Int J Thyroidol.
2024 Nov;17(2):286-294. 10.11106/ijt.2024.17.2.286.
Are TERT Promoter Mutations a Poor Prognostic Factor in Anaplastic Thyroid Carcinoma?
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Medicine, Thyroid Center, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 2Department of Endocrinology and Metabolism, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, Korea
- 3Department of Pathology and Translational Genomics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 4Division of Endocrinology and Metabolism, Department of Internal Medicine, Myongji Hospital, Hanyang University College of Medicine, Goyang, Korea
- 5Department of Internal Medicine, CHA Bundang Medical Center, CHA University, Seongnam, Korea
- KMID: 2561609
- DOI: http://doi.org/10.11106/ijt.2024.17.2.286
Abstract
- Background and Objectives
Telomerase reverse transcriptase (TERT) promoter mutations are a poor prognostic factor in differentiated thyroid carcinoma (DTC). However, their prognostic value in anaplastic thyroid carcinoma (ATC) is unclear. Therefore, we investigated whether TERT promoter mutations also act as an independent poor prognostic factor in ATC.
Materials and Methods
We reviewed the medical records of 41 patients with ATC who underwent the TERT promoter mutations test at Samsung Medical Center between November 1995 and December 2022. The aggressive treatment group was defined as patients who underwent surgery, external radiotherapy, and systemic therapy.
Results
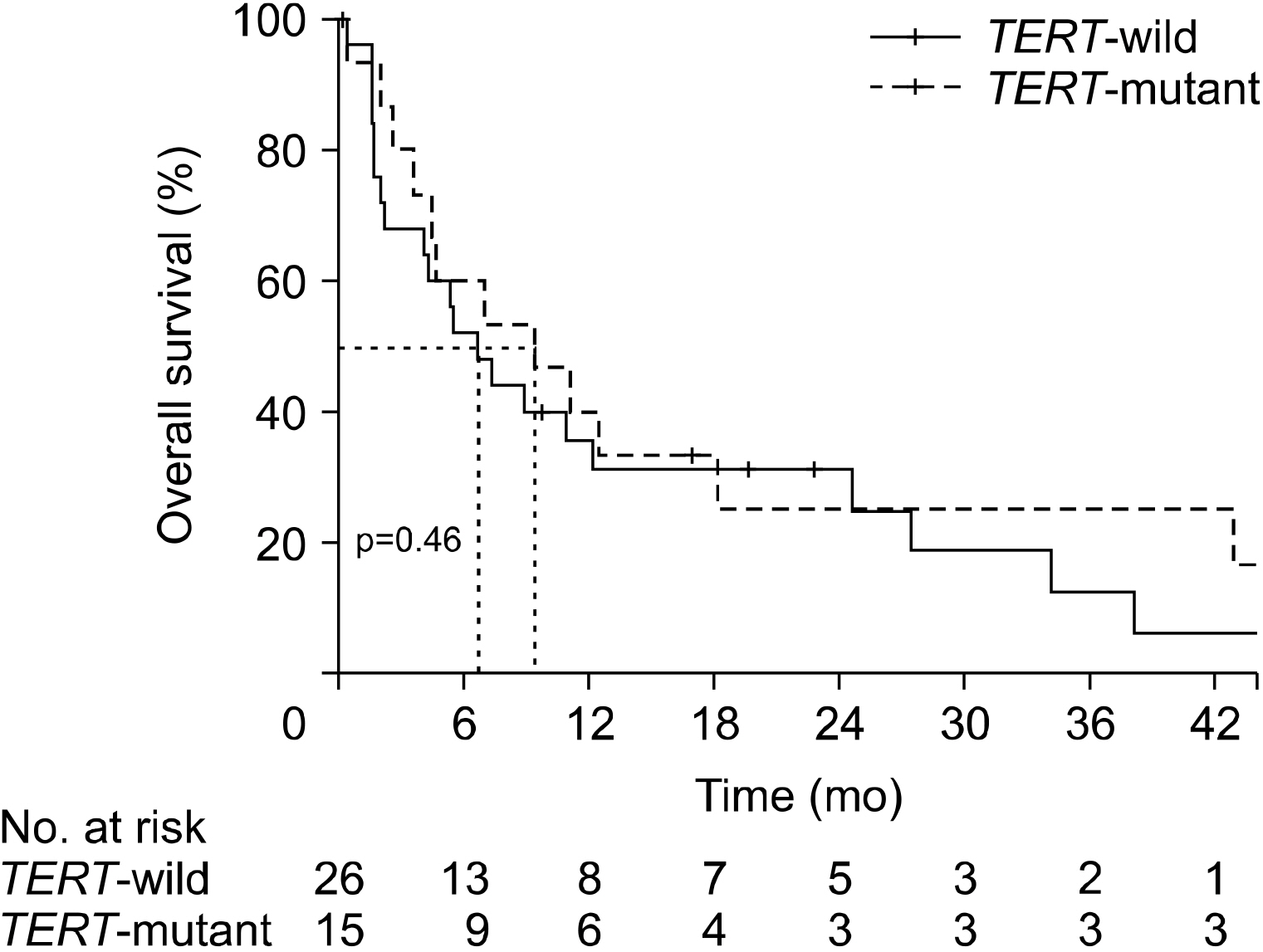
Among 41 patients, 15 (36.6%) showed TERT promoter mutations. There only differences in the clinicopathological characteristics between the TERT-mutant and wild-type groups were tumor size and coexistence of DTC. Median tumor size in the TERT-mutant group was 5.1 cm (3.0-11.0), which was significantly larger than that in the wild-type group (4.1 cm, 0.8-8.0, p=0.010). Nevertheless, the TERT-mutant group received relatively more aggressive treatment (53.3% vs. 19.2%, p=0.056), and the overall survival of the TERT-mutant group was longer than that of the wild-type group (9.4 months [0.4-51.5] vs. 7.1 months [0.4-49.5]), but its difference was not significant (p=0.458). In multiple regression analysis, distant metastasis was a significant prognostic factor, but TERT promoter mutation was not.
Conclusion
Unlike in DTC, TERT promoter mutations were not an independent poor prognostic factor in ATC.
Keyword
Figure
Reference
-
References
1. Horn S, Figl A, Rachakonda PS, Fischer C, Sucker A, Gast A, et al. 2013; TERT promoter mutations in familial and sporadic melanoma. Science. 339(6122):959–61. DOI: 10.1126/science.1230062. PMID: 23348503.2. Huang FW, Hodis E, Xu MJ, Kryukov GV, Chin L, Garraway LA. 2013; Highly recurrent TERT promoter mutations in human melanoma. Science. 