Clin Orthop Surg.
2013 Sep;5(3):216-224. 10.4055/cios.2013.5.3.216.
Role of Surgical Margin on Local Recurrence in High Risk Extremity Osteosarcoma: A Case-Controlled Study
- Affiliations
-
- 1Department of Orthopedic Surgery, Korea Cancer Center Hospital, Seoul, Korea. dgjeon@kcch.re.kr
- KMID: 1705548
- DOI: http://doi.org/10.4055/cios.2013.5.3.216
Abstract
- BACKGROUND
The relationship between surgical margin and local recurrence (LR) in osteosarcoma patients with poor responses to chemotherapy is unclear. Moreover, the incidences of LR according to three different resection planes (bone, soft tissue, and perineurovascular) are not commonly known.
METHODS
We evaluated the incidence of LR in three areas. To assess whether there is a role of surgical margin on LR in patients resistant to preoperative chemotherapy, we designed a case (35 patients with LR) and control (70 patients without LR) study. Controls were matched for age, location, initial tumor volume, and tumor volume change during preoperative chemotherapy.
RESULTS
LR occurred at the soft tissues in 18 cases (51.4%), at the perineurovascular tissues in 11 cases (31.4%), and at the bones in six cases (17.2%). The proportion of inadequate perineurovascular margin was higher in the case group than in the control group (p = 0.01). Within case-control group (105 patients), a correlation between each margin status and LR at corresponding area was found in the bone (p < 0.001) and perineurovascular area (p = 0.001).
CONCLUSIONS
LR is most common in soft tissues. In patients showing similar unfavorable responses to chemotherapy, the losses of perineurovascular fat plane on preoperative magnetic resonance imaging may be a valuable finding in predicting LR.
Keyword
MeSH Terms
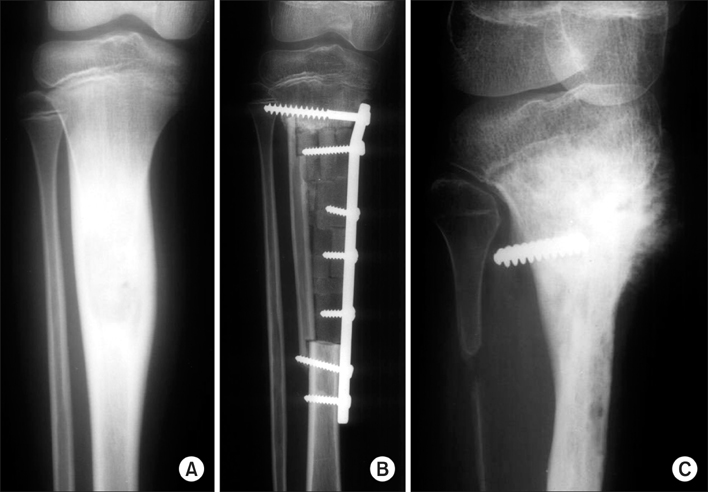
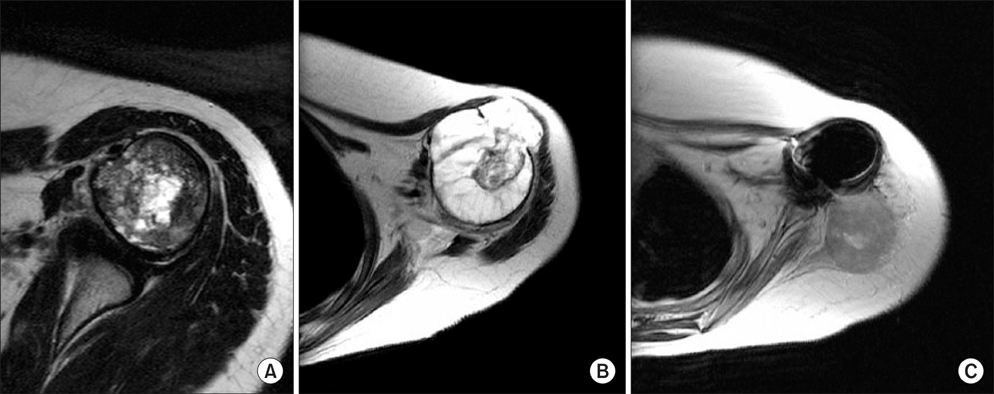
Figure
Reference
-
1. Bacci G, Ferrari S, Lari S, et al. Osteosarcoma of the limb: amputation or limb salvage in patients treated by neoadjuvant chemotherapy. J Bone Joint Surg Br. 2002; 84(1):88–92.2. Bacci G, Forni C, Longhi A, et al. Local recurrence and local control of non-metastatic osteosarcoma of the extremities: a 27-year experience in a single institution. J Surg Oncol. 2007; 96(2):118–123.
Article3. Chi SN, Conklin LS, Qin J, et al. The patterns of relapse in osteosarcoma: the Memorial Sloan-Kettering experience. Pediatr Blood Cancer. 2004; 42(1):46–51.
