence of p53 overexpression. Hum Pathol. 1999. 30:849–855.
Article6. Chan JK, Sin VC, Wong KF, Ng CS, Tsang WY, Chan CH, Cheung MM, Lau WH. Nonnasal lymphoma expressing the natural killer cell marker CD56: a clinicopathologic study of 49 cases of an uncommon aggressive neoplasm. Blood. 1997. 89:4501–4513.
Article7. Iwatsuki K, Yamamoto T, Tsuji K, Suzuki D, Fujii K, Matsuura H, Oono T. A spectrum of clinical manifestations caused by host immune responses against Epstein-Barr virus infections. Acta Med Okayama. 2004. 58:169–180.8. Quintanilla-Martinez L, Kumar S, Fend F, Reyes E, Teruya-Feldstein J, Kingma DW, Sorbara L, Raffeld M, Straus SE, Jaffe ES. Fulminant EBV (+) T-cell lymphoproliferative disorder following acute/chronic EBV infection: a distinct clinicopathologic syndrome. Blood. 2000. 96:443–451.9. Jaffe ES, Harris NL, Stein H, Vardiman JW. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. 2001. Lyon, France: IARC Press.10. Ko YH, Ree HJ, Kim WS, Choi WH, Moon WS, Kim SW. Clinicopathologic and genotypic study of extranodal nasal-type natural killer/T-cell lymphoma and natural killer precursor lymphoma among Koreans. Cancer. 2000. 89:2106–2116.
Article11. Lee J, Kim WS, Park YH, Park SH, Park KW, Kang JH, Lee SS, Lee SI, Lee SH, Kim K, Jung CW, Ahn YC, Ko YH, Park K. Nasal-type NK/T cell lymphoma: clinical features and treatment outcome. Br J Cancer. 2005. 92:1226–1230.
Article12. Kwong YL, Chan AC, Liang R, Chiang AK, Chim CS, Chan TK, Todd D, Ho FC. CD56+ NK lymphomas: clinicopathological features and prognosis. Br J Haematol. 1997. 97:821–829.13. Ko YH, Kim CW, Park CS, Jang HK, Lee SS, Kim SH, Ree HJ, Lee JD, Kim SW, Huh JR. REAL classification of malignant lymphomas in the Republic of Korea: incidence of recently recognized entities and changes in clinicopathologic features. Cancer. 1998. 83:806–812.14. Imashuku S. Clinical features and treatment strategies of Epstein-Barr virus-associated hemophagocytic lymphohistiocytosis. Crit Rev Oncol Hematol. 2002. 44:259–272.
Article15. Straus SE. The chronic mononucleosis syndrome. J Infect Dis. 1988. 157:405–412.
Article16. Lee J, Suh C, Park YH, Ko YH, Bang SM, Lee JH, Lee DH, Huh J, Oh SY, Kwon HC, Kim HJ, Lee SI, Kim JH, Park J, Oh SJ, Kim K, Jung C, Park K, Kim WS. Extranodal natural killer T-cell lymphoma, nasal-type: a prognostic model from a retrospective multicenter study. J Clin Oncol. 2006. 24:612–618.
Article17. Anderson JR, Armitage JO, Weisenburger DD. Epidemiology of the non-Hodgkin's lymphomas: distributions of the major subtypes differ by geographic locations. Non-Hodgkin's Lymphoma Classification Project. Ann Oncol. 1998. 9:717–720.18. Tsoukas CD, Lambris JD. Expression of EBV/C3d receptors on T cells: Biological significance. Immunol Today. 1993. 14:56–59.
Article19. De Campos-Lima PO, Gavioli R, Zhang QJ, Wallace LE, Dolcetti R, Rowe M, Rickinson AB, Masucci MG. HLA-A11 epitope loss isolates of Epstein-Barr virus from a highly A11+ population. Science. 1993. 260:98–100.
Article20. De Campos-Lima PO, Levitsky V, Brooks J, Lee SP, Hu LF, Rickinson AB, Masucci MG. T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues. J Exp Med. 1994. 179:1297–1305.
Article21. Kanno H, Kojya S, Li T, Ohsawa M, Nakatsuka S, Miyaguchi M, Harabuchi Y, Aozasa K. Low frequency of HLA-A*0201 allele in patients with Epstein-Barr virus-positive nasal lymphomas with polymorphic reticulosis morphology. Int J Cancer. 2000. 87:195–199.
Article22. Suzuki K, Ohshima K, Karube K, Suzumiya J, Ohga S, Ishihara S, Tamura K, Kikuchi M. Clinicopathological states of Epstein-Barr virus-associated T/NK-cell lymphoproliferative disorders (severe chronic active EBV infection) of children and young adults. Int J Oncol. 2004. 24:1165–1174.
Article23. Oh SH, Lee YA, Moon WY, Ko TS, Park YS, Moon HN, Hong CY, Kim DW. Prevalence of Epstein-Barr virus (EBV) antibody in Korean children. J Korean Pediatr Soc. 1994. 37:804–811.24. Oyama T, Ichimura K, Suzuki R, Suzumiya J, Ohshima K, Yatabe Y, Yokoi T, Kojima M, Kamiya Y, Ogura M, Saito H, Morishima Y, Nakamura S. Senile EBV+ B-cell lymphoproliferative disorders: a clinicopathologic study of 22 patients. Am J Surg Pathol. 2003. 27:16–26.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Epstein-Barr Virus Related Polymorphic Posttransplantation Lymphoproliferative Disease in a Patient with Latent Infection of JC Virus
- CD30-Positive T-Cell Lymphoproliferative Disease of the Oral Mucosa in Children: A Manifestation of Epstein-Barr Virus-Associated T-Lymphoproliferative Disorder
- Epstein-Barr Virus Associated Hydroa Vacciniforme-like Eruption
- Epstein-Barr virus-positive T/NK-cell lymphoproliferative disorders
- An Adult Case of Severe Chronic Active Epstein-Barr Virus Infection with T-Cell Lymphoproliferative Disorder