J Bacteriol Virol.
2007 Sep;37(3):177-191. 10.4167/jbv.2007.37.3.177.
Studies on the Development of Viral Detection Markers for the Quality Control of Blood
- Affiliations
-
- 1Department of Life Sciences, College of Natural Sciences, The University of Suwon, Whasung, Kyonggi-do 445-743, Republic of Korea. kimyh@suwon.ac.kr
- KMID: 1513573
- DOI: http://doi.org/10.4167/jbv.2007.37.3.177
Abstract
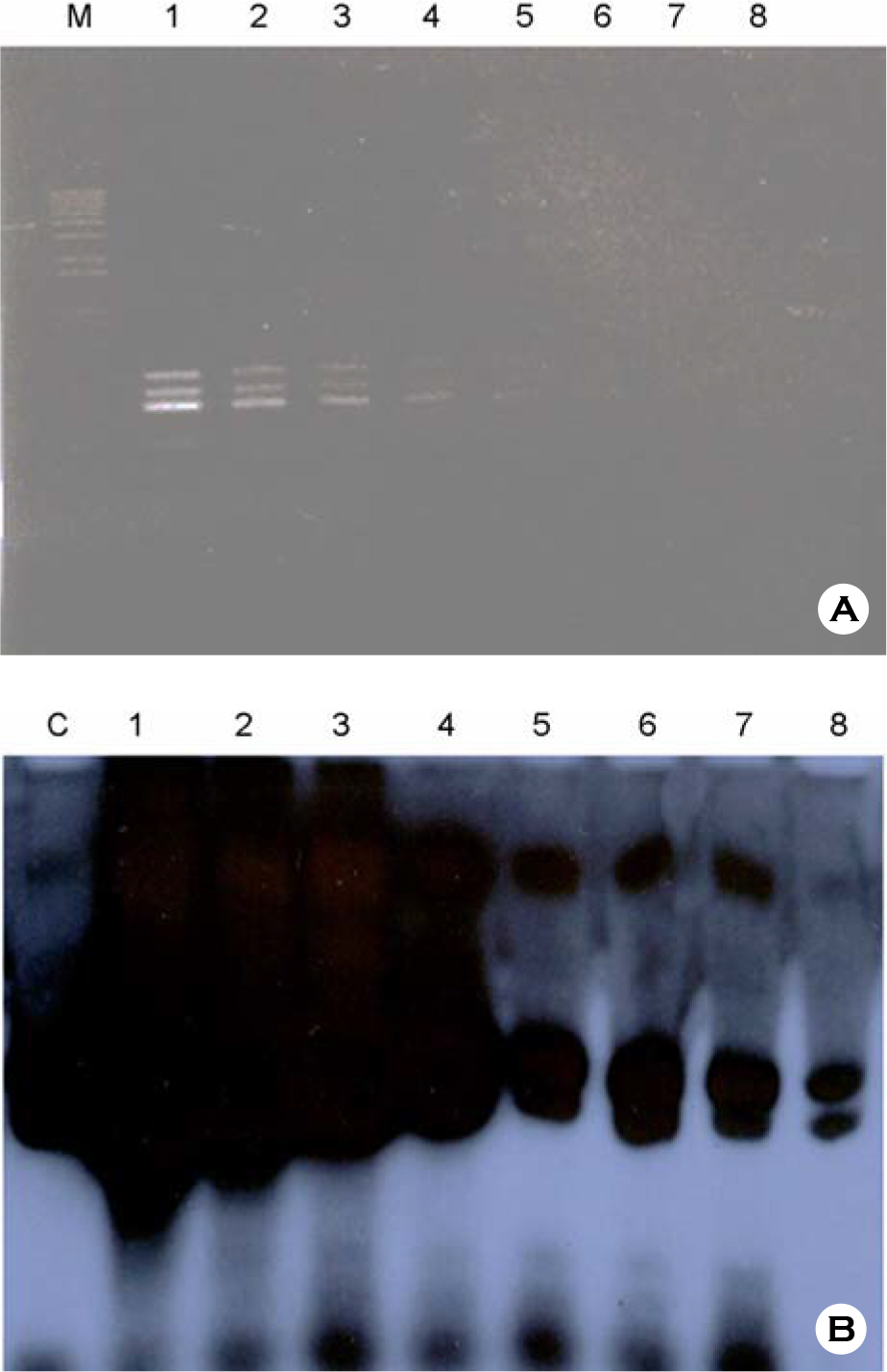
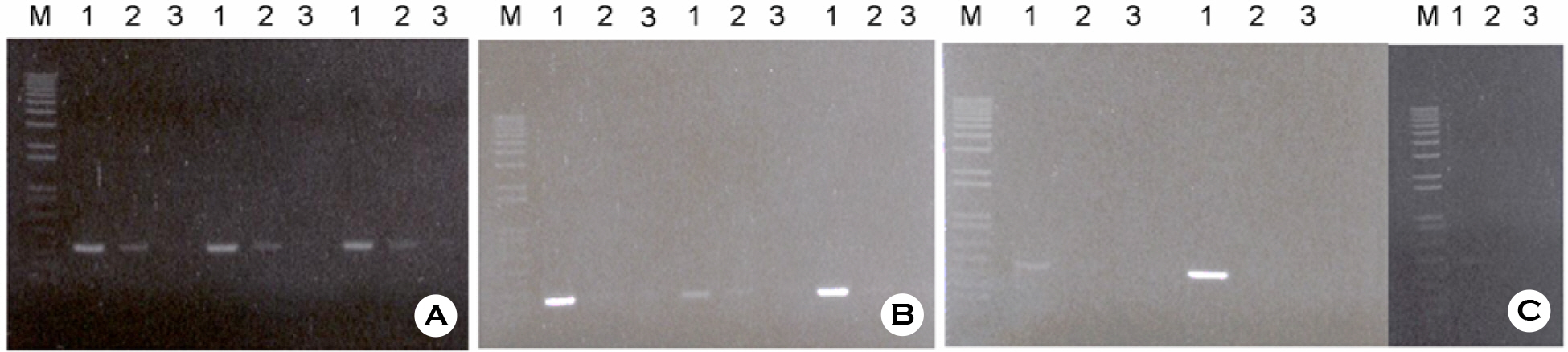
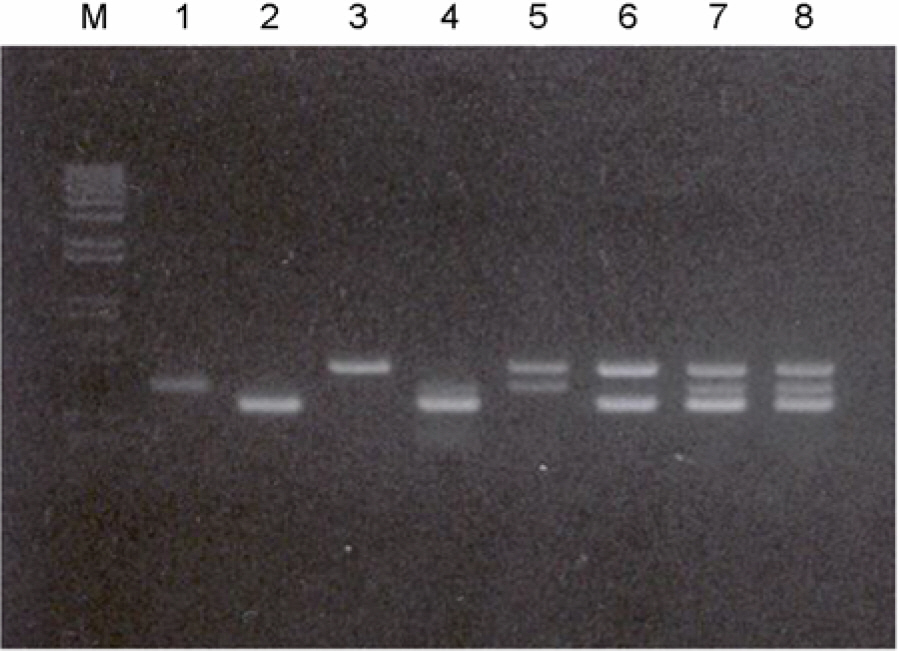
- According to the serological screening methods of antigen-antibody reaction such as ELISA, it has been known that the complete detection of viral infections of HBV, HCV, and HIV-1 viruses in the blood and blood related-products is not much reliable. Therefore, nucleic acid amplification testing methods (NAT) adopted to detect the small quantitative viral nucleic acids could support the basis of using and supplying the blood and its related products safely. This research work is basically designed to describe the simultaneous blood screening system by multiplex or duplex tests for detection of HBV, HCV, and HIV-1 viruses in the blood at one time with low price and labor. It is aimed at easy detection by using the conventional agarose gel electrophoresis. Thus, we tried to detect and identify the viral components in the blood sample according to their different size of PCR products. We decided a set of consensus sequences to recognize each viral DNA fragments after running the multiplex PCR in one tube. This was done by nested RT-PCR using two different RNA viral genomic templates followed by multiplex PCR with addition of viral DNA and their primers after purifying the viral genomic nucleic acids. Those specific primers could be used without any interference to amplify each viral genome in the blood samples. The sensitivities with different viral loads were evaluated on the agarose gel electrophoresis. Three different viral agents in the blood samples could be tested by this multiplex (RT)-PCR with three different primers.
