J Korean Med Sci.
2024 Mar;39(8):e70. 10.3346/jkms.2024.39.e70.
Chromosomal Microarray Analysis in Fetuses With Ultrasonographic Soft Markers: A Meta-Analysis of the Current Evidence
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Seoul National University College of Medicine, Seoul, Korea
- 2Department of Obstetrics and Gynecology, Korea University Guro Hospital, Korea University College of Medicine, Seoul, Korea
- 3Department of Biostatistics, Seoul Metropolitan Government-Seoul National University Hospital Boramae Medical Center, Seoul, Korea
- KMID: 2553324
- DOI: http://doi.org/10.3346/jkms.2024.39.e70
Abstract
- Background
Ultrasonographic soft markers are normal variants, rather than fetal abnormalities, and guidelines recommend a detailed survey of fetal anatomy to determine the necessity of antenatal karyotyping. Anecdotal reports have described cases with ultrasonographic soft markers in which chromosomal microarray analysis (CMA) revealed pathogenic copy number variants (CNVs) despite normal results on conventional karyotyping, but CMA for ultrasonographic soft markers remains a matter of debate. In this systematic review, we evaluated the clinical significance of CMA for pregnancies with isolated ultrasonographic soft markers and a normal fetal karyotype.
Methods
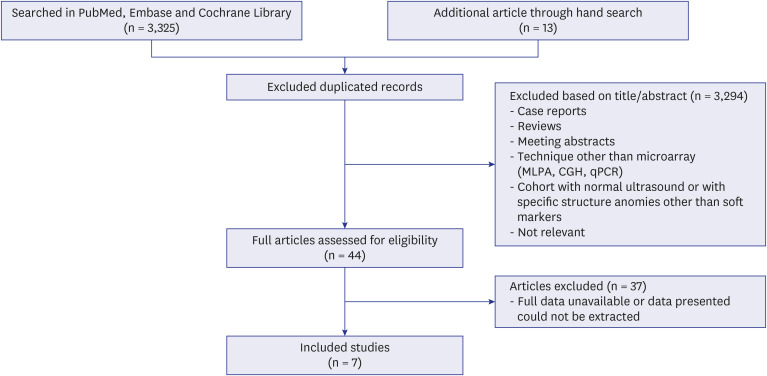
An electronic search was conducted by an experienced librarian through the MEDLINE, Embase, and Cochrane CENTRAL databases. We reviewed 3,338 articles (3,325 identified by database searching and 13 by a hand search) about isolated ultrasonographic soft markers, and seven ultrasonographic markers (choroid plexus cysts, echogenic bowel, echogenic intracardiac focus, hypoplastic nasal bone, short femur [SF], single umbilical artery, and urinary tract dilatation) were included for this study.
Results
Seven eligible articles were included in the final review. Pathogenic or likely pathogenic CNVs were found in fetuses with isolated ultrasonographic soft markers and a normal karyotype. The overall prevalence of pathogenic or likely pathogenic CNVs was 2.0% (41 of 2,048). The diagnostic yield of CMA was highest in fetuses with isolated SF (9 of 225, 3.9%).
Conclusion
CMA could aid in risk assessment and pregnancy counseling in pregnancies where the fetus has isolated ultrasonographic soft markers along with a normal karyotype.
Keyword
Figure
Reference
-
1. Nyberg DA, Souter VL. Sonographic markers of fetal trisomies: second trimester. J Ultrasound Med. 2001; 20(6):655–674. PMID: 11400940.2. Society for Maternal-Fetal Medicine (SMFM). Electronic address: pubs@smfm.org. Prabhu M, Kuller JA, Biggio JR. Society for Maternal-Fetal Medicine consult series #57: evaluation and management of isolated soft ultrasound markers for aneuploidy in the second trimester: (replaces consults #10, single umbilical artery, October 2010; #16, isolated echogenic bowel diagnosed on second-trimester ultrasound, August 2011; #17, evaluation and management of isolated renal pelviectasis on second-trimester ultrasound, December 2011; #25, isolated fetal choroid plexus cysts, April 2013; #27, isolated echogenic intracardiac focus, August 2013). Am J Obstet Gynecol. 2021; 225(4):B2–15.3. Cunningham FG, Leveno KJ, Dashe JS, Hoffman BL, Spong CY, Casey BM. Williams Obstetrics. 26th ed. New York, NY, USA: McGraw-Hill Medical;2022.4. Ko HS, Kwak DW, Oh SY, Choi SK, Hong JS, Hwang HS, et al. Clinical significance of soft markers in second trimester ultrasonography for pregnant Korean women: a multicenter study and literature review. Obstet Gynecol Sci. 2022; 65(2):145–155. PMID: 35184524.5. Bromley B, Lieberman E, Shipp TD, Benacerraf BR. The genetic sonogram: a method of risk assessment for down syndrome in the second trimester. J Ultrasound Med. 2002; 21(10):1087–1096. PMID: 12369663.6. American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics. Committee on Genetics. Society for Maternal-Fetal Medicine. Screening for fetal chromosomal abnormalities: ACOG practice bulletin, number 226. Obstet Gynecol. 2020; 136(4):e48–e69. PMID: 32804883.7. Lichtenbelt KD, Knoers NV, Schuring-Blom GH. From karyotyping to array-CGH in prenatal diagnosis. Cytogenet Genome Res. 2011; 135(3-4):241–250. PMID: 22086062.8. Manning M, Hudgins L. Professional Practice and Guidelines Committee. Array-based technology and recommendations for utilization in medical genetics practice for detection of chromosomal abnormalities. Genet Med. 2010; 12(11):742–745. PMID: 20962661.9. Michelson DJ, Shevell MI, Sherr EH, Moeschler JB, Gropman AL, Ashwal S. Evidence report: genetic and metabolic testing on children with global developmental delay: report of the quality standards subcommittee of the American Academy of Neurology and the practice committee of the Child Neurology Society. Neurology. 