J Vet Sci.
2011 Jun;12(2):133-142. 10.4142/jvs.2011.12.2.133.
Dynamic analysis of Ca2+ level during bovine oocytes maturation and early embryonic development
- Affiliations
-
- 1College of Veterinary Medicine, Northwest Agriculture and Forestry University, Yangling 712100, China.
- 2Laboratory of Genetic Resources, Institute of Animal Sciences, Chinese Academy of Agricultural Sciences, Beijing 100193, China. weijunguan301@gmail.com, Yuehui_Ma@hotmail.com
- KMID: 1106555
- DOI: http://doi.org/10.4142/jvs.2011.12.2.133
Abstract
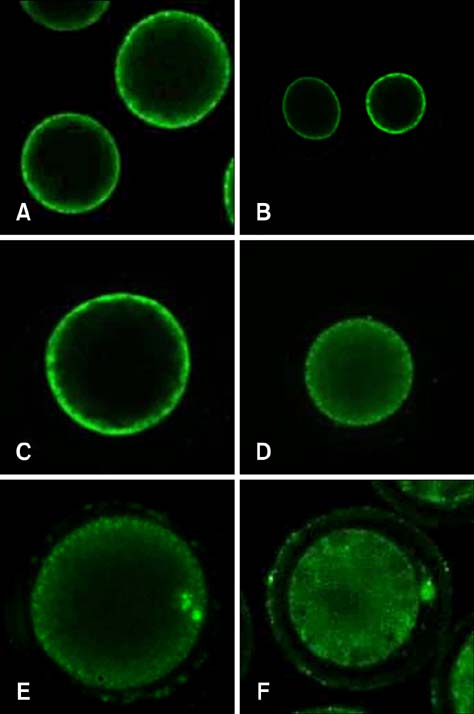
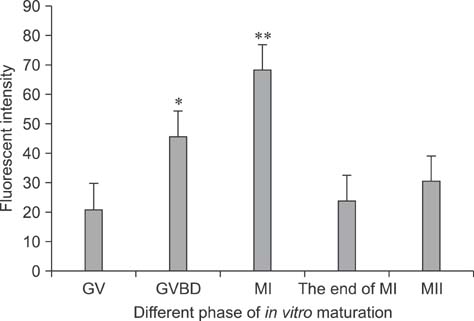
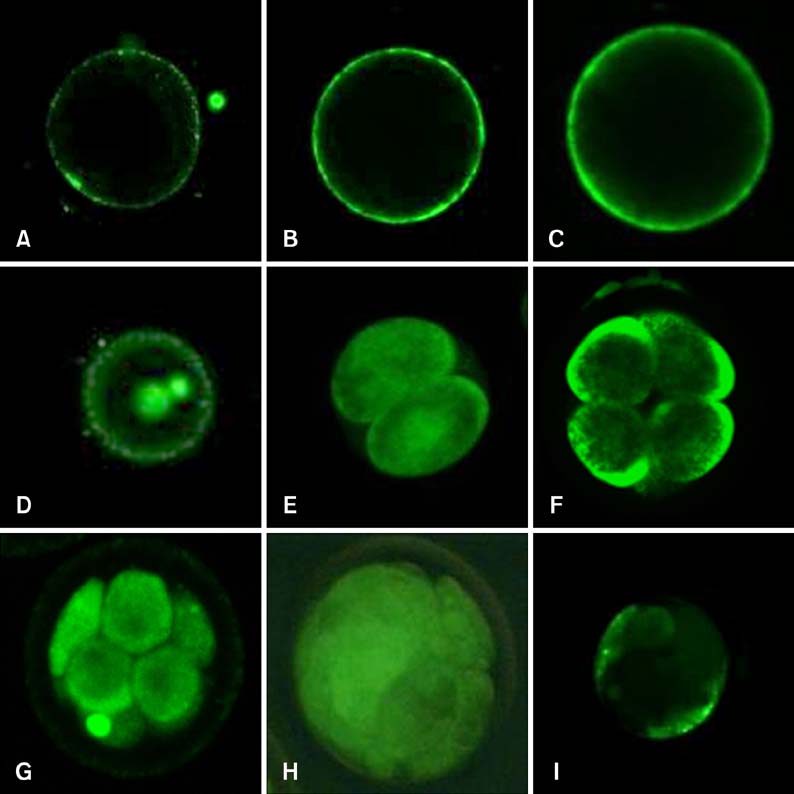
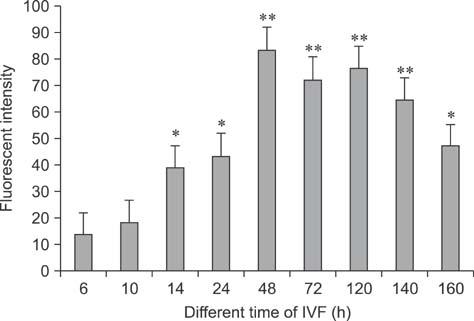
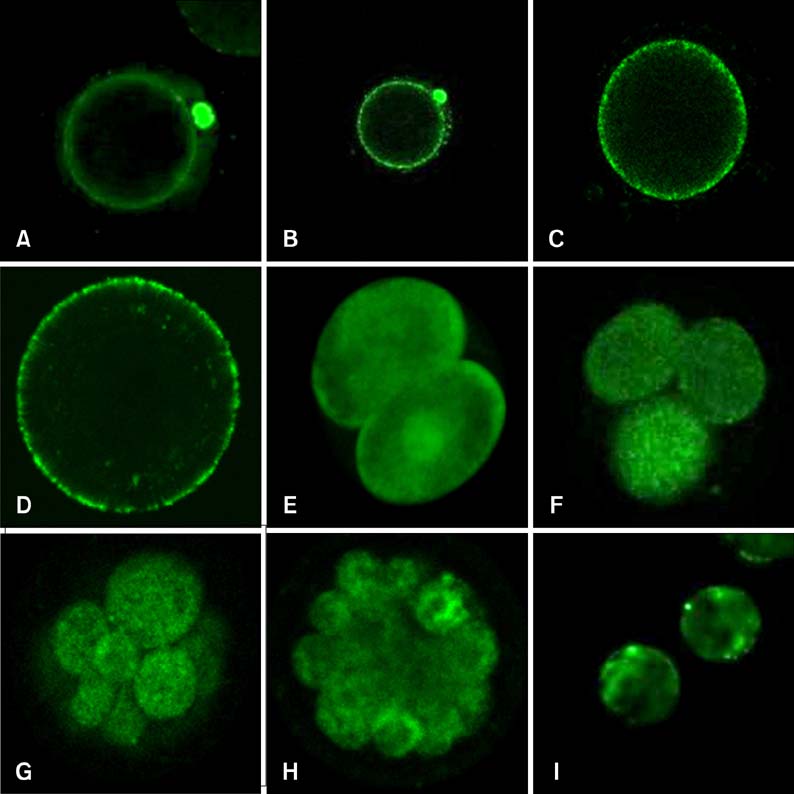
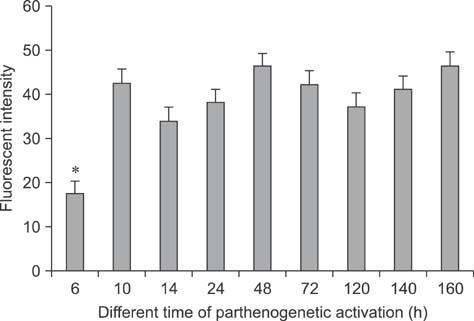
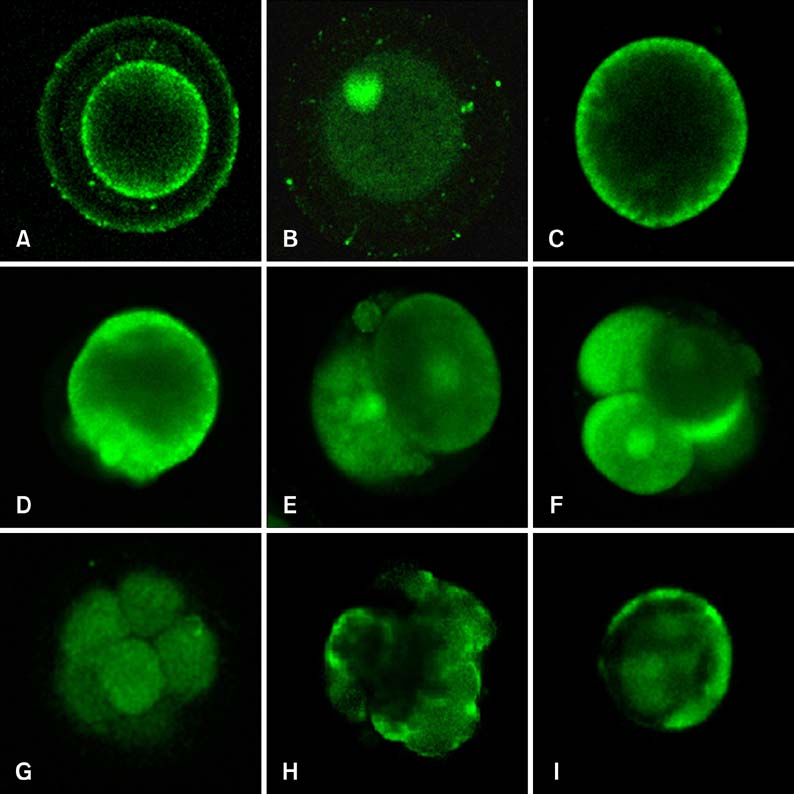
