Korean J Ophthalmol.
2011 Apr;25(2):98-104. 10.3341/kjo.2011.25.2.98.
Time-Lag between Subretinal Fluid and Pigment Epithelial Detachment Reduction after Polypoidal Choroidal Vasculopathy Treatment
- Affiliations
-
- 1Department of Ophthalmology, Chungbuk National University College of Medicine, Cheongju, Korea.
- 2Department of Ophthalmology, Gangneung Asan Hospital, University of Ulsan College of Medicine, Gangneung, Korea.
- 3Department of Ophthalmology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. yhyoon@amc.seoul.kr
- KMID: 1111870
- DOI: http://doi.org/10.3341/kjo.2011.25.2.98
Abstract
- PURPOSE
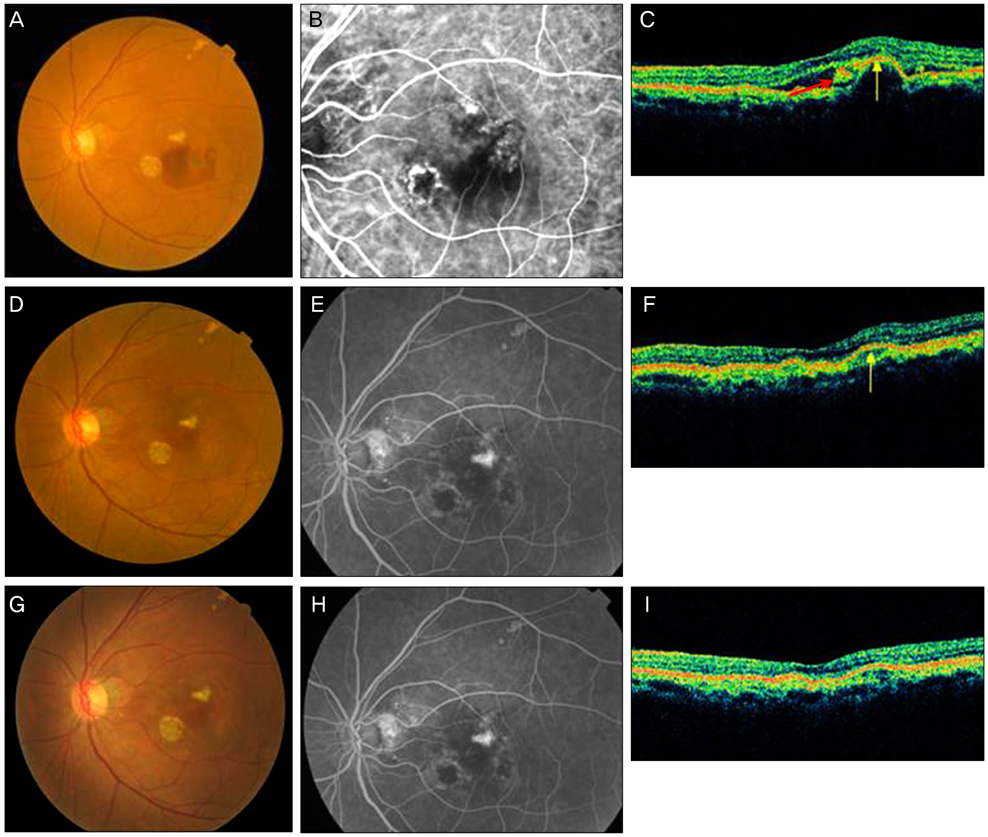
