Diabetes Metab J.
2024 Jul;48(4):487-502. 10.4093/dmj.2023.0362.
Protein Arginine Methyltransferases: Emerging Targets in Cardiovascular and Metabolic Disease
- Affiliations
-
- 1Department of Molecular Cell Biology, Single Cell Network Research Center, Sungkyunkwan University, Suwon, Korea
- 2Department of Biomedical Science and Engineering, Gwangju Institute of Science and Technology, Gwangju, Korea
- 3Muscle Physiome Institute, College of Pharmacy, Sookmyung Women’s University, Seoul, Korea
- 4Research Institute of Aging-Related Diseases, AniMusCure Inc., Suwon, Korea
- KMID: 2558032
- DOI: http://doi.org/10.4093/dmj.2023.0362
Abstract
- Cardiovascular diseases (CVDs) and metabolic disorders stand as formidable challenges that significantly impact the clinical outcomes and living quality for afflicted individuals. An intricate comprehension of the underlying mechanisms is paramount for the development of efficacious therapeutic strategies. Protein arginine methyltransferases (PRMTs), a class of enzymes responsible for the precise regulation of protein methylation, have ascended to pivotal roles and emerged as crucial regulators within the intrinsic pathophysiology of these diseases. Herein, we review recent advancements in research elucidating on the multifaceted involvements of PRMTs in cardiovascular system and metabolic diseases, contributing significantly to deepen our understanding of the pathogenesis and progression of these maladies. In addition, this review provides a comprehensive analysis to unveil the distinctive roles of PRMTs across diverse cell types implicated in cardiovascular and metabolic disorders, which holds great potential to reveal novel therapeutic interventions targeting PRMTs, thus presenting promising perspectives to effectively address the substantial global burden imposed by CVDs and metabolic disorders.
Keyword
Figure
Reference
-
1. Miranda AM, Janbandhu V, Maatz H, Kanemaru K, Cranley J, Teichmann SA, et al. Single-cell transcriptomics for the assessment of cardiac disease. Nat Rev Cardiol. 2023; 20:289–308.