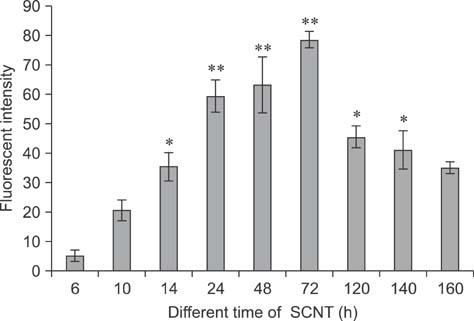
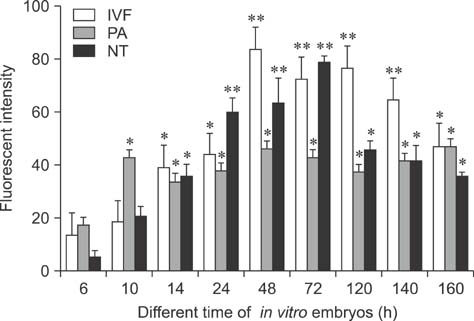
- Mammalian oocyte maturation and early embryo development processes are Ca(2+)-dependent. In this study, we used confocal microscopy to investigate the distribution pattern of Ca2+ and its dynamic changes in the processes of bovine oocytes maturation, in vitro fertilization (IVF), parthenogenetic activation (PA) and somatic cell nuclear transfer (SCNT) embryo development. During the germinal vesicle (GV) and GV breakdown stage, Ca2+ was distributed in the cortical ooplasm and throughout the oocytes from the MI to MII stage. In IVF embryos, Ca2+ was distributed in the cortical ooplasm before the formation of the pronucleus. In 4-8 cell embryos and morulas, Ca2+ was present throughout the blastomere. In PA embryos, Ca2+ was distributed throughout the blastomere at 48 h, similar to in the 4-cell and 8-cell phase and the morula. At 6 h after activation, there was almost no distribution of Ca2+ in the SCNT embryos. However, Ca2+ was distributed in the donor nucleus at 10 h and it was distributed throughout the blastomere in the 2-8 cell embryos. In this study, Ca2+ showed significant fluctuations with regularity of IVF and SCNT groups, but PA did not. Systematic investigation of the Ca2+ location and distribution changes during oocyte maturation and early embryo development processes should facilitate a better understanding of the mechanisms involved in oocyte maturation, reconstructed embryo activation and development, ultimately improving the reconstructed embryo development rate.