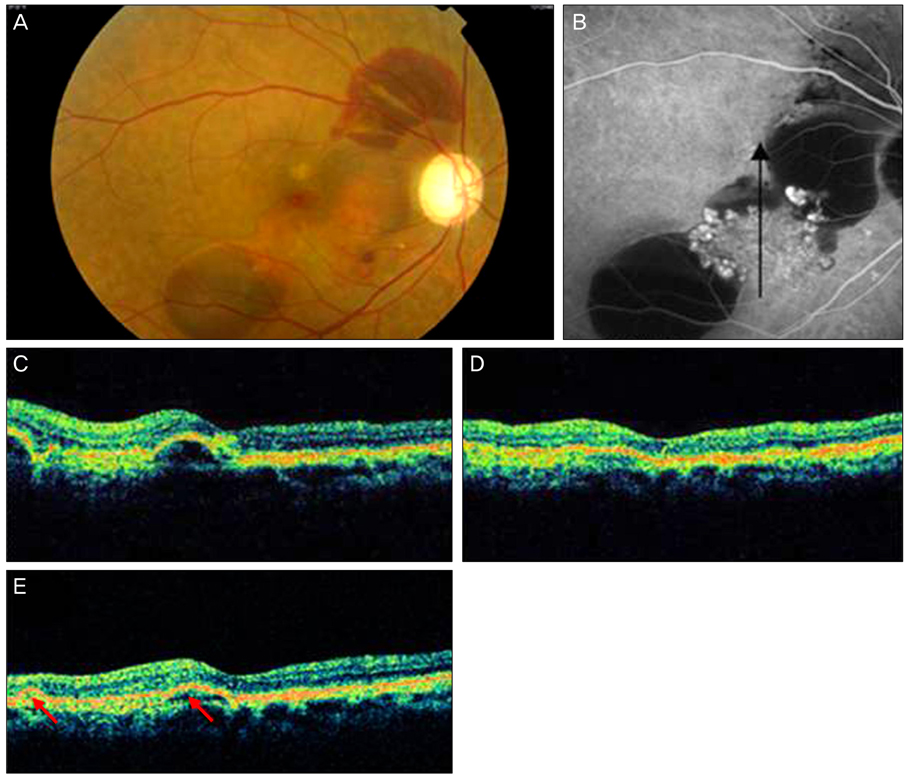
The goal of the present research was to study post-treatment changes in polypoidal choroidal vasculopathy (PCV) shown by optical coherence tomography (OCT).
METHODS
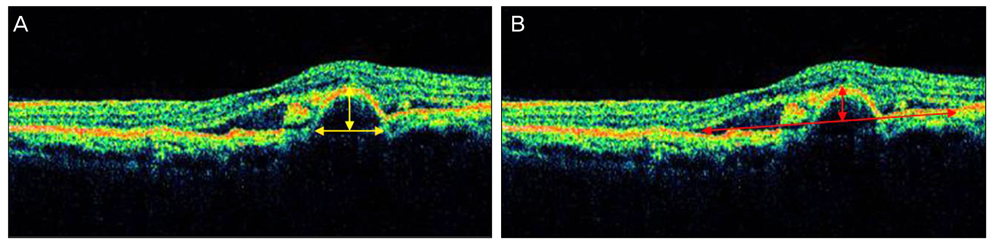
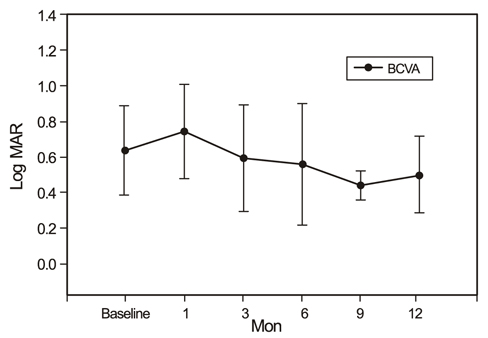
The study included 12 patients with naive PCV. Photodynamic therapy and 3 consecutive intravitreal bevacizumab injections at 6-week intervals were given. Best corrected visual acuity, subretinal fluid (SRF), pigment epithelium detachment (PED), central macular thickness (CMT), and total macular volume (TMV) were measured before and after treatment as assessed by Stratus OCT3.
