J Vet Sci.
2008 Dec;9(4):375-379. 10.4142/jvs.2008.9.4.375.
Low numbers of intestinal Shiga toxin-producing E. coli correlate with a poor prognosis in sheep infected with bovine leukemia virus
- Affiliations
-
- 1Department of Microbiology, Molecular Biology and Biochemistry, University of Idaho, Moscow, ID 83844-3052, USA. cbohach@uidaho.edu
- 2Department of Veterinary Microbiology and Pathology, Washington Animal Disease Diagnostic Laboratory, College of Veterinary Medicine, Washington State University, Pullman WA, 99164, USA.
- 3Field Disease Investigation Unit, Washington State University, Pullman WA, 99164-6610, USA.
- KMID: 1104912
- DOI: http://doi.org/10.4142/jvs.2008.9.4.375
Abstract
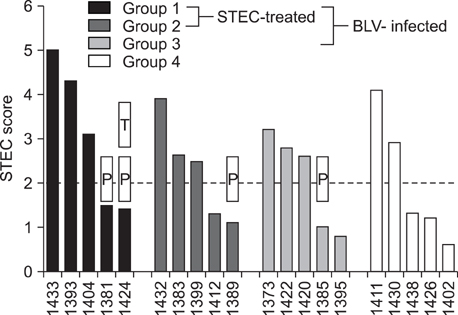
- Healthy ruminants carry intestinal Shiga toxin (Stx)-producing Escherichia coli (STEC). Stx has antiviral activities in vitro and STEC numbers correlate with reduced early viremia in sheep experimentally infected with bovine leukemia virus (BLV). This study assessed the impact of intestinal STEC on BLV-induced disease for one year post-BLV-challenge. High STEC scores (CFU/g feces x frequency of STEC-positive samples) correlated with good health, whereas poor weight gain, distress, and tumor development occurred only among animals with low STEC scores. STEC carriage was associated with increased percentages of B cells in peripheral blood.
MeSH Terms
Figure
Reference
-
1. Asakura H, Makino S, Shirahata T, Tsukamoto T, Kurazono H, Ikeda T, Takeshi K. Detection and genetical characterization of Shiga toxin-producing Escherichia coli from wild deer. Microbiol Immunol. 1998. 42:815–822.
Article2. Bettelheim KA, Bensink JC, Tambunan HS. Serotypes of verotoxin-producing (Shiga toxin-producing) Escherichia coli isolated from healthy sheep. Comp Immunol Microbiol Infect Dis. 2000. 23:1–7.
Article3. Beutin L, Geier D, Steinrück H, Zimmermann S, Scheutz F. Prevalence and some properties of verotoxin (Shiga-like toxin)-producing Escherichia coli in seven different species of healthy domestic animals. J Clin Microbiol. 1993. 31:2483–2488.
Article4. Butler T, Islam MR, Azad MA, Jones PK. Risk factors for development of hemolytic uremic syndrome during shigellosis. J Pediatr. 1987. 110:894–897.
Article5. Davis WC, Davis JE, Hamilton MJ. Use of monoclonal antibodies and flow cytometry to cluster and analyze leukocyte differentiation molecules. Methods Mol Biol. 1995. 45:149–167.
Article6. Djilali S, Parodi AL, Levy D, Cockerell GL. Development of leukemia and lymphosarcoma induced by bovine leukemia virus in sheep: a hematopathological study. Leukemia. 1987. 1:777–781.7. Endo Y, Mitsui K, Motizuki M, Tsurugi K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes. The site and the characteristics of the modification in 28 S ribosomal RNA caused by the toxins. J Biol Chem. 1987. 262:5908–5912.
Article8. Ferens WA, Hovde CJ. Antiviral activity of shiga toxin 1: suppression of bovine leukemia virus-related spontaneous lymphocyte proliferation. Infect Immun. 2000. 68:4462–4469.
Article9. Ferens WA, Cobbold R, Hovde CJ. Intestinal Shiga toxin-producing Escherichia coli bacteria mitigate bovine leukemia virus infection in experimentally infected sheep. Infect Immun. 2006. 74:2906–2916.
Article10. Ferens WA, Hovde CJ. The non-toxic A subunit of Shiga toxin type 1 prevents replication of bovine immunodeficiency virus in infected cells. Virus Res. 2007. 125:29–41.
Article11. Griffin PM, Tauxe RV. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic uremic syndrome. Epidemiol Rev. 1991. 13:60–98.
Article12. Kabeya H, Ohashi K, Onuma M. Host immune responses in the course of bovine leukemia virus infection. J Vet Med Sci. 2001. 63:703–708.
Article13. Karch H, Meyer T. Single primer pair for amplifying segments of distinct Shiga-like-toxin genes by polymerase chain reaction. J Clin Microbiol. 1989. 27:2751–2757.
Article14. Kenyon SJ, Ferrer JF, McFeely RA, Graves DC. Induction of lymphosarcoma in sheep by bovine leukemia virus. J Natl Cancer Inst. 1981. 67:1157–1163.15. Mirsky ML, Olmstead CA, Da Y, Lewin HA. The prevalence of proviral bovine leukemia virus in peripheral blood mononuclear cells at two subclinical stages of infection. J Virol. 1996. 70:2178–2183.
Article16. Nizetic D, Drmanac R, Lehrach H. An improved bacterial colony lysis procedure enables direct DNA hybridisation using short (10, 11 bases) oligonucleotides to cosmids. Nucleic Acids Res. 1991. 19:182.
Article17. Olson MC, Ramakrishnan S, Anand R. Ribosomal inhibitory proteins from plants inhibit HIV-1 replication in acutely infected peripheral blood mononuclear cells. AIDS Res Hum Retroviruses. 1991. 7:1025–1030.
Article18. Schwartz I, Bensaid A, Polack B, Perrin B, Berthelemy M, Levy D. In vivo leukocyte tropism of bovine leukemia virus in sheep and cattle. J Virol. 1994. 68:4589–4596.
Article19. Wang P, Tumer NE. Virus resistance mediated by ribosome inactivating proteins. Adv Virus Res. 2000. 55:325–355.
Article20. Yan W, Malik MN, Peterkin PI, Sharpe AN. Comparison of the hydrophobic grid-membrane filter DNA probe method and the Health Protection Branch standard method for the detection of Listeria monocytogenes in foods. Int J Food Microbiol. 1996. 30:379–384.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Shiga Toxin: Emerging Producer Strains, Prophylactic Approaches, and Application in Cancer Therapy
- Infectivity of bovine leukemia virus to Korean native goats I. antibody responses and syncytium assay for Korean native goats experimentally infected with bovine leukemia virus
- Shiga toxin-associated hemolytic uremic syndrome complicated by intestinal perforation in a child with typical hemolytic uremic syndrome
- A Case of Thrombotic Thrombocytopenic Purpura Associated with Escherichia coli O26
- Prevalence of Salmonella enterica and Shiga toxin-producing Escherichia coli in zoo animals from Chile