Article2. Litvinukova M, Talavera-Lopez C, Maatz H, Reichart D, Worth CL, Lindberg EL, et al. Cells of the adult human heart. Nature. 2020; 588:466–72.
Article3. Heindel JJ, Blumberg B, Cave M, Machtinger R, Mantovani A, Mendez MA, et al. Metabolism disrupting chemicals and metabolic disorders. Reprod Toxicol. 2017; 68:3–33.
Article4. di Somma M, Vliora M, Grillo E, Castro B, Dakou E, Schaafsma W, et al. Role of VEGFs in metabolic disorders. Angiogenesis. 2020; 23:119–30.5. Koh JH, Kim YW, Seo DY, Sohn TS. Mitochondrial TFAM as a signaling regulator between cellular organelles: a perspective on metabolic diseases. Diabetes Metab J. 2021; 45:853–65.
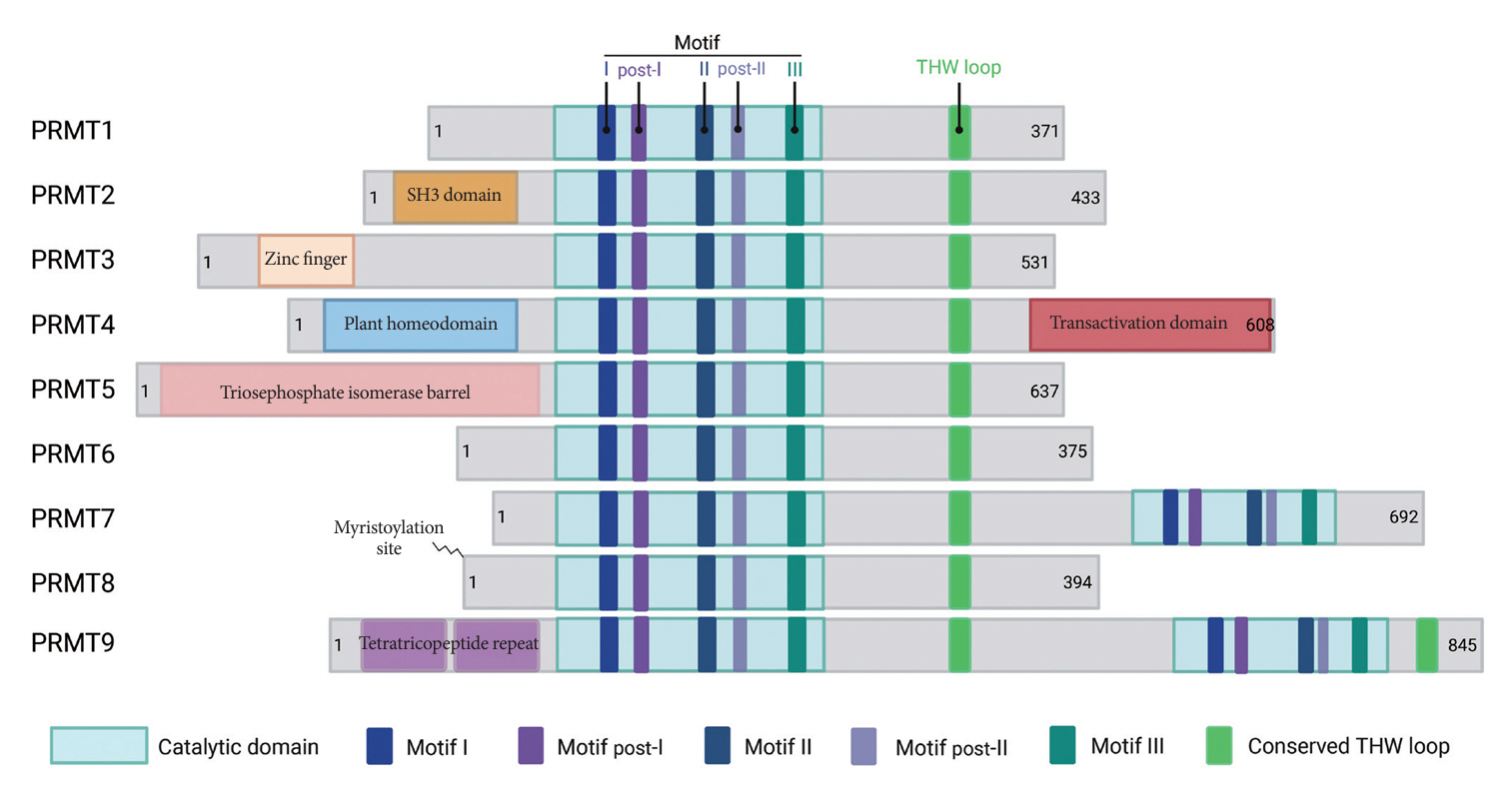
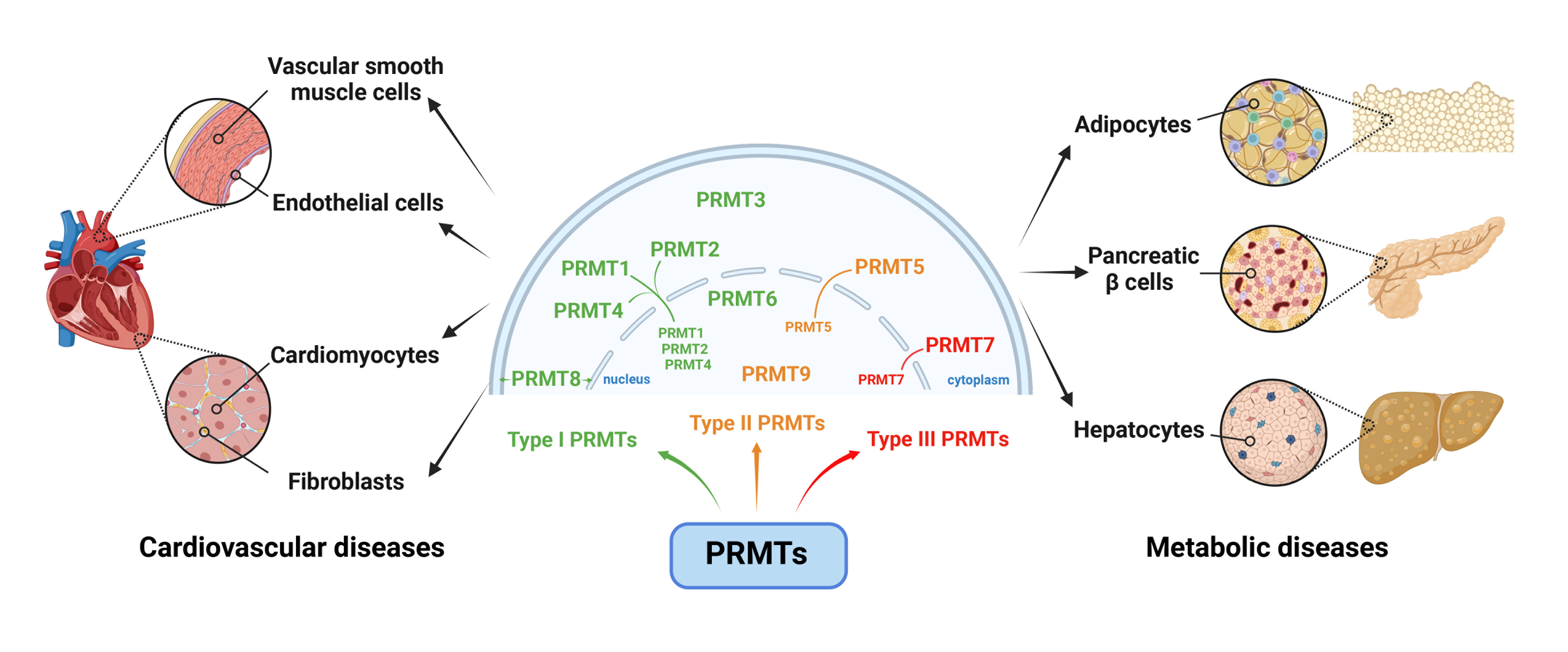
Article6. Couto E Silva A, Wu CY, Citadin CT, Clemons GA, Possoit HE, Grames MS, et al. Protein arginine methyltransferases in cardiovascular and neuronal function. Mol Neurobiol. 2020; 57:1716–32.
Article7. Blanc RS, Richard S. Arginine methylation: the coming of age. Mol Cell. 2017; 65:8–24.
Article8. Hwang JW, Cho Y, Bae GU, Kim SN, Kim YK. Protein arginine methyltransferases: promising targets for cancer therapy. Exp Mol Med. 2021; 53:788–808.
Article9. Guccione E, Richard S. The regulation, functions and clinical relevance of arginine methylation. Nat Rev Mol Cell Biol. 2019; 20:642–57.10. Smith E, Zhou W, Shindiapina P, Sif S, Li C, Baiocchi RA. Recent advances in targeting protein arginine methyltransferase enzymes in cancer therapy. Expert Opin Ther Targets. 2018; 22:527–45.
Article11. Baldwin RM, Haghandish N, Daneshmand M, Amin S, Paris G, Falls TJ, et al. Protein arginine methyltransferase 7 promotes breast cancer cell invasion through the induction of MMP9 expression. Oncotarget. 2015; 6:3013–32.
Article12. Jarrold J, Davies CC. PRMTs and arginine methylation: cancer’s best-kept secret? Trends Mol Med. 2019; 25:993–1009.
Article13. Hsu MC, Pan MR, Chu PY, Tsai YL, Tsai CH, Shan YS, et al. Protein arginine methyltransferase 3 enhances chemoresistance in pancreatic cancer by methylating hnRNPA1 to increase ABCG2 expression. Cancers (Basel). 2018; 11:8.
