J Bacteriol Virol.
2012 Mar;42(1):63-68. 10.4167/jbv.2012.42.1.63.
ASK1 is Involved in EBV LMP1-induced NF-kappaB Activation
- Affiliations
-
- 1Department of Life Science, Kyungwon University, Seongnam-Si, Kyeonggi-Do, Korea. songyj@kyungwon.ac.kr
- KMID: 1434774
- DOI: http://doi.org/10.4167/jbv.2012.42.1.63
Abstract
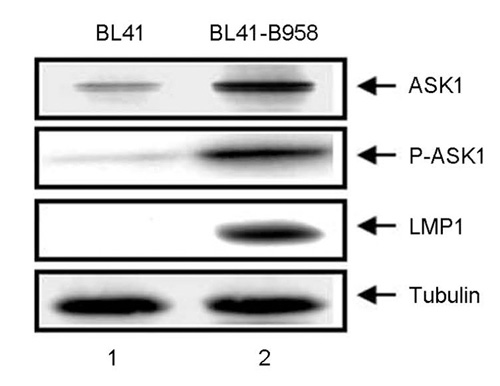
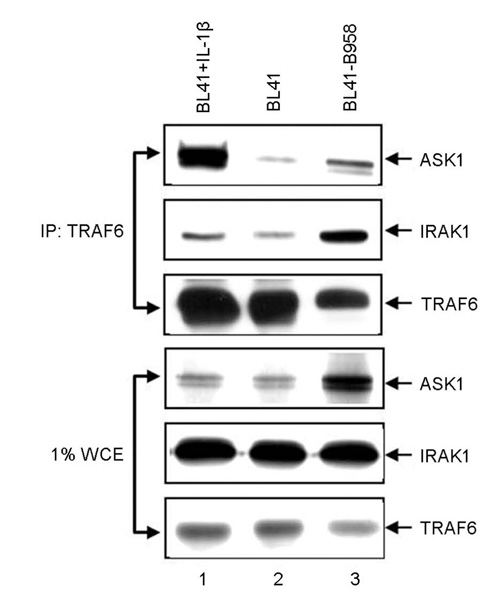
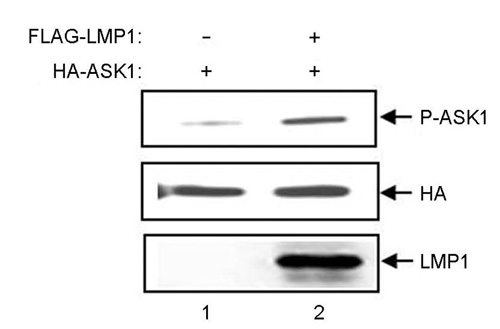
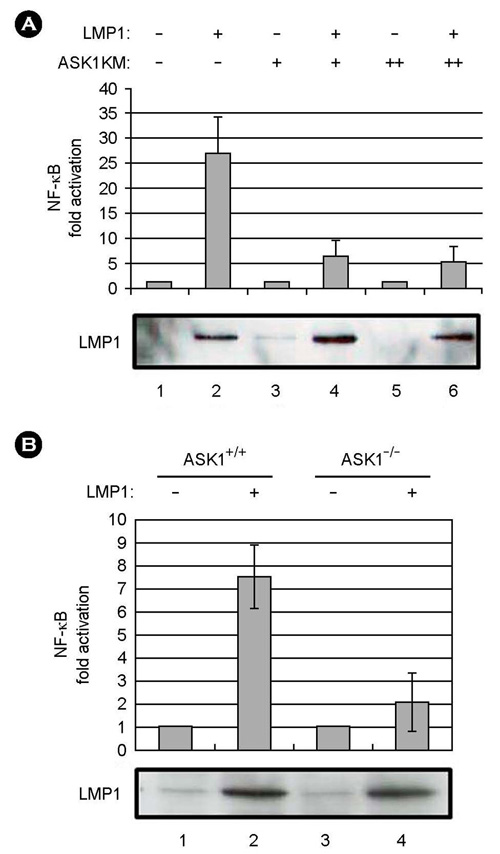
- Epstein-Barr virus (EBV) latent infection transforms B lymphocytes into proliferating lymphoblastoid cell lines (LCLs). EBV latent infection membrane protein 1 (LMP1) is required for EBV-mediated B lymphocyte transformation, and LMP1-induced NF-kappaB activation is essential for LCL survival. Previously, it was reported that the level of reactive oxygen species (ROS) and the expression of apoptosis signal-regulating kinase 1 (ASK1) are elevated in EBV-positive Burkitt's lymphoma (BL) cells, the potential role of ASK1 in LMP1-induced NF-kappaB activation was thus investigated in this study. In EBV-positive BL cells, ASK1 was highly expressed and activated. In addition, TRAF6-ASK1 interaction was significantly increased in EBV-positive BL cells. Interestingly, the expression of LMP1 alone facilitated ASK1 activation. The expression of a dominant negative ASK1 mutant (ASK1KM) strongly blocked LMP1-induced NF-kappaB activation. Furthermore, LMP1-induced NF-kappaB activation was significantly reduced in ASK1 knock out (ASK1-/-) mouse embryonic fibroblasts (MEFs). Taken together, these results demonstrate that ASK1 is activated by LMP1 and is critical for LMP1-induced NF-kappaB activation.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Herpesvirus-encoded Deubiquitinating Proteases and Their Roles in Regulating Immune Signaling Pathways
Ki Mun Kwon, Jin-Hyun Ahn
J Bacteriol Virol. 2013;43(4):244-252. doi: 10.4167/jbv.2013.43.4.244.