2011; 77(17):1629–1635. PMID: 21956720.10. American College of Obstetricians and Gynecologists Committee on Genetics. Committee opinion No. 581: the use of chromosomal microarray analysis in prenatal diagnosis. Obstet Gynecol. 2013; 122(6):1374–1377. PMID: 24264715.11. Norton ME. Callen's Ultrasonography in Obstetrics and Gynecology E-Book: Callen's Ultrasonography in Obstetrics and Gynecology E-Book. Amsterdam, The Netherlands: Elsevier Health Sciences;2016.12. Cai M, Huang H, Su L, Wu X, Xie X, Xu L, et al. Choroid plexus cysts: single nucleotide polymorphism array analysis of associated genetic anomalies and resulting obstetrical outcomes. Risk Manag Healthc Policy. 2021; 14:2491–2497. PMID: 34163268.13. Cai M, Lin N, Chen X, Fu M, Guo N, Xu L, et al. Evaluation of chromosomal abnormalities and copy number variations in fetuses with ultrasonic soft markers. BMC Med Genomics. 2021; 14:19. PMID: 33435955.14. Huang H, Cai M, Ma W, Lin N, Xu L. Chromosomal microarray analysis for the prenatal diagnosis in fetuses with nasal bone hypoplasia: a retrospective cohort study. Risk Manag Healthc Policy. 2021; 14:1533–1540. PMID: 33889037.15. Hu T, Tian T, Zhang Z, Wang J, Hu R, Xiao L, et al. Prenatal chromosomal microarray analysis in 2466 fetuses with ultrasonographic soft markers: a prospective cohort study. Am J Obstet Gynecol. 2021; 224(5):516.e1–516.16.16. Singer A, Maya I, Koifman A, Nasser Samra N, Baris HN, Falik-Zaccai T, et al. Microarray analysis in pregnancies with isolated echogenic bowel. Early Hum Dev. 2018; 119:25–28. PMID: 29522884.17. Moczulska H, Serafin M, Wojda K, Borowiec M, Sieroszewski P. Fetal nasal bone hypoplasia in the second trimester as a marker of multiple genetic syndromes. J Clin Med. 2022; 11(6):1513. PMID: 35329839.18. Shi X, Lu J, Li L, Wei R, Wu J. Prenatal chromosomal microarray analysis in foetuses with isolated absent or hypoplastic nasal bone. Ann Med. 2022; 54(1):1297–1302. PMID: 35506821.19. Chang H, Li L, Li M, Xiao X. Rare and common variants at 16p11.2 are associated with schizophrenia. Schizophr Res. 2017; 184:105–108. PMID: 27889382.20. D’Angelo D, Lebon S, Chen Q, Martin-Brevet S, Snyder LG, Hippolyte L, et al. Defining the effect of the 16p11.2 duplication on cognition, behavior, and medical comorbidities. JAMA Psychiatry. 2016; 73(1):20–30. PMID: 26629640.21. Maillard AM, Ruef A, Pizzagalli F, Migliavacca E, Hippolyte L, Adaszewski S, et al. The 16p11.2 locus modulates brain structures common to autism, schizophrenia and obesity. Mol Psychiatry. 2015; 20(1):140–147. PMID: 25421402.22. Steinman KJ, Spence SJ, Ramocki MB, Proud MB, Kessler SK, Marco EJ, et al. 16p11.2 deletion and duplication: characterizing neurologic phenotypes in a large clinically ascertained cohort. Am J Med Genet A. 2016; 170(11):2943–2955. PMID: 27410714.23. Maya I, Sharony R, Yacobson S, Kahana S, Yeshaya J, Tenne T, et al. When genotype is not predictive of phenotype: implications for genetic counseling based on 21,594 chromosomal microarray analysis examinations. Genet Med. 2018; 20(1):128–131. PMID: 28726807.24. Hassfurther A, Komini E, Fischer J, Leipoldt M. Clinical and genetic heterogeneity of the 15q13. 3 microdeletion syndrome. Mol Syndromol. 2016; 6(5):222–228. PMID: 26997942.25. Pizzo L, Andrieux J, Amor DJ, Girirajan S. Clinical utility gene card for: 16p12.2 microdeletion. Eur J Hum Genet. 2017; 25(2):271.26. Kirov G, Rees E, Walters JT, Escott-Price V, Georgieva L, Richards AL, et al. The penetrance of copy number variations for schizophrenia and developmental delay. Biol Psychiatry. 2014; 75(5):378–385. PMID: 23992924.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prenatal chromosomal microarray analysis of fetus with increased nuchal translucency
- Usefulness of Chromosomal Microarray in Hematologic Malignancies: A Case of Aggressive NK-cell Leukemia with 1q Abnormality
- The availability of ultrasonography as the method for early detection of fetal chromosomal abnormalities
- Clinical application of chromosomal microarray for pathogenic genomic imbalance in fetuses with increased nuchal translucency but normal karyotype
- An Introduction of the Systematic Review and Meta-Analysis