Article14. Hatanaka Y, Tsusaka T, Shimizu N, Morita K, Suzuki T, Machida S, et al. Histone H3 methylated at arginine 17 is essential for reprogramming the paternal genome in zygotes. Cell Rep. 2017; 20:2756–65.15. Waldmann T, Izzo A, Kamieniarz K, Richter F, Vogler C, Sarg B, et al. Methylation of H2AR29 is a novel repressive PRMT6 target. Epigenetics Chromatin. 2011; 4:11.
Article16. Hamey JJ, Rakow S, Bouchard C, Senst JM, Kolb P, Bauer UM, et al. Systematic investigation of PRMT6 substrate recognition reveals broad specificity with a preference for an RG motif or basic and bulky residues. FEBS J. 2021; 288:5668–91.
Article17. Casadio F, Lu X, Pollock SB, LeRoy G, Garcia BA, Muir TW, et al. H3R42me2a is a histone modification with positive transcriptional effects. Proc Natl Acad Sci U S A. 2013; 110:14894–9.
Article18. Fulton MD, Cao M, Ho MC, Zhao X, Zheng YG. The macromolecular complexes of histones affect protein arginine methyltransferase activities. J Biol Chem. 2021; 297:101123.
Article19. Wu Q, Schapira M, Arrowsmith CH, Barsyte-Lovejoy D. Protein arginine methylation: from enigmatic functions to therapeutic targeting. Nat Rev Drug Discov. 2021; 20:509–30.20. Jiang Y, Wei S, Koo JM, Kim HJ, Park W, Zhang Y, et al. Integrative evaluation of the clinical significance underlying protein arginine methyltransferases in hepatocellular carcinoma. Cancers (Basel). 2023; 15:4183.
Article21. Ishimaru T, Ishida J, Kim JD, Mizukami H, Hara K, Hashimoto M, et al. Angiodysplasia in embryo lacking protein arginine methyltransferase 1 in vascular endothelial cells. J Biochem. 2017; 161:255–8.
Article22. Zhu L, He X, Dong H, Sun J, Wang H, Zhu Y, et al. Protein arginine methyltransferase 1 is required for maintenance of normal adult hematopoiesis. Int J Biol Sci. 2019; 15:2763–73.
Article23. Berrens RV, Reik W. Prmt5: a guardian of the germline protects future generations. EMBO J. 2015; 34:689–90.
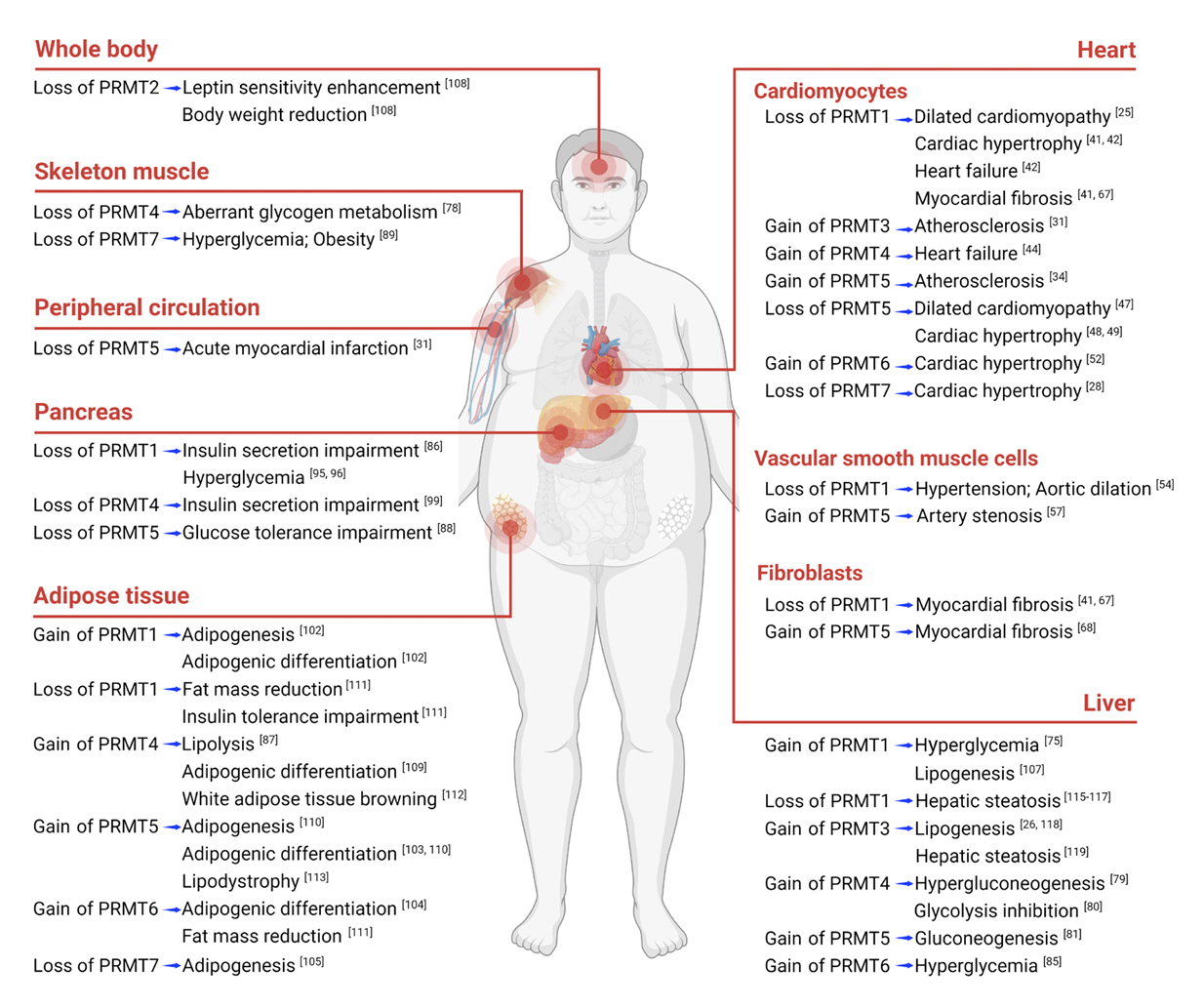
Article24. Tee WW, Pardo M, Theunissen TW, Yu L, Choudhary JS, Hajkova P, et al. Prmt5 is essential for early mouse development and acts in the cytoplasm to maintain ES cell pluripotency. Genes Dev. 2010; 24:2772–7.25. Murata K, Lu W, Hashimoto M, Ono N, Muratani M, Nishikata K, et al. PRMT1 deficiency in mouse juvenile heart induces dilated cardiomyopathy and reveals cryptic alternative splicing products. iScience. 2018; 8:200–13.