Keyword
MeSH Terms
-
Antigen-Antibody Reactions
Consensus Sequence
DNA, Viral
Electrophoresis, Agar Gel
Enzyme-Linked Immunosorbent Assay
Genome, Viral
HIV-1
Mass Screening
Multiplex Polymerase Chain Reaction
Nucleic Acid Amplification Techniques
Nucleic Acids
Polymerase Chain Reaction
Quality Control*
RNA
Running
Viral Load
Viral Structures
DNA, Viral
Nucleic Acids
RNA
Figure
Cited by 1 articles
-
Naturally Occurring Mutations of Hepatitis B virus and Hepatitis C Virus in Korean Chronic Patients by Distinct CD4 T Cell Responses
Hong Kim, Bum-Joon Kim
J Bacteriol Virol. 2014;44(1):37-43. doi: 10.4167/jbv.2014.44.1.37.
Reference
-
References
1). Allain J-P, Hewitt PE, Tedder RS, Williamson LM. Evidence that anti-HBc but not HBV DNA testing may prevent some HBV transmission by transfusion. Br J Hematol. 107:186–195. 1999.
Article2). Biswas R, Tabor E, Hsia CC, Wright DJ, Laycock MF, Fiebig EW, Peddada I, Smith R, Schreiber GB, Epstein JS, Nemo GJ, Busch MP. Comparative sensitivity of HBV NATs and HBsAg assays for detection of acute HBV infection. Transfusion. 43:788–798. 2003.
Article3). Candotti D, Adu-Darkodie Y, Davies F, Baldrich-Rubio E, Strirups F, Lee H, Allain J-P. AIDS in an HIV-seronegative Ghanaian women with intersubtype A/G recombinant HIV-1 infection. J Med Viro. 62:1–8. 2000.4). Candotti D, Sarkodie F, Allain J-P. Frequent recovery and broad genotype 2 diversity characterize HCV infection in Ghana, West Africa. J Virol. 77:7914–7923. 2003.5). Candotti D, Temple J, Owusu-Ofori S, Allain J-P. Multiplex real-time quantitative RT-PCR assay for hepatitis B virus, hepatitis C virus, and human immunodeficiency virus type 1. J of Virol Methods. 118:39–47. 2004.
Article6). Cuypers HTM, van Dijk R, Viret J-P, Schottstedt V, Lankinen M, Da Silba Cardoso M, Lelie PN. European multi-centre validation study of NucliSens Extractor in combination with HCV Amplicor 2.0 assay for HCV-NAT screening of plasma pools. Biologicals. 27:303–314. 1999.
Article7). Defoort J.-P, Marin M, Casano M, Prato S, Camilla C, Fert V. Simultaneous detection of multiplex-amplified HIV-1 RNA, HCV RNA, and HBV DNA using a flow cytometer microsphere-based hybridization assay. J Clinical Microbiol. 38:1066–1071. 2000.8). Elnifro EM, Ashshi AM, Cooper RJ, Klapper PE. Mutiplex PCR oprimization and application in diagnostic virology. Clinical Microbiol Rev. 13:559–570. 2000.9). FDA. Guideline for the manufacture of in vitro diagnostic products. 1994.10). Giachetti C, Linnen JM, Kolk DP, Dockter J, Gillotte-Tayler K, Park M, Ho M, McCormick MK, Minnes LT, McDonough ST. Highly sensitive multiplex assay for detection of HIV-1 and HCV RNA. J Clinical Microbiol. 40:2408–2419. 2002.11). Gobbers E, Oosterlaken TAM, van Bussel MJA, Melsert R, Kroes ACM, Class ECJ. Efficient extraction of virus DNA by Nuclisens extractor allows sensitive detection of HBV virus by PCR. J of Clinical Microbiol. 39(12):4339–43. 2001.12). Jackson BR, Busch MP, Stramer SL, AuBuchon JP. The cost-effectiveness of NAT for HIV, HCV, and HBV in whole blood donations. Transfusion. 43:721–729. 2003.13). Kolk DP, Dockter J, Linnen J, Ho-Sing-Loy M, Gillotte-Taylor K, McDonough SH, Mimms L, Giachetti C. Significant closure of the human immunodeficiency virus type 1 and hepatitis C virus preseroconversion detection windows with a transcription-mediated-amplification-driven assay. J Clin Microbiol. 40(5):1761–6. 2002.