Article4. Rodriguez-Galindo C, Shah N, McCarville MB, et al. Outcome after local recurrence of osteosarcoma: the St. Jude Children's Research Hospital experience (1970-2000). Cancer. 2004; 100(9):1928–1935.5. Weeden S, Grimer RJ, Cannon SR, Taminiau AH, Uscinska BM. European Osteosarcoma Intergroup. The effect of local recurrence on survival in resected osteosarcoma. Eur J Cancer. 2001; 37(1):39–46.
Article6. Picci P, Sangiorgi L, Bahamonde L, et al. Risk factors for local recurrences after limb-salvage surgery for high-grade osteosarcoma of the extremities. Ann Oncol. 1997; 8(9):899–903.
Article7. Picci P, Sangiorgi L, Rougraff BT, Neff JR, Casadei R, Campanacci M. Relationship of chemotherapy-induced necrosis and surgical margins to local recurrence in osteosarcoma. J Clin Oncol. 1994; 12(12):2699–2705.
Article8. Bacci G, Longhi A, Cesari M, Versari M, Bertoni F. Influence of local recurrence on survival in patients with extremity osteosarcoma treated with neoadjuvant chemotherapy: the experience of a single institution with 44 patients. Cancer. 2006; 106(12):2701–2706.
Article9. Song WS, Jeon DG, Kong CB, et al. Tumor volume increase during preoperative chemotherapy as a novel predictor of local recurrence in extremity osteosarcoma. Ann Surg Oncol. 2011; 18(6):1710–1716.
Article10. Kong CB, Song WS, Cho WH, Oh JM, Jeon DG. Local recurrence has only a small effect on survival in high-risk extremity osteosarcoma. Clin Orthop Relat Res. 2012; 470(5):1482–1490.
Article11. Kim MS, Cho WH, Song WS, Lee SY, Jeon DG. Time dependency of prognostic factors in patients with stage II osteosarcomas. Clin Orthop Relat Res. 2007; 463:157–165.
Article12. Göbel V, Jurgens H, Etspuler G, et al. Prognostic significance of tumor volume in localized Ewing's sarcoma of bone in children and adolescents. J Cancer Res Clin Oncol. 1987; 113(2):187–191.
Article13. Holscher HC, Bloem JL, Vanel D, et al. Osteosarcoma: chemotherapy-induced changes at MR imaging. Radiology. 1992; 182(3):839–844.
Article14. Grimer RJ, Taminiau AM, Cannon SR. Surgical Subcommittee of the European Osteosarcoma Intergroup. Surgical outcomes in osteosarcoma. J Bone Joint Surg Br. 2002; 84(3):395–400.
Article15. Grimer RJ, Sommerville S, Warnock D, et al. Management and outcome after local recurrence of osteosarcoma. Eur J Cancer. 2005; 41(4):578–583.
Article16. Nathan SS, Gorlick R, Bukata S, et al. Treatment algorithm for locally recurrent osteosarcoma based on local disease-free interval and the presence of lung metastasis. Cancer. 2006; 107(7):1607–1616.
Article17. Panicek DM, Hilton S, Schwartz LH. Assessment of neurovascular involvement by malignant musculoskeletal tumors. Sarcoma. 1997; 1(1):61–63.
Article18. Kawaguchi N, Ahmed AR, Matsumoto S, Manabe J, Matsushita Y. The concept of curative margin in surgery for bone and soft tissue sarcoma. Clin Orthop Relat Res. 2004; (419):165–172.
Article19. Andreou D, Bielack SS, Carrle D, et al. The influence of tumor- and treatment-related factors on the development of local recurrence in osteosarcoma after adequate surgery: an analysis of 1355 patients treated on neoadjuvant Cooperative Osteosarcoma Study Group protocols. Ann Oncol. 2011; 22(5):1228–1235.
Article20. Deijkers RL, Bloem RM, Kroon HM, Van Lent JB, Brand R, Taminiau AH. Epidiaphyseal versus other intercalary allografts for tumors of the lower limb. Clin Orthop Relat Res. 2005; 439:151–160.
Article21. Kumta SM, Chow TC, Griffith J, Li CK, Kew J, Leung PC. Classifying the location of osteosarcoma with reference to the epiphyseal plate helps determine the optimal skeletal resection in limb salvage procedures. Arch Orthop Trauma Surg. 1999; 119(5-6):327–331.22. Li J, Wang Z, Guo Z, Chen GJ, Yang M, Pei GX. Irregular osteotomy in limb salvage for juxta-articular osteosarcoma under computer-assisted navigation. J Surg Oncol. 2012; 106(4):411–416.
Article23. Tsuchiya H, Abdel-Wanis ME, Tomita K. Biological reconstruction after excision of juxta-articular osteosarcoma around the knee: a new classification system. Anticancer Res. 2006; 26(1B):447–453.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Osteosarcoma in Maxilla
- A Case Report of Extraskeletal Osteogenic Sarcoma
- Expression and Clinical Significance of Heparanasein Osteosarcoma
- Multiple Margin Positivity of Frozen Section Is an Independent Risk Factor for Local Recurrence in Breast-Conserving Surgery
- Clinical Outcome of Parosteal Osteosarcoma