339(6122):957–9. DOI: 10.1126/science.1229259. PMID: 23348506. PMCID: PMC4423787.3. Vinagre J, Pinto V, Celestino R, Reis M, Populo H, Boaventura P, et al. 2014; Telomerase promoter mutations in cancer: an emerging molecular biomarker? Virchows Arch. 465(2):119–33. DOI: 10.1007/s00428-014-1608-4. PMID: 25048572.4. Vinagre J, Almeida A, Pópulo H, Batista R, Lyra J, Pinto V, et al. 2013; Frequency of TERT promoter mutations in human cancers. Nat Commun. 4:2185. DOI: 10.1038/ncomms3185. PMID: 23887589.5. Alzahrani AS, Alsaadi R, Murugan AK, Sadiq BB. 2016; TERT promoter mutations in thyroid cancer. Horm Cancer. 7(3):165–77. DOI: 10.1007/s12672-016-0256-3. PMID: 26902827. PMCID: PMC10355936.6. Kim TH, Kim YE, Ahn S, Kim JY, Ki CS, Oh YL, et al. 2016; TERT promoter mutations and long-term survival in patients with thyroid cancer. Endocr Relat Cancer. 23(10):813–23. DOI: 10.1530/ERC-16-0219. PMID: 27528624.7. Yang H, Park H, Ryu HJ, Heo J, Kim JS, Oh YL, et al. 2022; Frequency of TERT promoter mutations in real-world analysis of 2,092 thyroid carcinoma patients. Endocrinol Metab (Seoul). 37(4):652–63. DOI: 10.3803/EnM.2022.1477. PMID: 35864728. PMCID: PMC9449103.8. Melo M, da Rocha AG, Vinagre J, Batista R, Peixoto J, Tavares C, et al. 2014; TERT promoter mutations are a major indicator of poor outcome in differentiated thyroid carcinomas. J Clin Endocrinol Metab. 99(5):E754–65. DOI: 10.1210/jc.2013-3734. PMID: 24476079. PMCID: PMC4191548.9. Xing M, Liu R, Liu X, Murugan AK, Zhu G, Zeiger MA, et al. 2014; BRAF V600E and TERT promoter mutations cooperatively identify the most aggressive papillary thyroid cancer with highest recurrence. J Clin Oncol. 32(25):2718–26. DOI: 10.1200/JCO.2014.55.5094. PMID: 25024077. PMCID: PMC4145183.10. Gandolfi G, Ragazzi M, Frasoldati A, Piana S, Ciarrocchi A, Sancisi V. 2015; TERT promoter mutations are associated with distant metastases in papillary thyroid carcinoma. Eur J Endocrinol. 172(4):403–13. DOI: 10.1530/EJE-14-0837. PMID: 25583906.11. George JR, Henderson YC, Williams MD, Roberts DB, Hei H, Lai SY, et al. 2015; Association of TERT promoter mutation, but not BRAF mutation, with increased mortality in PTC. J Clin Endocrinol Metab. 100(12):E1550–9. DOI: 10.1210/jc.2015-2690. PMID: 26461266. PMCID: PMC4667158.12. Bullock M, Ren Y, O'Neill C, Gill A, Aniss A, Sywak M, et al. 2016; TERT promoter mutations are a major indicator of recurrence and death due to papillary thyroid carcinomas. Clin Endocrinol (Oxf). 85(2):283–90. DOI: 10.1111/cen.12999. PMID: 26667986. PMCID: PMC5683578.13. Kim TH, Ki CS, Kim HS, Kim K, Choe JH, Kim JH, et al. 2017; Refining dynamic risk stratification and prognostic groups for differentiated thyroid cancer with TERT promoter mutations. J Clin Endocrinol Metab. 102(5):1757–64. DOI: 10.1210/jc.2016-3434. PMID: 28323925.14. Liu R, Bishop J, Zhu G, Zhang T, Ladenson PW, Xing M. 2017; Mortality risk stratification by combining BRAF V600E and TERT promoter mutations in papillary thyroid cancer: genetic duet of BRAF and TERT promoter mutations in thyroid cancer mortality. JAMA Oncol. 