Reference
-
1. Kieff ED, Rickinson AB. Knipe DM, Howley PM, editors. Epstein-Barr Virus and Its Replication. Fields Virology. 2007. Philadelphia: Lippincott, Williams, and Wilkins;2603–2655.2. Rickinson AB, Kieff ED. Knipe DM, Howley PM, editors. Epstein-Barr Virus. Fields Virology. 2007. Fifth ed. Philadelphia, Pa: Lippincott, Williams, and Wilkins;2655–2700.3. Li HP, Chang YS. Epstein-Barr virus latent membrane protein 1: structure and functions. J Biomed Sci. 2003. 10:490–504.
Article4. Mitchell T, Sugden B. Stimulation of NF-kappa B-mediated transcription by mutant derivatives of the latent membrane protein of Epstein-Barr virus. J Virol. 1995. 69:2968–2976.
Article5. Huen DS, Henderson SA, Croom-Carter D, Rowe M. The Epstein-Barr virus latent membrane protein-1 (LMP1) mediates activation of NF-kappa B and cell surface phenotype via two effector regions in its carboxy-terminal cytoplasmic domain. Oncogene. 1995. 10:549–560.6. Devergne O, Cahir McFarland ED, Mosialos G, Izumi KM, Ware CF, Kieff E. Role of the TRAF binding site and NF-kappaB activation in Epstein-Barr virus latent membrane protein 1-induced cell gene expression. J Virol. 1998. 72:7900–7908.
Article7. Devergne O, Hatzivassiliou E, Izumi KM, Kaye KM, Kleijnen MF, Kieff E, et al. Association of TRAF1, TRAF2, and TRAF3 with an Epstein-Barr virus LMP1 domain important for B-lymphocyte transformation: role in NF-kappaB activation. Mol Cell Biol. 1996. 16:7098–7108.
Article8. Mosialos G, Birkenbach M, Yalamanchili R, VanArsdale T, Ware C, Kieff E. The Epstein-Barr virus transforming protein LMP1 engages signaling proteins for the tumor necrosis factor receptor family. Cell. 1995. 80:389–399.
Article9. Kaye KM, Devergne O, Harada JN, Izumi KM, Yalamanchili R, Kieff E, et al. Tumor necrosis factor receptor associated factor 2 is a mediator of NF-kappa B activation by latent infection membrane protein 1, the Epstein-Barr virus transforming protein. Proc Natl Acad Sci U S A. 1996. 93:11085–11090.
Article10. Kaye KM, Izumi KM, Mosialos G, Kieff E. The Epstein-Barr virus LMP1 cytoplasmic carboxy terminus is essential for B-lymphocyte transformation; fibroblast cocultivation complements a critical function within the terminal 155 residues. J Virol. 1995. 69:675–683.
Article11. Izumi KM, Cahir McFarland ED, Riley EA, Rizzo D, Chen Y, Kieff E. The residues between the two transformation effector sites of Epstein-Barr virus latent membrane protein 1 are not critical for B-lymphocyte growth transformation. J Virol. 1999. 73:9908–9916.
Article12. Cahir-McFarland ED, Davidson DM, Schauer SL, Duong J, Kieff E. NF-kappa B inhibition causes spon taneous apoptosis in Epstein-Barr virus-transformed lymphoblastoid cells. Proc Natl Acad Sci U S A. 2000. 97:6055–6060.
Article13. Cahir-McFarland ED, Carter K, Rosenwald A, Giltnane JM, Henrickson SE, Staudt LM, et al. Role of NF-kappa B in cell survival and transcription of latent membrane protein 1-expressing or Epstein-Barr virus latency III-infected cells. J Virol. 2004. 78:4108–4119.