Article26. Kim DI, Park MJ, Lim SK, Park JI, Yoon KC, Han HJ, et al. PRMT3 regulates hepatic lipogenesis through direct interaction with LXRα. Diabetes. 2015; 64:60–71.
Article27. Cheng D, Gao G, Di Lorenzo A, Jayne S, Hottiger MO, Richard S, et al. Genetic evidence for partial redundancy between the arginine methyltransferases CARM1 and PRMT6. J Biol Chem. 2020; 295:17060–70.
Article28. Ahn BY, Jeong MH, Pyun JH, Jeong HJ, Vuong TA, Bae JH, et al. PRMT7 ablation in cardiomyocytes causes cardiac hypertrophy and fibrosis through β-catenin dysregulation. Cell Mol Life Sci. 2022; 79:99.
Article29. Zheng S, Zeng C, Huang A, Huang F, Meng A, Wu Z, et al. Relationship between protein arginine methyltransferase and cardiovascular disease (review). Biomed Rep. 2022; 17:90.30. Melikian N, Seddon MD, Casadei B, Chowienczyk PJ, Shah AM. Neuronal nitric oxide synthase and human vascular regulation. Trends Cardiovasc Med. 2009; 19:256–62.
Article31. Chen X, Niroomand F, Liu Z, Zankl A, Katus HA, Jahn L, et al. Expression of nitric oxide related enzymes in coronary heart disease. Basic Res Cardiol. 2006; 101:346–53.
Article32. Tan B, Liu Q, Yang L, Yang Y, Liu D, Liu L, et al. Low expression of PRMT5 in peripheral blood may serve as a potential independent risk factor in assessments of the risk of stable CAD and AMI. BMC Cardiovasc Disord. 2019; 19:31.
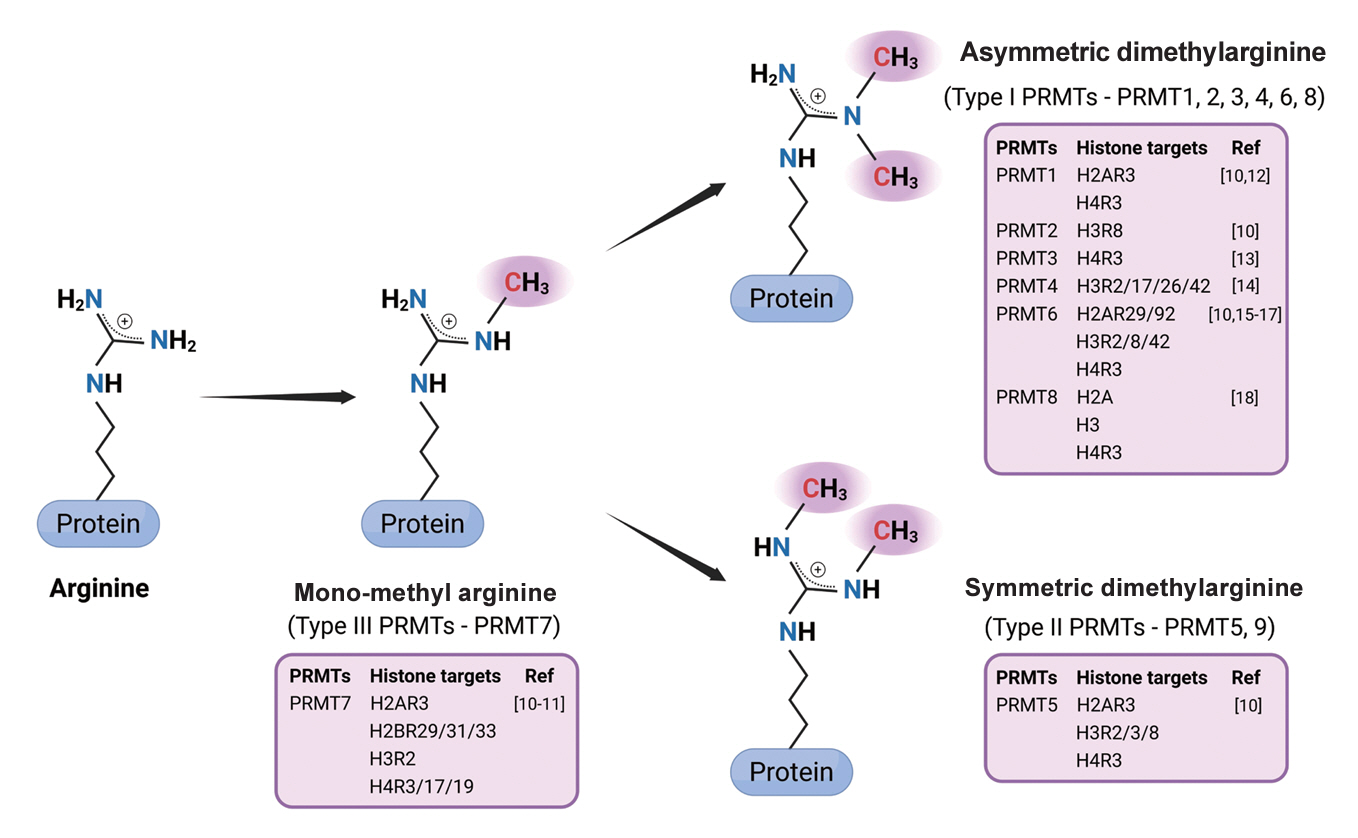
Article33. Fulton MD, Brown T, Zheng YG. The biological axis of protein arginine methylation and asymmetric dimethylarginine. Int J Mol Sci. 2019; 20:3322.