RESULTS
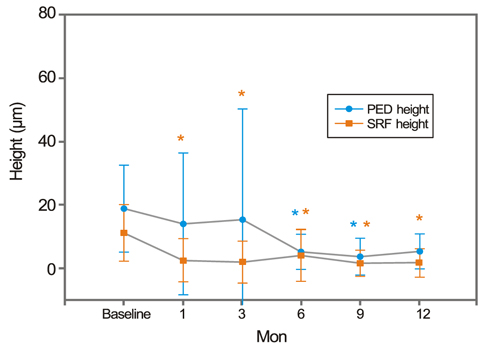
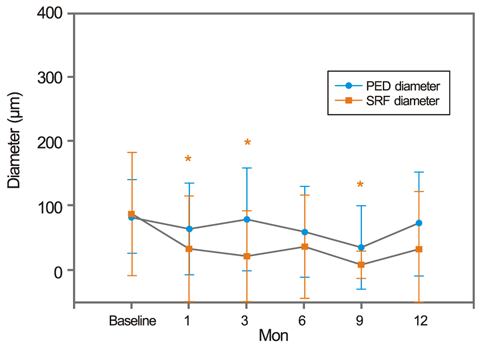
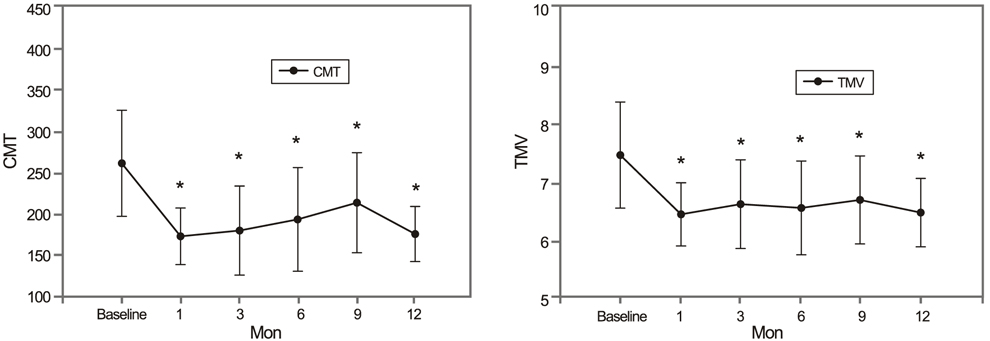
After treatment, the SRF height decreased earlier than the PED height. The SRF diameter decreased with statistical significance. However, the PED diameter did not show a statistically significant improvement, persisting at pre-treatment levels. Both CMT and TMV decreased significantly after treatment.
CONCLUSIONS
After PCV treatment, SRF and PED stabilized, as shown by OCT. However, the PED treatment response was both delayed and refractory compared to the SRF response. The small change in post-treatment PED diameter may suggest the possibility of PCV recurrence.
Keyword
MeSH Terms
-
Aged
Choroid/*pathology
Choroid Diseases
Choroidal Neovascularization/diagnosis/*drug therapy
Disease Progression
Female
Fluorescein Angiography
Follow-Up Studies
Fundus Oculi
Humans
Male
Photochemotherapy/*adverse effects
Prognosis
Retinal Detachment/diagnosis/*etiology
Retinal Pigment Epithelium/*pathology
Retrospective Studies
*Subretinal Fluid
Time Factors
Tomography, Optical Coherence
Visual Acuity
Figure
Reference
-
1. Spaide RF, Yannuzzi LA, Slakter JS, et al. Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina. 1995. 15:100–110.2. Yannuzzi LA, Ciardella A, Spaide RF, et al. The expanding clinical spectrum of idiopathic polypoidal choroidal vasculopathy. Arch Ophthalmol. 1997. 115:478–485.3. Yuzawa M, Mori R, Kawamura A. The origins of polypoidal choroidal vasculopathy. Br J Ophthalmol. 2005. 89:602–607.4. Uyama M, Matsubara T, Fukushima I, et al. Idiopathic polypoidal choroidal vasculopathy in Japanese patients. Arch Ophthalmol. 1999. 117:1035–1042.5. Uyama M, Wada M, Nagai Y, et al. Polypoidal choroidal vasculopathy: natural history. Am J Ophthalmol. 2002. 133:639–648.6. Yannuzzi LA, Wong DW, Sforzolini BS, et al. Polypoidal choroidal vasculopathy and neovascularized age-related macular degeneration. Arch Ophthalmol. 1999. 117:1503–1510.7. Moorthy RS, Lyon AT, Rabb MF, et al. Idiopathic polypoidal choroidal vasculopathy of the macula. Ophthalmology. 1998. 105:1380–1385.8. Costa RA, Navajas EV, Farah ME, et al. Polypoidal choroidal vasculopathy: angiographic characterization of the network vascular elements and a new treatment paradigm. Prog Retin Eye Res. 2005. 24:560–586.9. Ciardella AP, Donsoff IM, Huang SJ, et al. Polypoidal choroidal vasculopathy. Surv Ophthalmol. 2004. 49:25–37.10. Tateiwa H, Kuroiwa S, Gaun S, et al. Polypoidal choroidal vasculopathy with large vascular network. Graefes Arch Clin Exp Ophthalmol. 2002. 240:354–361.11. Yannuzzi LA, Flower RW, Slakter JS. Indocyanine green angiography. 1997. St. Louis: Mosby;329–339.12. Kwok AK, Lai TY, Chan CW, et al. Polypoidal choroidal vasculopathy in Chinese patients. Br J Ophthalmol. 2002. 86:892–897.13. Sho K, Takahashi K, Yamada H, et al. Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol. 2003. 