Article14. Ghosh S, Karin M. Missing pieces in the NF-kappaB puzzle. Cell. 2002. 109:S81–S96.15. Hayden MS, Ghosh S. Signaling to NF-kappaB. Genes Dev. 2004. 18:2195–2224.16. Matsuzawa A, Saegusa K, Noguchi T, Sadamitsu C, Nishitoh H, Nagai S, et al. ROS-dependent activation of the TRAF6-ASK1-p38 pathway is selectively required for TLR4-mediated innate immunity. Nat Immunol. 2005. 6:587–592.
Article17. Eliopoulos AG, Gallagher NJ, Blake SM, Dawson CW, Young LS. Activation of the p38 mitogen-activated protein kinase pathway by Epstein-Barr virus-encoded latent membrane protein 1 coregulates interleukin-6 and interleukin-8 production. J Biol Chem. 1999. 274:16085–16096.
Article18. Eliopoulos AG, Blake SM, Floettmann JE, Rowe M, Young LS. Epstein-Barr virus-encoded latent membrane protein 1 activates the JNK pathway through its extreme C terminus via a mechanism involving TRADD and TRAF2. J Virol. 1999. 73:1023–1035.
Article19. Eliopoulos AG, Stack M, Dawson CW, Kaye KM, Hodgkin L, Sihota S, et al. Epstein-Barr virus-encoded LMP1 and CD40 mediate IL-6 production in epithelial cells via an NF-kappaB pathway involving TNF receptor-associated factors. Oncogene. 1997. 14:2899–2916.
Article20. Eliopoulos AG, Dawson CW, Mosialos G, Floettmann JE, Rowe M, Armitage RJ, et al. CD40-induced growth inhibition in epithelial cells is mimicked by Epstein-Barr Virus-encoded LMP1: involvement of TRAF3 as a common mediator. Oncogene. 1996. 13:2243–2254.21. Kieser A, Kilger E, Gires O, Ueffing M, Kolch W, Hammerschmidt W. Epstein-Barr virus latent membrane protein-1 triggers AP-1 activity via the c-Jun N-terminal kinase cascade. EMBO J. 1997. 16:6478–6485.
Article22. Matsukawa J, Matsuzawa A, Takeda K, Ichijo H. The ASK1-MAP kinase cascades in mammalian stress response. J Biochem. 2004. 136:261–265.
Article23. Takeda K, Matsuzawa A, Nishitoh H, Tobiume K, Kishida S, Ninomiya-Tsuji J, et al. Involvement of ASK1 in Ca2+-induced p38 MAP kinase activation. EMBO Rep. 2004. 5:161–166.
Article24. Takeda K, Matsuzawa A, Nishitoh H, Ichijo H. Roles of MAPKKK ASK1 in stress-induced cell death. Cell Struct Funct. 2003. 28:23–29.
Article25. Cerimele F, Battle T, Lynch R, Frank DA, Murad E, Cohen C, et al. Reactive oxygen signaling and MAPK activation distinguish Epstein-Barr Virus (EBV)-positive versus EBV-negative Burkitt's lymphoma. Proc Natl Acad Sci U S A. 2005. 102:175–179.
Article26. Stinski MF. Synthesis of proteins and glycoproteins in cells infected with human cytomegalovirus. J Virol. 1977. 23:751–767.
Article27. Song YJ, Izumi KM, Shinners NP, Gewurz BE, Kieff E. IRF7 activation by Epstein-Barr virus latent membrane protein 1 requires localization at activation sites and TRAF6, but not TRAF2 or TRAF3. Proc Natl Acad Sci U S A. 2008. 105:18448–18453.
Article28. Bari W, Song YJ, Yoon SS. Suppressed induction of proinflammatory cytokines by a unique metabolite produced by Vibrio cholerae O1 El Tor biotype in cultured host cells. Infect Immun. 2011. 79:3149–3158.
Article29. Song YJ, Stinski MF. Inhibition of cell division by the human cytomegalovirus IE86 protein: role of the p53 pathway or cyclin-dependent kinase 1/cyclin B1. J Virol. 2005. 79:2597–2603.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- NF-kappaB Activation in T Helper 17 Cell Differentiation
- beta-Lapachone suppresses radiation-induced activation of nuclear factor-kappaB
- Genetic Analysis of Epstein-Barr Virus Latent Membrane Protein 1 and Immunohistochemical Expression of Transforming Growth Factor (TGF)-beta1, TGF-betaRII, p21, p16, E2F1, Thymidylate Synthase, and NF-kappaB in Epstein-Barr Virus Encoded RNA-positive Gastric Adenocarcinoma
- The Synergistic Effect of LMP1 and IRF7 on the Expression of TAP1 in DG 75 Cell Line
- Colocalization of Interferon Regulatory Factor 7 (IRF7) with Latent Membrane Protein 1 (LMP1) of Epstein-Barr Virus