Article34. Harris DP, Chandrasekharan UM, Bandyopadhyay S, Willard B, DiCorleto PE. PRMT5-mediated methylation of NF-κB p65 at Arg174 is required for endothelial CXCL11 gene induction in response to TNF-α and IFN-γ costimulation. PLoS One. 2016; 11:e0148905.35. Zhu N, Guo ZF, Kazama K, Yi B, Tongmuang N, Yao H, et al. Epigenetic regulation of vascular smooth muscle cell phenotypic switch and neointimal formation by PRMT5. Cardiovasc Res. 2023; 119:2244–55.
Article36. Cheng SL, Ramachandran B, Behrmann A, Shao JS, Mead M, Smith C, et al. Vascular smooth muscle LRP6 limits arteriosclerotic calcification in diabetic LDLR-/- mice by restraining noncanonical Wnt signals. Circ Res. 2015; 117:142–56.37. Kim E, Jang J, Park JG, Kim KH, Yoon K, Yoo BC, et al. Protein arginine methyltransferase 1 (PRMT1) selective inhibitor, TC-E 5003, has anti-inflammatory properties in TLR4 signaling. Int J Mol Sci. 2020; 21:3058.
Article38. Clemons GA, Silva AC, Acosta CH, Udo MS, Tesic V, Rodgers KM, et al. Protein arginine methyltransferase 4 modulates nitric oxide synthase uncoupling and cerebral blood flow in Alzheimer’s disease. J Cell Physiol. 2024; 239:e30858.
Article39. Yan S, Hu J, Li J, Wang P, Wang Y, Wang Z. PRMT4 drives post-ischemic angiogenesis via YB1/VEGF signaling. J Mol Med (Berl). 2021; 99:993–1008.40. An X, Lee J, Kim GH, Kim HJ, Pyo HJ, Kwon I, et al. Modulation of IKs channel-PIP2 interaction by PRMT1 plays a critical role in the control of cardiac repolarization. J Cell Physiol. 2022; 237:3069–79.41. Pyun JH, Kim HJ, Jeong MH, Ahn BY, Vuong TA, Lee DI, et al. Cardiac specific PRMT1 ablation causes heart failure through CaMKII dysregulation. Nat Commun. 2018; 9:5107.
Article42. Jeong MH, Jeong HJ, Ahn BY, Pyun JH, Kwon I, Cho H, et al. PRMT1 suppresses ATF4-mediated endoplasmic reticulum response in cardiomyocytes. Cell Death Dis. 2019; 10:903.
Article43. Kim SW, Ahn BY, Tran TT, Pyun JH, Kang JS, Leem YE. PRMT1 suppresses doxorubicin-induced cardiotoxicity by inhibiting endoplasmic reticulum stress. Cell Signal. 2022; 98:110412.
Article44. Wang Y, Ju C, Hu J, Huang K, Yang L. PRMT4 overexpression aggravates cardiac remodeling following myocardial infarction by promoting cardiomyocyte apoptosis. Biochem Biophys Res Commun. 2019; 520:645–50.45. Wang Y, Yan S, Liu X, Deng F, Wang P, Yang L, et al. PRMT4 promotes ferroptosis to aggravate doxorubicin-induced cardiomyopathy via inhibition of the Nrf2/GPX4 pathway. Cell Death Differ. 2022; 29:1982–95.
Article46. Liang Z, Wen C, Jiang H, Ma S, Liu X. Protein arginine methyltransferase 5 functions via interacting proteins. Front Cell Dev Biol. 2021; 9:725301.
Article47. Li Z, Xu J, Song Y, Xin C, Liu L, Hou N, et al. PRMT5 prevents dilated cardiomyopathy via suppression of protein O-GlcNAcylation. Circ Res. 2021; 129:857–71.
Article48. Cai S, Wang P, Xie T, Li Z, Li J, Lan R, et al. Histone H4R3 symmetric di-methylation by Prmt5 protects against cardiac hypertrophy via regulation of Filip1L/β-catenin. Pharmacol Res. 2020; 161:105104.
Article49. Zhang L, Wang H, Li W. PRMT5 up-regulation improves myocardial hypertrophy by mediating E2F-1/NF-κB/NLRP3 pathway. Prev Med. 2023; 172:107553.50. Chen M, Yi B, Sun J. Inhibition of cardiomyocyte hypertrophy by protein arginine methyltransferase 5. J Biol Chem. 2014; 289:24325–35.
Article51. Diao C, Chen Z, Qiu T, Liu H, Yang Y, Liu X, et al. Inhibition of PRMT5 attenuates oxidative stress-induced pyroptosis via activation of the Nrf2/HO-1 signal pathway in a mouse model of renal ischemia-reperfusion injury. Oxid Med Cell Longev. 2019; 2019:2345658.
Article52. Raveendran VV, Al-Haffar K, Kunhi M, Belhaj K, Al-Habeeb W, Al-Buraiki J, et al. Protein arginine methyltransferase 6 mediates cardiac hypertrophy by differential regulation of histone H3 arginine methylation. Heliyon. 2020; 6:e03864.
Article53. Touyz RM, Alves-Lopes R, Rios FJ, Camargo LL, Anagnostopoulou A, Arner A, et al. Vascular smooth muscle contraction in hypertension. Cardiovasc Res. 2018; 114:529–39.