121:1392–1396.14. Yannuzzi LA, Nogueira FB, Spaide RF, et al. Idiopathic polypoidal choroidal vasculopathy: a peripheral lesion. Arch Ophthalmol. 1998. 116:382–383.15. Gomez-Ulla F, Gonzalez F, Torreiro MG. Diode laser photocoagulation in idiopathic polypoidal choroidal vasculopathy. Retina. 1998. 18:481–483.16. Yuzawa M, Mori R, Haruyama M. A study of laser photocoagulation for polypoidal choroidal vasculopathy. Jpn J Ophthalmol. 2003. 47:379–384.17. Nishijima K, Takahashi M, Akita J, et al. Laser photocoagulation of indocyanine green angiographically identified feeder vessels to idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol. 2004. 137:770–773.18. Akaza E, Mori R, Yuzawa M. Long-term results of photodynamic therapy of polypoidal choroidal vasculopathy. Retina. 2008. 28:717–722.19. Lee SC, Seong YS, Kim SS, et al. Photodynamic therapy with verteporfin for polypoidal choroidal vasculopathy of the macula. Ophthalmologica. 2004. 218:193–201.20. Gomi F, Ohji M, Sayanagi K, et al. One-year outcomes of photodynamic therapy in age-related macular degeneration and polypoidal choroidal vasculopathy in Japanese patients. Ophthalmology. 2008. 115:141–146.21. Hussain N, Hussain A, Natarajan S. Role of photodynamic therapy in polypoidal choroidal vasculopathy. Indian J Ophthalmol. 2005. 53:101–104.22. Spaide RF, Donsoff I, Lam DL, et al. Treatment of polypoidal choroidal vasculopathy with photodynamic therapy. Retina. 2002. 22:529–535.23. Silva RM, Figueira J, Cachulo ML, et al. Polypoidal choroidal vasculopathy and photodynamic therapy with verteporfin. Graefes Arch Clin Exp Ophthalmol. 2005. 243:973–979.24. Chan WM, Lam DS, Lai TY, et al. Photodynamic therapy with verteporfin for symptomatic polypoidal choroidal vasculopathy: one-year results of a prospective case series. Ophthalmology. 2004. 111:1576–1584.25. Sato T, Kishi S, Watanabe G, et al. Tomographic features of branching vascular networks in polypoidal choroidal vasculopathy. Retina. 2007. 27:589–594.26. Tsujikawa A, Sasahara M, Otani A, et al. Pigment epithelial detachment in polypoidal choroidal vasculopathy. Am J Ophthalmol. 2007. 143:102–111.27. Lee WK, Lee PY, Lee SK. Photodynamic therapy for polypoidal choroidal vasculopathy: vaso-occlusive effect on the branching vascular network and origin of recurrence. Jpn J Ophthalmol. 2008. 52:108–115.28. Yamashiro K, Tsujikawa A, Nishida A, et al. Recurrence of polypoidal choroidal vasculopathy after photodynamic therapy. Jpn J Ophthalmol. 2008. 52:457–462.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Subretinal Hematoma Secondary to Polypoidal Choroidal Vasculopathy Misunderstood as a Subretinal Mass
- Initial Factors Associated with Resistance to Intravitreal Aflibercept Injection in Polypoidal Choroidal Vasculopathy
- Characteristics of Polypoidal Choroidal Vasculopathy Associated with Subretinal Hemorrhage
- A Case of Polypoidal Choroidal Vasculopathy and Serous Retinal Detachment in a Bilateral Dome-shaped Macula
- Short-Term Efficacy of Intravitreal Aflibercept for Polypoidal Choroidal Vasculopathy