Keyword
MeSH Terms
Figure
Reference
-
1. Bandyopadhyay J, Lee J, Lee J, Lee JI, Yu JR, Jee C, Cho JH, Jung S, Lee MH, Zannoni S, Singson A, Kim DH, Koo HS, Ahnn J. Calcineurin, a Calcium/Calmodulin-dependent protein phosphatase, is involved in movement, fertility, egg laying, and growth in caenorhabditis elegans. Mol Biol Cell. 2002. 13:3281–3293.
Article2. Berridge MJ, Lipp P, Bootman MD. The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol. 2000. 1:11–21.
Article3. Bi CM, C Y, Dai G, Lu JC, Li CJ, Zhang XR. The Calcium oscillation and distribution before GVBD in mouse oocyte. J Nanjing Norm Univ. 2000. 23:80–83.4. Boni R, Cuomo A, Tosti E. Developmental potential in bovine oocytes is related to cumulus-oocyte complex grade, calcium current activity, and calcium stores. Biol Reprod. 2002. 66:836–842.
Article5. Carroll J. The initiation and regulation of Ca2+ signalling at fertilization in mammals. Semin Cell Dev Biol. 2001. 12:37–43.
Article6. Carroll J, Swann K, Whittingham D, Whitaker M. Spatiotemporal dynamics of intracellular [Ca2+]i oscillations during the growth and meiotic maturation of mouse oocytes. Development. 1994. 120:3507–3517.
Article7. Carroll J, Swann K. Spontaneous cytosolic calcium oscillations driven by inositol trisphosphate occur during in vitro maturation of mouse oocytes. J Biol Chem. 1992. 267:11196–11201.
Article8. Choi YH, Love CC, Chung YG, Varner DD, Westhusin ME, Burghardt RC, Hinrichs K. Use of Piezo-driven direct nuclear injection and activation with stallion sperm extract to produce horse nuclear transfer embryos. Theriogenology. 2002. 58:771–774.
Article9. Ducibella T, Huneau D, Angelichio E, Xu Z, Schultz RM, Kopf GS, Fissore R, Madoux S, Ozil JP. Egg-to-embryo transition is driven by differential responses to Ca2+ oscillation number. Dev Biol. 2002. 250:280–291.
Article10. Ducibella T, Schultz RM, Ozil JP. Role of calcium signals in early development. Semin Cell Dev Biol. 2006. 17:324–332.
Article11. Duesbery NS, Masui Y. The role of Ca2+ in progesterone-induced germinal vesicle breakdown of Xenopus laevis oocytes: the synergic effects of microtubule depolymerization and Ca2+. Dev Genes Evol. 1996. 206:110–124.
Article12. Fujiwara T, Nakada K, Shirakawa H, Miyazaki S. Development of inositol trisphosphate-induced calcium release mechanism during maturation of hamster oocytes. Dev Biol. 1993. 156:69–79.
Article13. Heilbrunn LV. The action of calcium on muscle protoplasm. Physiol Zool. 1940. 13:88–94.
Article14. Homa ST, Carroll J, Swann K. The role of calcium in mammalian oocyte maturation and egg activation. Hum Reprod. 1993. 8:1274–1281.15. Hyslop LA, Nixon VL, Levasseur M, Chapman F, Chiba K, McDougall A, Venables JP, Elliott DJ, Jones KT. Ca2+-promoted cyclin B1 degradation in mouse oocytes requires the establishment of a metaphase arrest. Dev Biol. 2004. 269:206–219.
