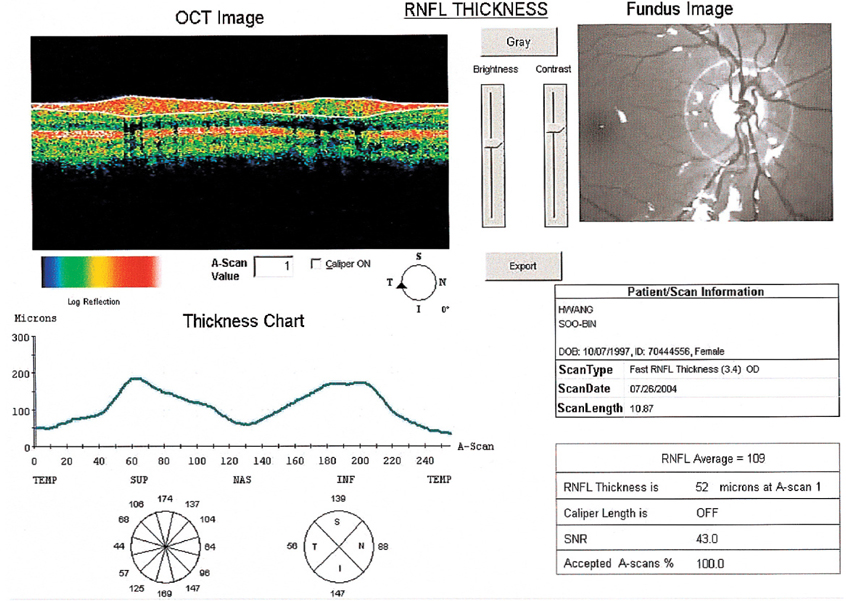
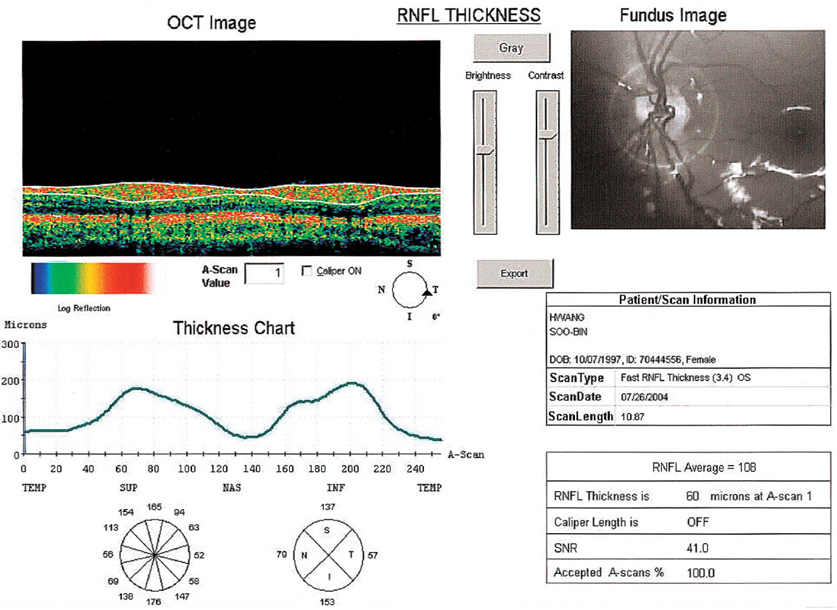
Thicknesses of Macular Retinal Layer and Peripapillary Retinal Nerve Fiber Layer in Patients with Hyperopic Anisometropic Amblyopia
- Affiliations
-
- 1Department of Ophthalmology, Kim's Eye Hospital, Myung-Gok Eye Research Institute, Konyang University College of Medicine, Seoul, Korea. slitlamp2@hanmail.net
- KMID: 1099087
- DOI: http://doi.org/10.3341/kjo.2005.19.1.62
Abstract
- This prospective study was performed to measure the macular and the peripapillary retinal nerve fiber layer (RNFL) thicknesses using optical coherence tomography (OCT) in patients with anisometropic amblyopia. Thirty-one patients with hyperopic anisometropic amblyopia were included. The macular retinal thickness and the peripapillary RNFL thickness were measured using OCT. The mean refractive error was +3.71 diopters (D) and +1.00 D, the mean macular retinal thickness was 252.5 micrometer and 249.7 micrometer, and the mean RNFL thickness was 115.2 micrometer and 109.6 micrometer, in the amblyopic eye and the normal eye, respectively. OCT assessment of RNFL thickness revealed a significantly thicker RNFL in hyperopic anisometropic amblyopia (P=0.019), but no statistically significant difference was found in macular retinal thickness (P> 0.05). In conclusion, the amblyopic process may involve the peripapillary RNFL, but not the macula. However, further evaluation is needed.