3(2):202–8. DOI: 10.1001/jamaoncol.2016.3288. PMID: 27581851.15. Oishi N, Kondo T, Ebina A, Sato Y, Akaishi J, Hino R, et al. 2017; Molecular alterations of coexisting thyroid papillary carcinoma and anaplastic carcinoma: identification of TERT mutation as an independent risk factor for transformation. Mod Pathol. 30(11):1527–37. DOI: 10.1038/modpathol.2017.75. PMID: 28731042.16. Chintakuntlawar AV, Foote RL, Kasperbauer JL, Bible KC. 2019; Diagnosis and management of anaplastic thyroid cancer. Endocrinol Metab Clin North Am. 48(1):269–84. DOI: 10.1016/j.ecl.2018.10.010. PMID: 30717908.17. Lin B, Ma H, Ma M, Zhang Z, Sun Z, Hsieh IY, et al. 2019; The incidence and survival analysis for anaplastic thyroid cancer: a SEER database analysis. Am J Transl Res. 11(9):5888–96.18. Iyer PC, Dadu R, Gule-Monroe M, Busaidy NL, Ferrarotto R, Habra MA, et al. 2018; Salvage pembrolizumab added to kinase inhibitor therapy for the treatment of anaplastic thyroid carcinoma. J Immunother Cancer. 6(1):68. DOI: 10.1186/s40425-018-0378-y. PMID: 29996921. PMCID: PMC6042271.19. Keam B, Kreitman RJ, Wainberg ZA, Cabanillas ME, Cho DC, Italiano A, et al. 2018; Updated efficacy and safety data of dabrafenib (D) and trametinib (T) in patients (pts) with BRAF V600E-mutated anaplastic thyroid cancer (ATC). Ann Oncol. 29(Suppl 8):viii645–8. DOI: 10.1093/annonc/mdy302.002.20. Subbiah V, Kreitman RJ, Wainberg ZA, Cho JY, Schellens JHM, Soria JC, et al. 2018; Dabrafenib and trametinib treatment in patients with locally advanced or metastatic BRAF V600-mutant anaplastic thyroid cancer. J Clin Oncol. 36(1):7–13. DOI: 10.1200/JCO.2017.73.6785. PMID: 29072975. PMCID: PMC5791845.21. Capdevila J, Wirth LJ, Ernst T, Ponce Aix S, Lin CC, Ramlau R, et al. 2020; PD-1 blockade in anaplastic thyroid carcinoma. J Clin Oncol. 38(23):2620–7. DOI: 10.1200/JCO.19.02727. PMID: 32364844. PMCID: PMC7476256.22. Dierks C, Seufert J, Aumann K, Ruf J, Klein C, Kiefer S, et al. 2021; Combination of lenvatinib and pembrolizumab is an effective treatment option for anaplastic and poorly differentiated thyroid carcinoma. Thyroid. 31(7):1076–85. DOI: 10.1089/thy.2020.0322. PMID: 33509020. PMCID: PMC8290324.23. Leboulleux S, Godbert Y, Penel N, Hescot S, De la Fouchardiere C, Blonski M, et al. 2021; Benefits of pembrolizumab in progressive radioactive iodine refractory thyroid cancer: results of the AcSé pembrolizumab study from unicancer. J Clin Oncol. 39(15_suppl):6082. DOI: 10.1200/JCO.2021.39.15_suppl.6082.24. Park J, Jung HA, Shim JH, Park WY, Kim TH, Lee SH, et al. 2021; Multimodal treatments and outcomes for anaplastic thyroid cancer before and after tyrosine kinase inhibitor therapy: a real-world experience. Eur J Endocrinol. 184(6):837–45. DOI: 10.1530/EJE-20-1482. PMID: 33852431.25. Subbiah V, Kreitman RJ, Wainberg ZA, Cho JY, Schellens JHM, Soria JC, et al. 2022; Dabrafenib plus trametinib in patients with BRAF V600E-mutant anaplastic thyroid cancer: updated analysis from the phase II ROAR basket study. Ann Oncol. 