Anat Cell Biol.
2010 Dec;43(4):340-346. 10.5115/acb.2010.43.4.340.
Ultrastructural analysis of low-threshold mechanoreceptive vibrissa afferent boutons in the cat trigeminal caudal nucleus
- Affiliations
-
- 1Department of Oral Anatomy and Neurobiology, School of Dentistry, Kyungpook National University, Daegu, Korea. ycbae@knu.ac.kr
- 2Department of Oral Physiology, School of Dentistry, Kyungpook National University, Daegu, Korea.
- 3Department of Oral Anatomy and Neurobiology, Graduate School of Dentistry, Osaka University, Suita, Osaka, Japan.
- KMID: 1455349
- DOI: http://doi.org/10.5115/acb.2010.43.4.340
Abstract
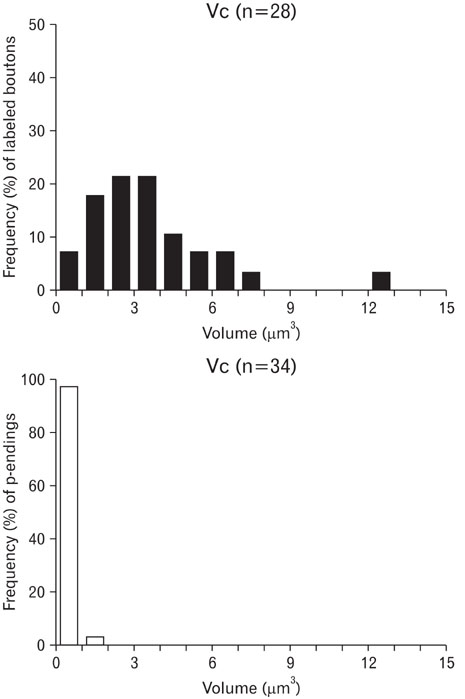
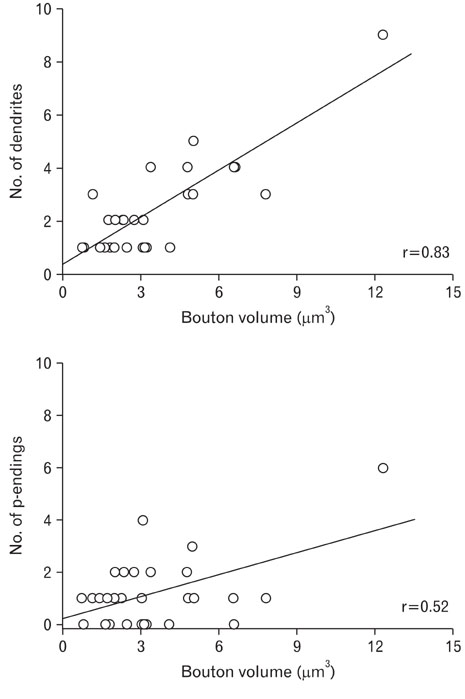
- Ultrastructural parameters related to synaptic release and their correlation with synaptic connectivity were analyzed in the low-threshold mechanoreceptive vibrissa afferent boutons in laminae III and IV of the trigeminal caudal nucleus (Vc). Rapidly adapting vibrissa afferents were intra-axonally labeled, and quantitative ultrastructural analyses with serial sections were performed on the labeled boutons and their presynaptic endings (p-endings). The volume of the labeled boutons was widely distributed from small to large ones (0.8~12.3 microm3), whereas the p-endings were small and uniform in size. The volume of the labeled boutons was positively correlated with the ultrastructural parameters such as mitochondrial volume (correlation coefficient, r=0.96), active zone area (r=0.82) and apposed surface area (r=0.79). Vesicle density (r=-0.18) showed little correlation to the volume of labeled boutons, suggesting that the total vesicle number of a bouton is proportional to its volume. In addition, the bouton volume was positively correlated with the number of p-endings (r=0.52) and with the number of dendrites postsynaptic to the labeled bouton (r=0.83). These findings suggest that low-threshold mechanoreception conveyed through vibrissa afferents is processed in a bouton size-dependent manner in the Vc, which may contribute to the sensory-motor function of laminae III/IV in Vc.