Article16. Hyttel P, Viuff D, Laurincik J, Schmidt M, Thomsen PD, Avery B, Callesen H, Rath D, Niemann H, Rosenkranz C, Schellander K, Ochs RL, Greve T. Risks of in-vitro production of cattle and swine embryos: aberrations in chromosome numbers, ribosomal RNA gene activation and perinatal physiology. Hum Reprod. 2000. 15:Suppl 5. 87–97.
Article17. Ito M, Shikano T, Oda S, Horiguchi T, Tanimoto S, Awaji T, Mitani H, Miyazaki S. Difference in Ca2+ oscillation-inducing activity and nuclear translocation ability of plcz1, an egg-activating sperm factor candidate, between mouse, rat, human, and medaka fish. Biol Reprod. 2008. 78:1081–1090.
Article18. Jaffe LF. On the conservation of fast calcium wave speeds. Cell Calcium. 2002. 32:217–229.
Article19. Kline D, Kline JT. Repetitive calcium transients and the role of calcium in exocytosis and cell cycle activation in the mouse egg. Dev Biol. 1992. 149:80–89.
Article20. Kong SK, Tsang D, Leung KN, Lee CY. Nuclear envelope acts as a calcium barrier in C6 glioma cells. Biochem Biophys Res Commun. 1996. 218:595–600.
Article21. Li CJ, Wang B, Fan BQ. The 3-dimensional distribution of free calcium in mouse oocyte during meiotic divisions. Acta Anatomica Sinica. 1995. 26:71–76.22. Malcuit C, Kurokawa M, Fissore RA. Calcium oscillations and mammalian egg activation. J Cell Physiol. 2006. 206:565–573.
Article23. Markoulaki S, Kurokawa M, Yoon SY, Matson S, Ducibella T, Fissore R. Comparison of Ca2+ and CaMKII responses in IVF and ICSI in the mouse. Mol Hum Reprod. 2007. 13:265–272.24. Markoulaki S, Matson S, Abbott AL, Ducibella T. Oscillatory CaMKII activity in mouse egg activation. Dev Biol. 2003. 258:464–474.
Article25. Mehlmann LM. Oocyte-specific expression of Gpr3 is required for the maintenance of meiotic arrest in mouse oocytes. Dev Biol. 2005. 288:397–404.
Article26. Mitalipov SM, Zhou Q, Byrne JA, Ji WZ, Norgren RB, Wolf DP. Reprogramming following somatic cell nuclear transfer in primates is dependent upon nuclear remodeling. Hum Reprod. 2007. 22:2232–2242.
Article27. Miyazaki S, Ito M. Calcium signals for egg activation in mammals. J Pharmacol Sci. 2006. 100:545–552.
Article28. Moosmang S, Lenhardt P, Haider N, Hofmann F, Wegener JW. Mouse models to study L-type calcium channel function. Pharmacol Ther. 2005. 106:347–355.
Article29. Nedambale TL, Du F, Xu J, Chaubal SA, Dinnyes A, Groen W, Faber D, Dobrinsky JR, Yang X, Tian XC. Prolonging bovine sperm-oocyte incubation in modified medium 199 improves embryo development rate and the viability of vitrified blastocysts. Theriogenology. 2006. 66:1951–1960.
Article30. Niemann H, Wrenzycki C. Alterations of expression of developmentally important genes in preimplantation bovine embryos by in vitro culture conditions: implications for subsequent development. Theriogenology. 2000. 53:21–34.
Article31. Ozil JP, Huneau D. Activation of rabbit oocytes: the impact of the Ca2+ signal regime on development. Development. 2001. 128:917–928.
Article32. Ozil JP, Markoulaki S, Toth S, Matson S, Banrezes B, Knott JG, Schultz RM, Huneau D, Ducibella T. Egg activation events are regulated by the duration of a sustained [Ca2+]cyt signal in the mouse. Dev Biol. 2005. 282:39–54.