33(4):406–15. DOI: 10.1016/j.annonc.2021.12.014. PMID: 35026411. PMCID: PMC9338780.26. Bible KC, Kebebew E, Brierley J, Brito JP, Cabanillas ME, Clark TJ Jr, et al. 2021; 2021 American Thyroid Association guidelines for management of patients with anaplastic thyroid cancer. Thyroid. 31(3):337–86. DOI: 10.1089/thy.2020.0944. PMID: 33728999. PMCID: PMC8349723.27. Landa I, Ibrahimpasic T, Boucai L, Sinha R, Knauf JA, Shah RH, et al. 2016; Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J Clin Invest. 126(3):1052–66. DOI: 10.1172/JCI85271. PMID: 26878173. PMCID: PMC4767360.28. Molinaro E, Romei C, Biagini A, Sabini E, Agate L, Mazzeo S, et al. 2017; Anaplastic thyroid carcinoma: from clinicopathology to genetics and advanced therapies. Nat Rev Endocrinol. 13(11):644–60. DOI: 10.1038/nrendo.2017.76. PMID: 28707679.29. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. 2009; New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 45(2):228–47. DOI: 10.1016/j.ejca.2008.10.026. PMID: 19097774.30. Liu T, Wang N, Cao J, Sofiadis A, Dinets A, Zedenius J, et al. 2014; The age- and shorter telomere-dependent TERT promoter mutation in follicular thyroid cell-derived carcinomas. Oncogene. 33(42):4978–84. DOI: 10.1038/onc.2013.446. PMID: 24141777.31. Shi X, Liu R, Qu S, Zhu G, Bishop J, Liu X, et al. 2015; Association of TERT promoter mutation 1,295,228 C>T with BRAF V600E mutation, older patient age, and distant metastasis in anaplastic thyroid cancer. J Clin Endocrinol Metab. 100(4):E632–7. DOI: 10.1210/jc.2014-3606. PMID: 25584719. PMCID: PMC4399285.32. Xu B, Zhang L, Setoodeh R, Mohanty AS, Landa I, Balzer B, et al. 2022; Prolonged survival of anaplastic thyroid carcinoma is associated with resectability, low tumor-infiltrating neutrophils/myeloid-derived suppressor cells, and low peripheral neutrophil- to-lymphocyte ratio. Endocrine. 76(3):612–9. DOI: 10.1007/s12020-022-03008-9. PMID: 35149932. PMCID: PMC10173871.33. Chung JH. 2020; BRAF and TERT promoter mutations: clinical application in thyroid cancer. Endocr J. 67(6):577–84. DOI: 10.1507/endocrj.EJ20-0063. PMID: 32321884.34. Gu H, Wang J, Ran W, Li G, Hu S, Zhao H, et al. 2024; Anaplastic and poorly differentiated thyroid carcinomas: genetic evidence of high-grade transformation from differentiated thyroid carcinoma. J Pathol Clin Res. 10(2):e356. DOI: 10.1002/cjp2.356. PMID: 38602501. PMCID: PMC10796291.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Mechanisms of TERT Reactivation and Its Interaction with BRAFV600E
- Thyroid Cancer, Iodine, and Gene Mutation
- Frequency of TERT Promoter Mutations in Real-World Analysis of 2,092 Thyroid Carcinoma Patients
- TERT Promoter Mutations and Tumor Persistence/Recurrence in Papillary Thyroid Cancer
- Low Prevalence of Somatic TERT Promoter Mutations in Classic Papillary Thyroid Carcinoma