Article54. Pyun JH, Ahn BY, Vuong TA, Kim SW, Jo Y, Jeon J, et al. Inducible Prmt1 ablation in adult vascular smooth muscle leads to contractile dysfunction and aortic dissection. Exp Mol Med. 2021; 53:1569–79.55. Zeng SY, Luo JF, Quan HY, Xiao YB, Liu YH, Lu HQ, et al. Protein arginine methyltransferase 2 inhibits angiotensin II-induced proliferation and inflammation in vascular smooth muscle cells. Biomed Res Int. 2018; 2018:1547452.
Article56. Liu H, Jia K, Ren Z, Sun J, Pan LL. PRMT5 critically mediates TMAO-induced inflammatory response in vascular smooth muscle cells. Cell Death Dis. 2022; 13:299.
Article57. Liu H, Dong X, Jia K, Yuan B, Ren Z, Pan X, et al. Protein arginine methyltransferase 5-mediated arginine methylation stabilizes Kruppel-like factor 4 to accelerate neointimal formation. Cardiovasc Res. 2023; 119:2142–56.
Article58. Souilhol C, Harmsen MC, Evans PC, Krenning G. Endothelial-mesenchymal transition in atherosclerosis. Cardiovasc Res. 2018; 114:565–77.
Article59. Kruger-Genge A, Blocki A, Franke RP, Jung F. Vascular endothelial cell biology: an update. Int J Mol Sci. 2019; 20:4411.60. Raj V, Natarajan S, Marimuthu C, Chatterjee S, Ramasamy M, Ramanujam GM, et al. Cholecalciferol and metformin protect against lipopolysaccharide-induced endothelial dysfunction and senescence by modulating sirtuin-1 and protein arginine methyltransferase-1. Eur J Pharmacol. 2021; 912:174531.
Article61. Cao Y, Fang Y, Mu J, Liu X. High salt medium activates RhoA/ROCK and downregulates eNOS expression via the upregulation of ADMA. Mol Med Rep. 2016; 14:606–12.
Article62. Ye Q, Zhang J, Zhang C, Yi B, Kazama K, Liu W, et al. Endothelial PRMT5 plays a crucial role in angiogenesis after acute ischemic injury. JCI Insight. 2022; 7:e152481.
Article63. Bandyopadhyay S, Harris DP, Adams GN, Lause GE, McHugh A, Tillmaand EG, et al. HOXA9 methylation by PRMT5 is essential for endothelial cell expression of leukocyte adhesion molecules. Mol Cell Biol. 2012; 32:1202–13.
Article64. Chen M, Qu X, Zhang Z, Wu H, Qin X, Li F, et al. Cross-talk between Arg methylation and Ser phosphorylation modulates apoptosis signal-regulating kinase 1 activation in endothelial cells. Mol Biol Cell. 2016; 27:1358–66.65. Travers JG, Kamal FA, Robbins J, Yutzey KE, Blaxall BC. Cardiac fibrosis: the fibroblast awakens. Circ Res. 2016; 118:1021–40.66. Kurose H. Cardiac fibrosis and fibroblasts. Cells. 2021; 10:1716.
Article67. Zhang Z, Ding S, Wang Z, Zhu X, Zhou Z, Zhang W, et al. Prmt1 upregulated by Hdc deficiency aggravates acute myocardial infarction via NETosis. Acta Pharm Sin B. 2022; 12:1840–55.
Article68. Dong XL, Yuan BH, Yu SZ, Liu H, Pan XH, Sun J, et al. Adriamycin induces cardiac fibrosis in mice via PRMT5-mediated cardiac fibroblast activation. Acta Pharmacol Sin. 2023; 44:573–83.
Article69. vanLieshout TL, Ljubicic V. The emergence of protein arginine methyltransferases in skeletal muscle and metabolic disease. Am J Physiol Endocrinol Metab. 2019; 317:E1070–80.70. Han HS, Choi D, Choi S, Koo SH. Roles of protein arginine methyltransferases in the control of glucose metabolism. Endocrinol Metab (Seoul). 2014; 29:435–40.
Article71. Kim DS, Scherer PE. Obesity, diabetes, and increased cancer progression. Diabetes Metab J. 2021; 45:799–812.
Article72. Lee SH, Park SY, Choi CS. Insulin resistance: from mechanisms to therapeutic strategies. Diabetes Metab J. 2022; 46:15–37.
Article73. Asahara SI, Inoue H, Kido Y. Regulation of pancreatic β-cell mass by gene-environment interaction. Diabetes Metab J. 2022; 46:38–48.
Article74. Lee K, Kim H, Lee J, Oh CM, Song H, Kim H, et al. Essential role of protein arginine methyltransferase 1 in pancreas development by regulating protein stability of neurogenin 3. Diabetes Metab J. 2019; 43:649–58.75. Choi D, Oh KJ, Han HS, Yoon YS, Jung CY, Kim ST, et al. Protein arginine methyltransferase 1 regulates hepatic glucose production in a FoxO1-dependent manner. Hepatology. 2012; 56:1546–56.
Article76. Ma Y, Liu S, Jun H, Wang J, Fan X, Li G, et al. A critical role for hepatic protein arginine methyltransferase 1 isoform 2 in glycemic control. FASEB J. 2020; 34:14863–77.
Article77. Porta M, Amione C, Barutta F, Fornengo P, Merlo S, Gruden G, et al. The co-activator-associated arginine methyltransferase 1 (CARM1) gene is overexpressed in type 2 diabetes. Endocrine. 2019; 63:284–92.
Article78. Wang SC, Dowhan DH, Eriksson NA, Muscat GE. CARM1/PRMT4 is necessary for the glycogen gene expression programme in skeletal muscle cells. Biochem J. 2012; 444:323–31.
Article79. Krones-Herzig A, Mesaros A, Metzger D, Ziegler A, Lemke U, Bruning JC, et al. Signal-dependent control of gluconeogenic key enzyme genes through coactivator-associated arginine methyltransferase 1. J Biol Chem. 2006; 281:3025–9.80. Zhong XY, Yuan XM, Xu YY, Yin M, Yan WW, Zou SW, et al. CARM1 methylates GAPDH to regulate glucose metabolism and is suppressed in liver cancer. Cell Rep. 2018; 24:3207–23.