Article33. Rizos D, Gutiérrez-Adán A, Pérez-Garnelo S, De La Fuente J, Boland MP, Lonergan P. Bovine embryo culture in the presence or absence of serum: implications for blastocyst development, cryotolerance, and messenger RNA expression. Biol Reprod. 2003. 68:236–243.
Article34. Schultz RM, Kopf GS. 2 Molecular basis of mammalian egg activation. Curr Top Dev Boil. 1995. 30:21–62.
Article35. Steinhardt R, Zucker R, Schatten G. Intracellular calcium release at fertilization in the sea urchin egg. Dev Biol. 1977. 58:185–196.
Article36. Steinhardt RA, Epel D. Activation of sea-urchin eggs by a calcium ionophore. Proc Natl Acad Sci USA. 1974. 71:1915–1919.
Article37. Stricker SA, Smythe TL. Endoplasmic reticulum reorganizations and Ca2+ signaling in maturing and fertilized oocytes of marine protostome worms: the roles of MAPKs and MPF. Development. 2003. 130:2867–2879.
Article38. Sun L, Hodeify R, Haun S, Charlesworth A, MacNicol AM, Ponnappan S, Ponnappan U, Prigent C, Machaca K. Ca2+ homeostasis regulates Xenopus oocyte maturation. Biol Reprod. 2008. 78:726–735.39. Sun L, Machaca K. Ca2+cyt negatively regulates the initiation of oocyte maturation. J Cell Biol. 2004. 165:63–75.40. Tan SJ, Xie ZJ, Shi DS, Xie TS, Li H, Deng YF, Liu JL. Effects of porcine oocyte activation induced by a cytosolic sperm factor. J South China Agric Univ. 2009. 30:78–81.41. Tombes RM, Simerly C, Borisy GG, Schatten G. Meiosis, egg activation, and nuclear envelope breakdown are differentially reliant on Ca2+, whereas germinal vesicle breakdown is Ca2+ independent in the mouse oocyte. J Cell Biol. 1992. 117:799–811.
Article42. Tosti E, Boni R, Cuomo A. Ca2+ current activity decreases during meiotic progression in bovine oocytes. Am J Physiol Cell Physiol. 2000. 279:C1795–C1800.43. Tosti E, Boni R, Cuomo A. Fertilization and activation currents in bovine oocytes. Reproduction. 2002. 124:835–846.
Article44. Wang CG, Deng MQ, Sun FZ. Intracellular Ca2+ distribution and its role in mouse oocytes maturation. Shi Yan Sheng Wu Xue Bao. 1998. 31:147–153.45. Whitaker M, Smith J. Introduction. Calcium signals and developmental patterning. Philos Trans R Soc Lond B Biol Sci. 2008. 363:1307–1310.
Article46. Whitaker M. Calcium at fertilization and in early development. Physiol Rev. 2006. 86:25–88.
Article47. Whitaker M. Calcium signalling in early embryos. Philos Trans R Soc Lond B Biol Sci. 2008. 363:1401–1418.
Article48. Yanagimachi R. Intracytoplasmic sperm injection experiments using the mouse as a model. Hum Reprod. 1998. 13:Suppl 1. 87–98.
Article49. Yoda A, Oda S, Shikano T, Kouchi Z, Awaji T, Shirakawa H, Kinoshita K, Miyazaki S. Ca2+ oscillation-inducing phospholipase C zeta expressed in mouse eggs is accumulated to the pronucleus during egg activation. Dev Biol. 2004. 268:245–257.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Maternal effect genes: Findings and effects on mouse embryo development
- Developmental Capacity of Bovine Follicular Oocytes after Ultra-Rapid Freezing by Electron Microscope Grid I. Cryopreservation of Bovine Immature Oocytes
- Various macromolecules in in vitro growth medium influence growth, maturation, and parthenogenetic development of pig oocytes derived from small antral follicles
- Effects of sperm insemination on the final meiotic maturation of mouse oocytes arrested at metaphase I after in vitro maturation
- Effect of Reproductive Status on In Vitro Developmental Competence of Bovine Oocytes