Figure
Reference
-
1. Atwood HL, Marin L. Ultrastructure of synapses with different transmitter-releasing characteristics on motor axon terminals of a crab, Hyas areneas. Cell Tissue Res. 1983. 231:103–115.2. Bae YC, Ahn HJ, Park KP, et al. The synaptic microcircuitry associated with primary afferent terminals in the interpolaris and caudalis of trigeminal sensory nuclear complex. Brain Res. 2005. 1060:118–125.3. Bae YC, Ihn HJ, Park MJ, et al. Identification of signal substances in synapses made between primary afferents and their associated axon terminals in the rat trigeminal sensory nuclei. J Comp Neurol. 2000. 418:299–309.4. Bae YC, Paik SK, Park KP, et al. Quantitative analysis of tooth pulp afferent terminals in the rat brain stem. Neuroreport. 2004. 15:2485–2489.5. Hu JW, Sessle BJ. Comparison of responses of cutaneous nociceptive and nonnociceptive brain stem neurons in trigeminal subnucleus caudalis (medullary dorsal horn) and subnucleus oralis to natural and electrical stimulation of tooth pulp. J Neurophysiol. 1984. 52:39–53.6. Lovick TA. Primary afferent depolarization of tooth pulp afferents by stimulation in nucleus raphe magnus and the adjacent reticular formation in the cat: effects of bicuculline. Neurosci Lett. 1981. 25:173–178.7. Lovick TA. The role of 5-HT, GABA and opioid peptides in presynaptic inhibition of tooth pulp input from the medial brainstem. Brain Res. 1983. 289:135–142.8. Mizuno N, Yasui Y, Nomura S, et al. A light and electron microscopic study of premotor neurons for the trigeminal motor nucleus. J Comp Neurol. 1983. 215:290–298.9. Moon YS, Paik SK, Seo JH, et al. GABA- and glycine-like immunoreactivity in axonal endings presynaptic to the vibrissa afferents in the cat trigeminal interpolar nucleus. Neuroscience. 2008. 152:138–145.10. Nakagawa S, Kurata S, Yoshida A, et al. Ultrastructural observations of synaptic connections of vibrissa afferent terminals in cat principal sensory nucleus and morphometry of related synaptic elements. J Comp Neurol. 1997. 389:12–33.11. Olszewski J. On the anatomical and functional organization of the spinal trigeminal nucleus. J Comp Neurol. 1950. 92:401–413.12. Pierce JP, Lewin GR. An ultrastructural size principle. Neuroscience. 1994. 58:441–446.13. Pierce JP, Mendell LM. Quantitative ultrastructure of Ia boutons in the ventral horn: scaling and positional relationships. J Neurosci. 1993. 13:4748–4763.14. Propst JW, Ko CP. Correlations between active zone ultrastructure and synaptic function studied with freeze-fracture of physiologically identified neuromuscular junctions. J Neurosci. 1987. 7:3654–3664.15. Sessle BJ. The neurobiology of facial and dental pain: present knowledge, future directions. J Dent Res. 1987. 66:962–981.16. Sessle BJ. Acute and chronic craniofacial pain: brainstem mechanisms of nociceptive transmission and neuroplasticity, and their clinical correlates. Crit Rev Oral Biol Med. 2000. 11:57–91.17. Shigenaga Y, Matano S, Kusuyama M, Sakai A. Cortical neurons responding to electrical stimulations of the rat's incisor pulp. Brain Res. 1974. 67:153–156.18. Shigenaga Y, Matano S, Okada K, Sakai A. The effects of tooth pulp stimulation in the thalamus and hypothalamus of the rat. Brain Res. 1973. 63:402–407.19. Shigenaga Y, Nakatani Z, Nishimori T, Suemune S, Kuroda R, Matano S. The cells of origin of cat trigeminothalamic projections: especially in the caudal medulla. Brain Res. 1983. 277:201–222.20. Shigenaga Y, Sakai A, Okada K. Effects of tooth pulp stimulation in trigeminal nucleus caudalis and adjacent reticular formation in rat. Brain Res. 1976. 103:400–406.21. Shigenaga Y, Yoshida A, Mitsuhiro Y, Tsuru K, Doe K. Morphological and functional properties of trigeminal nucleus oralis neurons projecting to the trigeminal motor nucleus of the cat. Brain Res. 1988. 461:143–149.22. Yeow MB, Peterson EH. Active zone organization and vesicle content scale with bouton size at a vertebrate central synapse. J Comp Neurol. 1991. 307:475–486.23. Yoshida A, Fukami H, Nagase Y, et al. Quantitative analysis of synaptic contacts made between functionally identified oralis neurons and trigeminal motoneurons in cats. J Neurosci. 2001. 21:6298–6307.24. Zhang LF, Moritani M, Honma S, Yoshida A, Shigenaga Y. Quantitative ultrastructure of slowly adapting lingual afferent terminals in the principal and oral nuclei in the cat. Synapse. 2001. 41:96–111.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Quantitative Ultrastructural Analysis of Periodontal Afferent Terminals in the Trigeminal Motor Nucleus
- Ultrastructural analysis of glutamate immunoreactive primary afferent terminals in the trigeminal nucleus principalis and trigeminal nucleus oralis of the rat
- Synaptic connection and GABA-immunoreactivity of periodontal afferent terminals and their presynaptic endings in the trigeminal motor nucleus of the rat
- Synaptic connectivity and ultrastructrue of the masseteric muscle spindle afferent trigeminals according to geometric location in the trigeminal motor nucleus of the cat
- Quantitative Ultrastructural Analysis of Endings Presynaptic to the Tooth Pulp Afferent Terminals in the Trigeminal Oral Nucleus