Article81. Tsai WW, Niessen S, Goebel N, Yates JR 3rd, Guccione E, Montminy M. PRMT5 modulates the metabolic response to fasting signals. Proc Natl Acad Sci U S A. 2013; 110:8870–5.
Article82. Wu J, Nagy LE, Wang L. The long and the small collide: LncRNAs and small heterodimer partner (SHP) in liver disease. Mol Cell Endocrinol. 2021; 528:111262.
Article83. Ohn JH, Hwang JY, Moon MK, Ahn HY, Kim HH, Koo YD, et al. Small heterodimer partner (SHP) deficiency protects myocardia from lipid accumulation in high fat diet-fed mice. PLoS One. 2017; 12:e0186021.
Article84. Kanamaluru D, Xiao Z, Fang S, Choi SE, Kim DH, Veenstra TD, et al. Arginine methylation by PRMT5 at a naturally occurring mutation site is critical for liver metabolic regulation by small heterodimer partner. Mol Cell Biol. 2011; 31:1540–50.85. Han HS, Jung CY, Yoon YS, Choi S, Choi D, Kang G, et al. Arginine methylation of CRTC2 is critical in the transcriptional control of hepatic glucose metabolism. Sci Signal. 2014; 7:ra19.
Article86. Iwasaki H. Impaired PRMT1 activity in the liver and pancreas of type 2 diabetic Goto-Kakizaki rats. Life Sci. 2009; 85:161–6.
Article87. Li Y, Peng M, Zeng T, Zheng J, Liao Y, Zhang H, et al. Protein arginine methyltransferase 4 regulates adipose tissue lipolysis in type 1 diabetic mice. Diabetes Metab Syndr Obes. 2020; 13:535–44.88. Ma J, He X, Cao Y, O’Dwyer K, Szigety KM, Wu Y, et al. Islet-specific Prmt5 excision leads to reduced insulin expression and glucose intolerance in mice. J Endocrinol. 2020; 244:41–52.
Article89. Jeong HJ, Lee HJ, Vuong TA, Choi KS, Choi D, Koo SH, et al. Prmt7 deficiency causes reduced skeletal muscle oxidative metabolism and age-related obesity. Diabetes. 2016; 65:1868–82.90. Chen Y, Xu X, Sheng M, Zhang X, Gu Q, Zheng Z. PRMT-1 and DDAHs-induced ADMA upregulation is involved in ROS- and RAS-mediated diabetic retinopathy. Exp Eye Res. 2009; 89:1028–34.
Article91. Kim DI, Park MJ, Choi JH, Kim IS, Han HJ, Yoon KC, et al. PRMT1 and PRMT4 regulate oxidative stress-induced retinal pigment epithelial cell damage in SIRT1-dependent and SIRT1-independent manners. Oxid Med Cell Longev. 2015; 2015:617919.
Article92. Kim DI, Park MJ, Lim SK, Choi JH, Kim JC, Han HJ, et al. Highglucose-induced CARM1 expression regulates apoptosis of human retinal pigment epithelial cells via histone 3 arginine 17 dimethylation: role in diabetic retinopathy. Arch Biochem Biophys. 2014; 560:36–43.
Article93. Chen YY, Peng XF, Liu GY, Liu JS, Sun L, Liu H, et al. Protein arginine methyltranferase-1 induces ER stress and epithelialmesenchymal transition in renal tubular epithelial cells and contributes to diabetic nephropathy. Biochim Biophys Acta Mol Basis Dis. 2019; 1865:2563–75.
Article94. Kim D, Lim S, Park M, Choi J, Kim J, Han H, et al. Ubiquitination-dependent CARM1 degradation facilitates Notch1-mediated podocyte apoptosis in diabetic nephropathy. Cell Signal. 2014; 26:1774–82.95. Kim H, Yoon BH, Oh CM, Lee J, Lee K, Song H, et al. PRMT1 is required for the maintenance of mature β-cell identity. Diabetes. 2020; 69:355–68.
Article96. Lv L, Wang X, Shen J, Cao Y, Zhang Q. MiR-574-3p inhibits glucose toxicity-induced pancreatic β-cell dysfunction by suppressing PRMT1. Diabetol Metab Syndr. 2022; 14:99.
Article97. Lv L, Chen H, Sun J, Lu D, Chen C, Liu D. PRMT1 promotes glucose toxicity-induced β cell dysfunction by regulating the nucleo-cytoplasmic trafficking of PDX-1 in a FOXO1-dependent manner in INS-1 cells. Endocrine. 2015; 49:669–82.
Article98. Bayen S, Saini S, Gaur P, Duraisamy AJ, Kumar Sharma A, Pal K, et al. PRMT1 promotes hyperglycemia in a FoxO1-dependent manner, affecting glucose metabolism, during hypobaric hypoxia exposure, in rat model. Endocrine. 2018; 59:151–63.
Article99. Kim JK, Lim Y, Lee JO, Lee YS, Won NH, Kim H, et al. PRMT4 is involved in insulin secretion via the methylation of histone H3 in pancreatic β cells. J Mol Endocrinol. 2015; 54:315–24.100. An SM, Cho SH, Yoon JC. Adipose tissue and metabolic health. Diabetes Metab J. 2023; 47:595–611.
Article101. Zhang AM, Wellberg EA, Kopp JL, Johnson JD. Hyperinsulinemia in obesity, inflammation, and cancer. Diabetes Metab J. 2021; 45:285–311.
Article102. Zhu Q, Wang D, Liang F, Tong X, Liang Z, Wang X, et al. Protein arginine methyltransferase PRMT1 promotes adipogenesis by modulating transcription factors C/EBPβ and PPARγ. J Biol Chem. 2022; 298:102309.