Article14). Linnen J, Broulik A, Umali A, Kolk D, Dockter J, McDonogh S, Mimms L, Giachetti C. Analytical sensitivity of the TMA-based Procleix Ultrio assay and effect of assay sensitivity on closing the HBV detection window. Transfusion. 42:8S. 2002.15). Meng Q, Wong C, Rangachari A, Tamatsukuri S, Sasaki M, Fiss E, Cheng L, Ramankutty T, Clarke D, Yawata H, Sakakura Y, Hirose T, Impraim C. Automated Multiplex assay system for simultaneous detection of HBV DNA, HCV RNA, HIV-1 RNA. J of Clinical Microbiol. 39(8):2937–45. 2001.16). Mercier B, Burlot L, Ferec C. Simultaneous screening for HBV DNA and HCV RNA genomes in blood donations using a novel TaqMan PCR assay. J Virol Methods. 77:1–9. 1999.
Article17). Mine H, Emura H, Miyamoto M, Tomono T, Minegishi K, Murokaa H, Yamanak R, Yoshikawa A, Nishioka K. Japanese red Cross NAT Research Group. High throughput screening of 16 million serologically negative blood donors for HBV, HCV, HIV-1 by NAT testing with specific and sensitive multiplex reagent in Japan. J of Virol Methods. 112:145–151.18). Miyachi H, Masukawa A, Ohshima T, Hirose T, Impraim C, Ando Y. Automated specific capture of HCV RNA with probes and paramagnetic separation. J of Clinical Microbiol. 38:18–21. 2003,. 2000.19). Ohnuma H, Tanaka T, Yoshikawa A, Murokawa H, Minegishi K, Yamanaka R, Lizuka HY, Miyamoto M, Satoh S, Nakahira S, Tomono T, Murozuka T, Takeda Y, Doi Y, Mine H, Yokoyama S, Hirose T, Nishioka K. Japanese Red Cross NAT Screening Research Group. The first large-scale nucleic acid amplification testing (NAT) of donated blood using multiplex reagent for simultaneous detection of HBV, HCV, and HIV-1 and significance of NAT for HBV. Microbial Immunol. 45(9):667–672. 2001.
Article20). Polz MF, Cavanaugh CM. Bias in template-to product ratios in multitemplate PCR. Appl Environ Microbiol. 64:3724–3730. 1998.21). Roth WK, Weber M, Seifried E. Feasibility and efficacy of routine PCR screening of blood donations for HCV, HBV, and HIV-1 in a blood-bank setting. Lancet. 353:359–363. 1999.22). Schreiber GB, Busch MP, Kleinman SH, Korelitz JJ. The risk of transfusion-transmitted viral infection. The retrovirus epidemiology donor study. New Eng J Med. 334:1685–1690. 1996.23). Soldan K, Barbara JA, Ramsey ME, Hall AJ. Estimation of the risk of HBV, HCV and HIV infectious donations entering the blood supply in England. Vox Sang. 84:274–286. 2003.24). Stramer SL, Waldman KT, Brodsky P, Davis JT. Comparison of HIV-1/HCV nucleic acid test reactivities of diluted and undiluted donations. Transfusion. 40:S94–040A. 2000.25). Vet JA, Majithia AR, Marras SAE, Tyagi S, Dube S, Poiesz BJ, Kramer FR. Multiplex detection of four pathogenic retro-viruses using molecular beacons. PNAS USA. 96:6394–6399. 1999.
Article26). Weinberger KM, Widenmann E, Bohm S, Jilg W. Sensitive and accurate quantitation of HBV DNA using kinetic fluorescence detection system (TaqMan PCR). J Virol Methods. 85:75–82. 2000.27). Widel A, Edmund H, Persson MH, Jonsson M. Transmission of HCV via both erythrocyte and platelet transfusions from a single donor in serological window-phase of HCV. Vox Sang. 71:55–57. 1966.28). Yates S, Penning M, Goudsmit J, Frantzen I, Weije B, Strijp D, Gemen B. Quantitative detection of HBV DNA by real-time NA sequence-based amplification with molecular Beacon detection. J of Clinical Microbiol. 39:3656–3665. 2001.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of Blood Culture and Quality Improvement
- Quality Management of Clinical Chemistry Tests in Blood Center
- Predictive Value of Maternal Serum Markers for Preeclampsia
- Detection Rate of Hepatitis B Virus DNA among Blood Donors showing HBsAg Positivity or Negativity and relatedness with other hepatitis B viral markers
- A Study on ALT levels in Hepatitis Marker Positive Donors -Anti-HBc Positive Rate in Blood Donors-