Keyword
MeSH Terms
Figure
Cited by 4 articles
-
Macular Thickness in Moderate to Severe Amblyopia
Zhale Rajavi, Hamideh Sabbaghi, Narges Behradfar, Mehdi Yaseri, Mohammad Aghazadeh Amiri, Mohammad Faghihi
Korean J Ophthalmol. 2018;32(4):312-318. doi: 10.3341/kjo.2017.0101.Retinal Nerve Fiber Layer Thickness and Optic Disc Parameters in Dominant Compared with Non-Dominant Eyes
Min Soo Lee, Kyong Jin Cho, Woo Hyung Cho, Sung Eun Kyung, Moo Hwan Chang
J Korean Ophthalmol Soc. 2013;54(5):784-788. doi: 10.3341/jkos.2013.54.5.784.The Analysis of Retinal Nerve Fiber Layer in Amblyopia Using Spectral Domain Optical Coherence Tomography
Chan Keum Park, Su Jin Kim
J Korean Ophthalmol Soc. 2016;57(10):1631-1639. doi: 10.3341/jkos.2016.57.10.1631.The Analysis of Peripapillary RNFL, Macula and Macular Ganglion Cell Layer Thickness in Patients with Monocular Amblyopia Using SD-OCT
Ji Man Park, Young Je Choi, Dae Hyun Kim
J Korean Ophthalmol Soc. 2016;57(1):98-105. doi: 10.3341/jkos.2016.57.1.98.
Reference
-
1. Von Noorden GK. Mechanism of amblyopia. Doc Ophthalmol. 1977. 34:93.2. Headon MP, Powell TC. Cellular changes in the lateral geniculate nucleus of infant monkeys after suture of the eyelids. J Anat. 1973. 116:135–145.3. Sherman SM, Wilson JR. Behavioral and morphological evidence for binocular competition in the postnatal development of the dog's visual system. J Comp Neurol. 1975. 161:183–195.4. Von Noorden GK. Histological studies of the visual system in monkeys with experimental amblyopia. Invest Ophthalmol Vis Sci. 1973. 12:727–738.5. Wiesel TN, Hubel DH. Effect of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J Neurophysiol. 1963. 26:978–993.6. Von Noorden GK, Crawford MLJ, Levacy RA. The lateral geniculate nucleus in human anisometropic amblyopia. Invest Ophthalmol Vis Sci. 1983. 24:788–790.7. Von Noorden GK, Crawford ML. The lateral geniculate nucleus in human strabismic amblyopia. Invest Ophthalmol Vis Sci. 1992. 33:2729–2732.8. Rasch E, Swift H, Riesen AH, Chow KL. Altered structure and composition of retinal cells in dark-reared mammals. Exp Cell Res. 1961. 25:348–363.9. Wendell-Smith CP. Effect of light deprivation on the postnatal development of the optic nerve. Nature. 1964. 204:707.10. Chow KL. Failure to demonstrate changes in the visual system of monkeys kept in darkness or colored light. J Comp Neurol. 1955. 102:597–606.11. Chauban S, Marshall J. The interpretation of optical coherence tomography image of the retina. Invest Ophthalmol Vis Sci. 1999. 40:2332–2342.12. Hee MR, Puliafito CA, Wong C. Quantitative assessment of macular edema with optical coherence tomography. Arch Ophthalmol. 1995. 113:1019–1029.13. Puliafito CA, Hee MR, Lin CP. Imaging of macular diseases with optical coherence tomography. Ophthalmology. 1995. 102:217–229.14. DaCunha D, Jenkins EM. Amblyopia in three year olds. Med Officer. 1961. 