Article103. LeBlanc SE, Wu Q, Lamba P, Sif S, Imbalzano AN. Promoterenhancer looping at the PPARγ2 locus during adipogenic differentiation requires the Prmt5 methyltransferase. Nucleic Acids Res. 2016; 44:5133–47.
Article104. Hwang JW, So YS, Bae GU, Kim SN, Kim YK. Protein arginine methyltransferase 6 suppresses adipogenic differentiation by repressing peroxisome proliferator-activated receptor γ activity. Int J Mol Med. 2019; 43:2462–70.105. Leem YE, Bae JH, Jeong HJ, Kang JS. PRMT7 deficiency enhances adipogenesis through modulation of C/EBP-β. Biochem Biophys Res Commun. 2019; 517:484–90.
Article106. Hu YJ, Sif S, Imbalzano AN. Prmt7 is dispensable in tissue culture models for adipogenic differentiation. F1000Res. 2013; 2:279.
Article107. Park MJ, Kim DI, Lim SK, Choi JH, Kim JC, Yoon KC, et al. Thioredoxin-interacting protein mediates hepatic lipogenesis and inflammation via PRMT1 and PGC-1α regulation in vitro and in vivo. J Hepatol. 2014; 61:1151–7.
Article108. Iwasaki H, Kovacic JC, Olive M, Beers JK, Yoshimoto T, Crook MF, et al. Disruption of protein arginine N-methyltransferase 2 regulates leptin signaling and produces leanness in vivo through loss of STAT3 methylation. Circ Res. 2010; 107:992–1001.
Article109. Yadav N, Cheng D, Richard S, Morel M, Iyer VR, Aldaz CM, et al. CARM1 promotes adipocyte differentiation by coactivating PPARgamma. EMBO Rep. 2008; 9:193–8.110. LeBlanc SE, Konda S, Wu Q, Hu YJ, Oslowski CM, Sif S, et al. Protein arginine methyltransferase 5 (Prmt5) promotes gene expression of peroxisome proliferator-activated receptor γ2 (PPARγ2) and its target genes during adipogenesis. Mol Endocrinol. 2012; 26:583–97.
Article111. Choi S, Choi D, Lee YK, Ahn SH, Seong JK, Chi SW, et al. Depletion of Prmt1 in adipocytes impairs glucose homeostasis in diet-induced obesity. Diabetes. 2021; 70:1664–78.
Article112. Zhong Y, Wang Y, Li X, Qin H, Yan S, Rao C, et al. PRMT4 facilitates white adipose tissue browning and thermogenesis by methylating PPARγ. Diabetes. 2023; 72:1095–111.
Article113. Jia Z, Yue F, Chen X, Narayanan N, Qiu J, Syed SA, et al. Protein arginine methyltransferase PRMT5 regulates fatty acid metabolism and lipid droplet biogenesis in white adipose tissues. Adv Sci (Weinh). 2020; 7:2002602.
Article114. Rius-Perez S, Torres-Cuevas I, Millan I, Ortega AL, Perez S. PGC-1α, inflammation, and oxidative stress: an integrative view in metabolism. Oxid Med Cell Longev. 2020; 2020:1452696.115. Xu L, Huang Z, Lo TH, Lee JT, Yang R, Yan X, et al. Hepatic PRMT1 ameliorates diet-induced hepatic steatosis via induction of PGC1α. Theranostics. 2022; 12:2502–18.
Article116. Zhao J, Adams A, Weinman SA, Tikhanovich I. Hepatocyte PRMT1 protects from alcohol induced liver injury by modulating oxidative stress responses. Sci Rep. 2019; 9:9111.
Article117. Zhao J, Adams A, Roberts B, O’Neil M, Vittal A, Schmitt T, et al. Protein arginine methyl transferase 1- and Jumonji C domain-containing protein 6-dependent arginine methylation regulate hepatocyte nuclear factor 4 alpha expression and hepatocyte proliferation in mice. Hepatology. 2018; 67:1109–26.
Article118. Nahon JE, Groeneveldt C, Geerling JJ, van Eck M, Hoekstra M. Inhibition of protein arginine methyltransferase 3 activity selectively impairs liver X receptor-driven transcription of hepatic lipogenic genes in vivo. Br J Pharmacol. 2018; 175:3175–83.119. de Jong LM, Zhang Z, den Hartog Y, Sijsenaar TJ, Martins Cardoso R, Manson ML, et al. PRMT3 inhibitor SGC707 reduces triglyceride levels and induces pruritus in Western-type diet-fed LDL receptor knockout mice. Sci Rep. 2022; 12:483.120. Feustel K, Falchook GS. Protein arginine methyltransferase 5 (PRMT5) inhibitors in oncology clinical trials: a review. J Immunother Precis Oncol. 2022; 5:58–67.
Article121. Vieito M, Moreno V, Spreafico A, Brana I, Wang JS, Preis M, et al. Phase 1 study of JNJ-64619178, a protein arginine methyltransferase 5 inhibitor, in advanced solid tumors. Clin Cancer Res. 2023; 29:3592–602.
Article122. Watts JM, Bradley TJ, Thomassen A, Brunner AM, Minden MD, Papadantonakis N, et al. A phase I/II study to investigate the safety and clinical activity of the protein arginine methyltransferase 5 inhibitor GSK3326595 in subjects with myelodysplastic syndrome and acute myeloid leukemia. Blood. 2019; 134(Suppl 1):2656.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Arginine methylation as a key post-translational modification in skeletal muscle homeostasis: a review
- Roles of Protein Arginine Methyltransferases in the Control of Glucose Metabolism
- Identification of Protein Arginine Methyltransferase 5 as a Regulator for Encystation of Acanthamoeba
- Protein methylation in cellular proliferation and differentiation: Non-histone nuclear methyl acceptor protein(s) during 3'-methyl-4-dimethylaminoazobenzeneinduced hepatocarcinogenesis
- Effects of Regular Exercise on the Endothelial Function in Cardiovascular Disease