106:146.15. Flom MC, Neummaier RW. Prevalence of amblyopia. Public Health Rep. 1966. 81:329.16. Friedmann Z, Neumann E, Hyams SW, Peleg B. Ophthalmic screening of 38,000 children, age 1 to 2.5 years, in child welfare clinics. J Pediatr Ophthalmol Strabismus. 1980. 17:261–267.17. Vereecken E, Feron A, Evens L. Importance de la detection prococe du strabisme et de l'amblyopie (in French). Bull Soc Belge Ophthalmol. 1966. 143:729–739.18. McNeil NL. Patterns of visual defects in children. Br J Ophthalmol. 1955. 39:688–670.19. Russell EL, Kada JM, Hufhines DM. Orange County vision screening project. Ophthalmologic evaluation. Sight Saving Rev. 1961. 31:215–219.20. Chow KL, Riesen AH, Newell FN. Degeneration of retinal ganglion cells in infant chimpanzees reared in darkness. J Comp Neurol. 1957. 107:27–42.21. Rasch E, Swift H, Riesen AH, Chow KL. Altered structure and composition of retinal cells in dark-reared mammals. Exp Cell Res. 1961. 25:348–363.22. Arden GB, Wooding SL. Pattern ERG in amblyopia. Invest Ophthalmol Vis Sci. 1985. 26:88–96.23. Hess RF, Baker CL, Nerhoeve JN. The pattern evoked electroretinogram: its variability in normals and its relationship to amblyopia. Invest Ophthalmol Vis Sci. 1985. 26:1610–1623.24. Deline PJ, Weissenbruch C, Berendschot TT, Norren DV. Photoreceptor function in unilateral amblyopia. Vision Res. 1998. 38:613–617.25. Colen TP, de Faber JT, Lemij HG. Retinal nerve fiber layer thickness in human strabismic amblyopia. Binocul Vis Strabismus Q. 2000. 15:141–146.26. Bozkurt B, Irkec M, Orhan M. Thickness of the retinal nerve fiber layer in patients with anisometropic and strabismic amblyopia. Strabismus Binocul Vis Strabismus Q. 2003. 11:1–7.27. Yen MY, Cheng CY, Wang AG. Retinal nerve fiber layer thickness in unilateral amblyopia. Invest Ophthalmol Vis Sci. 2004. 45:2224–2230.28. Mrugacz M, Lazarczyk AB, Kita DS. Use of optical coherence tomography in myopia. J Pediatr Ophthalmol Strabismus. 2004. 41:159–162.29. Hee MR, Puliafito CA, Duker JS. Topography of diabetic macular edema with optical coherence tomography. Ophthalmology. 1998. 105:360–370.30. Kang JH, Kim SA, Song WG. Macular thickness changes with age in normal subjects measured by optical coherence tomography. J Korean Ophthalmol Soc. 2004. 45:592–598.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Thicknesses of the Fovea and Retinal Nerve Fiber Layer in Amblyopic and Normal Eyes in Children
- Associations of Peripapillary Retinal Nerve Fiber Layer and Macular Retinal Layer Thickness with Serum Homocysteine Concentration
- The Relationship between the OCT Parameters and Visual Acuity after Occlusion Therapy in Hyperopic Anisometropic Amblyopia
- The Analysis of Peripapillary RNFL, Macula and Macular Ganglion Cell Layer Thickness in Patients with Monocular Amblyopia Using SD-OCT
- Comparison of OCT Parameters between the Dominant and Nondominant Eye